Advanced Asymmetric Synthesis of Optically Active Nicotine for Commercial Scale-up and High Purity
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce high-value alkaloids, and the recent disclosure in Chinese patent CN111233829A presents a significant breakthrough in the preparation of optically active nicotine. This innovative method addresses the critical limitations of traditional extraction and synthetic routes by employing a sophisticated asymmetric catalytic reduction of imine salt derivatives. Unlike conventional processes that rely on harsh conditions or expensive precursors, this technology utilizes common metal catalysts and chiral ligands to achieve high synthesis yields and exceptional chemical purity under mild thermal conditions. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of nicotine intermediates. The ability to control stereochemistry precisely through ligand selection offers a robust solution for producing specific enantiomers required for advanced pharmaceutical applications and high-end electronic cigarette formulations. This report analyzes the technical depth and commercial viability of this patent to demonstrate its value as a reliable nicotine intermediate supplier strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of nicotine has been dominated by extraction from tobacco plants or synthetic routes that suffer from significant economic and technical drawbacks. As detailed in the background of the patent, traditional extraction methods are heavily influenced by external factors such as raw material availability, climate conditions, and growth cycles, leading to inconsistent supply chains and potential health hazards from tobacco-specific impurities. On the synthetic side, prior art methods often rely on expensive starting materials like 3-bromopyridine and require harsh reaction conditions involving butyl lithium, which poses severe safety risks and complicates industrial scale-up. Furthermore, existing synthetic routes frequently produce racemic mixtures, necessitating costly and inefficient resolution steps that discard up to 50% of the material, such as the tartrate resolution process. These inefficiencies result in high production costs, low overall yields, and significant waste generation, making them unsuitable for the demanding requirements of modern cost reduction in fine chemical manufacturing.
The Novel Approach
The novel approach disclosed in patent CN111233829A fundamentally reengineers the synthesis pathway by utilizing imine salt derivatives as the primary precursors for asymmetric reduction. This method eliminates the need for expensive halogenated pyridine starting materials and avoids the use of dangerous organolithium reagents, instead employing common reducing agents and metal catalysts that are readily available in the global chemical market. The reaction conditions are remarkably mild, typically operating within a temperature range of -10 to 50°C, which significantly reduces energy consumption and simplifies reactor engineering requirements. By directly synthesizing the optically active form through chiral catalysis, the process bypasses the need for resolution steps, theoretically doubling the atom economy compared to racemic synthesis followed by separation. This streamlined workflow not only enhances the chemical purity of the final product but also drastically simplifies the downstream processing, offering a compelling value proposition for supply chain heads focused on reducing lead time for high-purity nicotine.
Mechanistic Insights into Asymmetric Transfer Hydrogenation
The core of this technological advancement lies in the precise orchestration of chiral ligands and metal centers to facilitate enantioselective reduction. The process begins with the in situ formation of an active organic metal catalyst, where a chiral ligand containing nitrogen or phosphorus coordinates with a rhodium or ruthenium metal derivative. The structural versatility of these ligands, ranging from TsDPEN derivatives to complex ferrocenyl phosphines, allows for fine-tuning of the steric and electronic environment around the metal center. This tailored environment is crucial for discriminating between the prochiral faces of the imine salt substrate during the hydride transfer step. The catalytic cycle involves the activation of the reducing agent, such as formic acid or potassium formate, followed by the stereoselective delivery of hydrogen to the imine bond. This mechanism ensures that the resulting nicotine molecule possesses the desired optical configuration, whether it be the (R) or (S) enantiomer, depending on the chirality of the ligand employed.
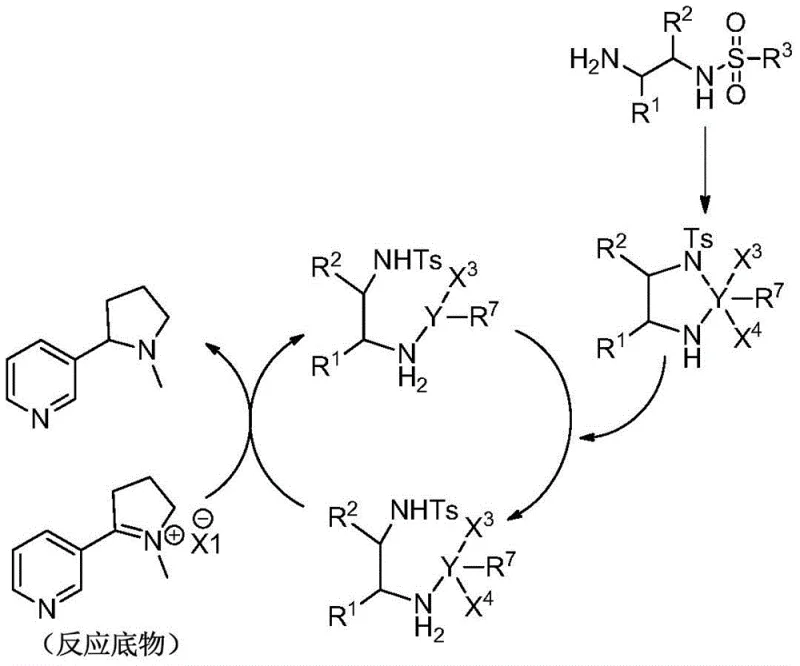
Impurity control is inherently built into this mechanistic design through the specificity of the catalytic interaction and the mildness of the reaction conditions. Harsh conditions often promote side reactions such as over-reduction or decomposition of sensitive functional groups, but the controlled temperature range of -10 to 50°C minimizes these thermal degradation pathways. Furthermore, the use of specific imine salt derivatives ensures that the reaction proceeds through a well-defined intermediate, reducing the formation of complex by-product mixtures that are difficult to separate. The quenching step, which involves the addition of an alkaline solution to terminate the reduction, is also optimized to prevent further reaction of the product with excess reducing agent. This careful management of reaction kinetics and thermodynamics results in a crude product with high chemical purity, reducing the burden on purification units and ensuring that the final isolated nicotine meets stringent purity specifications required for pharmaceutical and consumer applications.
