Revolutionizing Bio-Based Monomer Production: Continuous Green Synthesis of Furandicarboxylic Acid
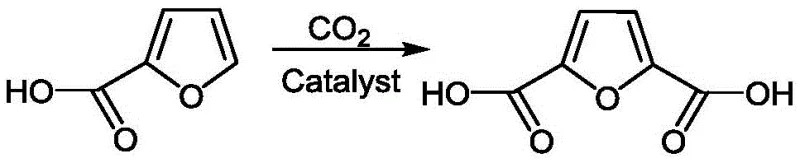
The global shift towards sustainable bio-based materials has intensified the search for efficient alternatives to petroleum-derived monomers, with 2,5-Furandicarboxylic Acid (FDCA) emerging as a cornerstone molecule for next-generation polyesters and polyamides. A significant breakthrough in this domain is documented in Chinese Patent CN111153877B, which discloses a novel method for the continuous green synthesis of furandicarboxylic acid utilizing furoic acid and carbon dioxide. This technology represents a paradigm shift from traditional batch processes to a sophisticated continuous flow system, addressing critical bottlenecks in atom economy and process scalability. By leveraging a fixed-bed reactor loaded with transition metal supported catalysts, the invention achieves high conversion rates under remarkably mild conditions, typically ranging from 70°C to 100°C at atmospheric pressure. For R&D directors and procurement strategists seeking a reliable FDCA supplier, this patent outlines a pathway that not only simplifies the synthetic route but also aligns perfectly with modern environmental, social, and governance (ESG) mandates by utilizing CO2 as a C1 building block.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of FDCA has been dominated by the oxidation of 5-hydroxymethylfurfural (HMF), a route that, while chemically viable, presents substantial engineering challenges for large-scale implementation. The HMF pathway often necessitates harsh reaction conditions, including elevated temperatures exceeding 165°C and high-pressure oxygen environments, which impose severe safety risks and increase energy consumption significantly. Furthermore, the separation of the final product from the catalyst and reaction byproducts in batch systems is notoriously difficult, leading to lower overall yields and higher purification costs. Alternative routes, such as the cyclization of hexaric acid or acylation of furan, suffer from poor atom economy, multi-step complexities, and the generation of stoichiometric waste streams that complicate downstream processing. These inherent limitations in conventional methodologies create a volatile supply chain for high-purity furandicarboxylic acid, making it difficult for downstream polymer manufacturers to secure consistent quality and volume.
The Novel Approach
In stark contrast, the methodology described in patent CN111153877B introduces a streamlined continuous process that directly carbonylates furoic acid using carbon dioxide, effectively bypassing the instability issues associated with HMF intermediates. This approach utilizes a fixed-bed reactor configuration where the catalyst is immobilized, eliminating the need for complex filtration steps to remove homogeneous catalysts from the product stream. The process operates under near-ambient pressure and moderate temperatures, drastically reducing the energy footprint and equipment stress compared to high-pressure oxidation methods. Moreover, the integration of a vacuum rectification tower allows for the immediate separation of the product while recycling unreacted starting materials back into the reactor inlet. This closed-loop design ensures that the commercial scale-up of complex bio-based monomers becomes technically feasible, offering a robust solution for cost reduction in polymer intermediate manufacturing by maximizing raw material utilization and minimizing waste disposal costs.
Mechanistic Insights into Transition Metal-Catalyzed Carbonylation
The core of this innovative synthesis lies in the precise interaction between the furoic acid substrate and carbon dioxide mediated by a specialized transition metal supported catalyst. The reaction mechanism involves the activation of the C-H bond on the furan ring, followed by the insertion of CO2 to form the second carboxyl group, effectively transforming furoic acid into FDCA. The catalyst system, typically comprising metals such as Palladium, Nickel, Rhodium, or Copper supported on molecular sieves like 4A, Y-type, or ZSM-5, provides the necessary active sites for this transformation while maintaining structural integrity under continuous flow conditions. The choice of support material is critical, as it influences the dispersion of the metal active centers and the diffusion of reactants within the pore structure, directly impacting the turnover frequency and selectivity of the reaction. By optimizing the metal-to-support weight ratio between 1.0:100 and 1.6:100, the process ensures maximum catalytic efficiency without leaching of metal species into the product stream.
Impurity control is inherently managed through the continuous nature of the fixed-bed reactor and the subsequent vacuum distillation step. Unlike batch processes where side reactions can accumulate over time, the steady-state operation of the fixed-bed reactor maintains consistent reaction parameters, minimizing the formation of degradation products or oligomers. The vacuum distillation unit operates at a top temperature of approximately 100°C and a pressure of 0.01MPa, effectively separating the high-boiling FDCA product from the volatile solvent and unreacted gases. Any unreacted furoic acid that vaporizes during this stage is condensed in a recovery tank and pumped back to the reactor inlet for secondary reaction. This rigorous recycling mechanism not only boosts the overall yield to over 95% but also ensures that the final product meets stringent purity specifications of ≥99.0%, which is essential for applications in high-performance polymers and pharmaceutical intermediates where trace impurities can compromise material properties.