How to Synthesize Optically Active Nicotine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for translating this laboratory-scale innovation into a robust manufacturing process. The procedure involves three critical stages: catalyst preparation, asymmetric reduction, and product isolation, each designed to maximize efficiency and safety. The initial step requires the precise mixing of the chiral ligand and metal catalyst in a suitable organic solvent, allowing sufficient time for the active catalytic species to form before the introduction of the substrate. This pre-formation step is vital for ensuring consistent catalytic activity and reproducibility across different batches. Following this, the imine salt derivative and reducing agent are introduced under controlled thermal conditions, where the reaction progress is monitored to ensure complete conversion without compromising enantiomeric excess. The detailed standardized synthesis steps see the guide below.
- Prepare the organic metal catalyst by reacting a chiral nitrogen or phosphorus ligand with a rhodium or ruthenium derivative in an organic solvent at -10 to 50°C.
- Add the imine salt derivative precursor and a reducing agent to the catalyst mixture, maintaining the temperature between -10 to 50°C for 0.5 to 20 hours to effect reduction.
- Quench the reaction with an alkaline solution, extract the product using an organic solvent, and concentrate to isolate high-purity optically active nicotine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers transformative benefits that extend beyond mere technical feasibility. The shift from extraction or complex multi-step synthesis to a direct catalytic reduction significantly streamlines the supply chain by reducing the number of unit operations and raw material dependencies. By utilizing common chemical substances for catalysts and reducing agents, the method mitigates the risk of supply disruptions associated with specialized or scarce reagents. This reliability is crucial for maintaining continuous production schedules and meeting the fluctuating demands of the global market. Furthermore, the mild reaction conditions reduce the need for specialized high-pressure or cryogenic equipment, lowering capital expenditure and operational costs associated with plant maintenance and safety compliance. These factors collectively contribute to a more resilient and cost-efficient supply chain architecture.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven by the elimination of expensive starting materials and the avoidance of wasteful resolution steps. Traditional methods that produce racemic mixtures inherently discard half of the synthesized material during chiral separation, effectively doubling the raw material cost per unit of active product. By achieving high enantiomeric excess directly through asymmetric catalysis, this process maximizes atom economy and reduces the overall consumption of raw materials. Additionally, the use of common reducing agents and solvents minimizes procurement costs, while the mild conditions lower energy consumption for heating and cooling. These cumulative efficiencies result in substantial cost savings without compromising on the quality or purity of the final nicotine product.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly enhanced by the use of readily available and stable raw materials. Unlike tobacco extraction, which is subject to agricultural variability and seasonal constraints, this synthetic route relies on industrial chemicals that can be sourced consistently throughout the year. The robustness of the catalyst system also contributes to process stability, reducing the likelihood of batch failures or deviations that can disrupt delivery schedules. Furthermore, the scalability of the reaction allows for flexible production volumes, enabling manufacturers to respond quickly to changes in market demand. This agility ensures that customers receive their orders on time, fostering stronger long-term partnerships and trust in the supply relationship.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with modern green chemistry principles and regulatory requirements. The reduction in waste generation, particularly the avoidance of large amounts of resolving agents and by-products, simplifies waste treatment processes and lowers disposal costs. The mild operating conditions also reduce the carbon footprint of the manufacturing process by minimizing energy usage. As regulatory scrutiny on chemical manufacturing intensifies, adopting cleaner and more efficient technologies becomes a strategic advantage. This process is designed for commercial scale-up of complex alkaloids, ensuring that production can be expanded to meet industrial volumes while maintaining compliance with environmental standards and safety regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate and reliable information for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production lines or for sourcing high-quality nicotine intermediates from external partners. The insights provided here cover aspects of stereocontrol, raw material selection, and process optimization to ensure a comprehensive understanding of the technology's capabilities.
Q: What are the advantages of using imine salt derivatives for nicotine synthesis?
A: Using imine salt derivatives as precursors allows for mild reaction conditions near normal temperature, avoiding the harsh conditions and expensive starting materials like 3-bromopyridine found in conventional methods, thereby reducing overall production costs.
Q: How is optical purity controlled in this asymmetric reduction process?
A: Optical purity is controlled by the specific configuration of the chiral ligand employed; for instance, using an S-configuration ligand yields R-nicotine, while an R-configuration ligand yields S-nicotine, with ee values reaching up to 95% in optimized examples.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes common chemical substances for catalysts and reducing agents and operates under mild conditions, which simplifies safety protocols and equipment requirements, making it highly convenient for large-scale industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Nicotine Supplier
The technical potential of this asymmetric reduction pathway underscores the importance of partnering with a CDMO expert capable of navigating complex chemical transformations. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our team is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of optically active nicotine meets the highest standards of quality and consistency. We understand the critical nature of chiral purity in pharmaceutical and specialty chemical applications and have the infrastructure to support the precise control required for such sensitive syntheses.
We invite you to engage with our technical procurement team to discuss how this technology can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments to verify the compatibility of this method with your product specifications. Our commitment to transparency and technical excellence ensures that you receive the support needed to make informed sourcing decisions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