How to Synthesize Furandicarboxylic Acid Efficiently
To implement this synthesis route effectively, operators must adhere to a precise sequence of preparation and reaction steps that leverage the continuous flow architecture. The process begins with the preparation of the feed solution, where furoic acid is dissolved in a suitable solvent such as ethylene glycol dimethyl ether, dioxane, or acetonitrile at a specific solid-liquid ratio. This solution is then mixed with carbon dioxide gas before entering the heated fixed-bed reactor containing the pre-activated catalyst. Detailed standard operating procedures regarding catalyst activation, flow rate calibration, and temperature zoning are critical for maintaining the stability of the reaction over extended periods.
- Dissolve furoic acid in a solvent such as ethylene glycol dimethyl ether to create a feed solution.
- Mix the feed solution with carbon dioxide at a molar ratio of 1: 1.5 to 1:2.5 and introduce into a fixed-bed reactor containing a transition metal supported catalyst.
- Maintain reaction temperature between 70°C and 100°C at atmospheric pressure, then separate product via vacuum distillation while recycling unreacted materials.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this continuous green synthesis technology offers transformative benefits that extend far beyond simple chemical yield improvements. The ability to utilize carbon dioxide, an abundant and low-cost industrial byproduct, as a primary reactant fundamentally alters the cost structure of FDCA production, decoupling it from the volatility of expensive oxidants or complex sugar-derived precursors. The continuous fixed-bed design facilitates 24/7 operation with minimal downtime for catalyst changeovers or cleaning, ensuring a steady and predictable output that stabilizes supply chains against market fluctuations. Furthermore, the elimination of heavy metal contamination risks and the reduction of hazardous waste streams simplify regulatory compliance and lower the total cost of ownership for manufacturing facilities. These factors collectively contribute to reducing lead time for high-purity polymer intermediates and enhance the overall resilience of the supply network.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the substitution of expensive oxidizing agents with carbon dioxide and the implementation of a continuous recycling loop. By recovering and reusing unreacted furoic acid and solvents, the process minimizes raw material loss, leading to substantial cost savings in variable production expenses. Additionally, the mild reaction conditions reduce energy consumption for heating and pressurization, while the heterogeneous catalyst system eliminates the need for costly metal scavenging steps post-reaction. These efficiencies compound to create a highly competitive cost profile for the final product.
- Enhanced Supply Chain Reliability: The continuous nature of the fixed-bed reactor system ensures a consistent throughput of product, mitigating the risks associated with batch-to-batch variability that often plague traditional chemical manufacturing. The robustness of the transition metal supported catalysts allows for extended run times without significant loss of activity, reducing the frequency of maintenance shutdowns. This reliability is crucial for downstream customers who require just-in-time delivery of critical monomers for their own polymerization processes, thereby strengthening long-term supplier-buyer relationships.
- Scalability and Environmental Compliance: Scaling this technology from pilot to commercial production is straightforward due to the modular nature of fixed-bed reactors, allowing capacity to be increased by numbering up rather than scaling up vessel size. The process generates minimal waste, as the only major byproduct is water or unreacted gases which are recycled, aligning with strict environmental regulations and sustainability goals. This green chemistry profile not only reduces disposal costs but also enhances the brand value of the end-products by certifying them as sustainably sourced.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous synthesis of furandicarboxylic acid, derived directly from the experimental data and claims of the underlying patent literature. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this material into their existing supply chains.
Q: What are the primary advantages of this continuous FDCA synthesis method over traditional HMF routes?
A: This method utilizes mild reaction conditions (70-100°C, atmospheric pressure) compared to the harsh conditions often required for HMF oxidation. Furthermore, the continuous fixed-bed design allows for superior process integration and easier catalyst separation, significantly enhancing production efficiency and reducing operational complexity.
Q: Which catalysts are most effective for this carbonylation process?
A: The patent specifies transition metal supported catalysts, particularly those using Palladium (Pd), Nickel (Ni), Rhodium (Rh), or Copper (Cu) supported on molecular sieves like 4A, Y-type, or oxides such as Al2O3 and SiO2. These catalysts demonstrate high activity and stability under the described continuous flow conditions.
Q: How does the recycling loop impact the overall yield of furandicarboxylic acid?
A: The process incorporates a vacuum distillation step where unreacted furoic acid and solvent are condensed and recycled back into the reactor. This closed-loop system ensures that raw materials are utilized efficiently, driving the total yield of furandicarboxylic acid to exceed 95%, which is critical for commercial viability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furandicarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the continuous green synthesis route for FDCA and have invested heavily in mastering similar advanced catalytic technologies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most stringent purity specifications. Our rigorous QC labs are equipped to analyze complex impurity profiles, guaranteeing that every batch of high-purity furandicarboxylic acid delivered adheres to the exacting standards required for bio-based polymer applications. We are committed to bridging the gap between innovative patent chemistry and reliable industrial supply.
We invite forward-thinking organizations to collaborate with us to optimize their supply chains and reduce manufacturing costs through advanced process chemistry. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your product development timelines while ensuring supply continuity.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
