Advanced Asymmetric Synthesis of Levo-Nicotine for Commercial Scale-Up and High Purity Standards
Advanced Asymmetric Synthesis of Levo-Nicotine for Commercial Scale-Up and High Purity Standards
The global demand for high-purity chiral alkaloids has intensified, particularly for levo-nicotine, a critical component in pharmaceutical research and advanced agrochemical formulations. Traditional supply chains relying on tobacco extraction face inherent limitations regarding purity consistency and the presence of carcinogenic minor alkaloids. A groundbreaking technical solution detailed in patent CN114702474A introduces a robust, fully synthetic pathway that circumvents these biological constraints. This method leverages advanced asymmetric catalysis to deliver levo-nicotine with an optical purity as high as 99.7% e.e. and a total synthesis yield ranging from 60% to 70%. For R&D directors and procurement strategists, this represents a paradigm shift from variable agricultural sourcing to precise, controllable chemical manufacturing, ensuring a reliable levo-nicotine supplier status for downstream applications requiring stringent quality specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial acquisition of levo-nicotine has been dominated by extraction from natural tobacco sources or resolution of racemic synthetic mixtures, both of which present significant technical and commercial bottlenecks. Natural extraction is inherently plagued by variability; the alkaloid profile of tobacco leaves fluctuates based on climate, soil composition, and harvest timing, leading to batch-to-batch inconsistency that complicates downstream formulation. Furthermore, natural nicotine typically contains a complex matrix of related alkaloids that are structurally similar and difficult to separate, often resulting in final purities below 95% and introducing potential health risks associated with tobacco-specific nitrosamines. On the synthetic side, prior art routes often rely on resolution techniques where half of the material is discarded as the unwanted enantiomer, theoretically capping the yield at 50% and generating substantial chemical waste. Other synthetic approaches utilize hazardous reagents such as butyllithium at cryogenic temperatures of -78°C, creating severe safety hazards and high energy costs that are untenable for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The innovative methodology disclosed in the reference patent fundamentally restructures the synthesis logic by employing a direct asymmetric hydrogenation strategy. Instead of building the chiral center through resolution or dangerous lithiation, this route installs the stereochemistry early in the sequence using a tailored chiral catalyst system. This approach allows for the conversion of achiral starting materials directly into the desired levo-isomer with exceptional efficiency. By bypassing the resolution step, the process theoretically doubles the material efficiency compared to classical resolution methods, drastically simplifying the mass balance and reducing raw material consumption. The operation conditions are significantly milder, utilizing hydrogenation pressures between 0.5 to 2.0 MPa and temperatures ranging from 30°C to 40°C, which are far more compatible with standard stainless steel reactor infrastructure found in modern multipurpose plants. This transition from extreme conditions to moderate catalytic processes enhances the commercial scale-up of complex alkaloids by lowering the barrier to entry for safe, large-volume production.
Mechanistic Insights into Asymmetric Hydrogenation and Cyclization
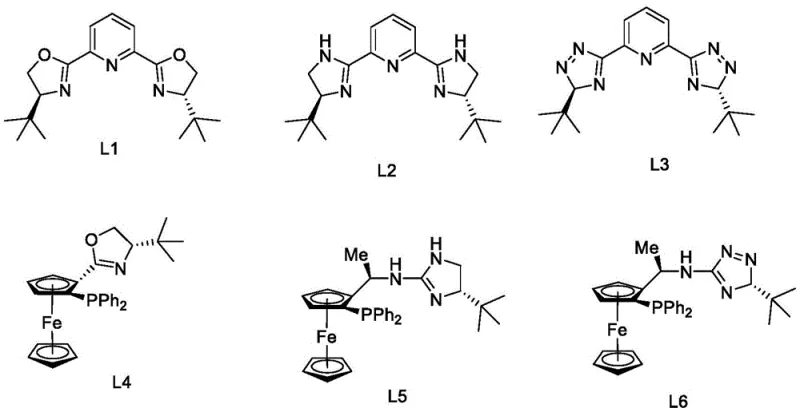
The core technological breakthrough lies in the stereoselective reduction of the ketone precursor (Formula I) to the chiral alcohol intermediate (Formula II). This transformation is mediated by an in situ generated catalyst system comprising a transition metal source, such as Ni(COD)2 or Rh(COD)Cl2, coordinated with a specialized chiral ligand. The patent highlights a series of pyridine-bis(oxazoline) or ferrocene-based ligands (L1-L6) that create a specific chiral environment around the metal center. During the hydrogenation cycle, the substrate coordinates to the metal, and the bulky substituents on the ligand sterically direct the approach of the hydride species to one specific face of the carbonyl group. This precise spatial control ensures that the resulting alcohol possesses the (S)-configuration required for levo-nicotine, achieving enantiomeric excess values exceeding 99.5% e.e. in optimized examples. The ability to tune the ligand structure allows chemists to maximize turnover numbers and selectivity, providing a robust mechanism for impurity control at the molecular level.
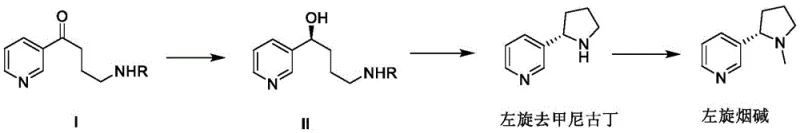
Following the asymmetric reduction, the chiral alcohol undergoes a cyclization reaction to form the pyrrolidine ring, yielding levo-nornicotine. This step involves activation of the hydroxyl group, typically via sulfonylation, followed by intramolecular nucleophilic substitution. The mild basic conditions used in this stage prevent racemization of the sensitive chiral center established in the first step, preserving the high optical purity throughout the sequence. Finally, the secondary amine is methylated using a reductive amination protocol with formaldehyde and formic acid, a classic Eschweiler-Clarke type modification that is highly efficient and atom-economical. The entire sequence is designed to minimize purification burdens; for instance, the intermediate alcohol can be precipitated directly from the reaction mixture by adding non-polar solvents like n-hexane, avoiding energy-intensive distillation steps until the final product isolation. This streamlined workflow demonstrates how high-purity levo-nicotine can be manufactured with reduced solvent usage and simplified work-up procedures.
How to Synthesize Levo-Nicotine Efficiently
The implementation of this synthesis route requires careful attention to catalyst preparation and reaction parameter control to ensure reproducibility at scale. The process begins with the in situ formation of the active catalyst species under an inert atmosphere, followed by the introduction of hydrogen gas to drive the reduction of the ketone substrate. Subsequent steps involve precise pH control during the cyclization and methylation phases to manage the solubility of the amine intermediates and facilitate clean phase separations. While the general chemistry is robust, the specific stoichiometry, temperature profiles, and solvent choices detailed in the patent are critical for achieving the reported 99.7% purity. For technical teams looking to adopt this technology, adhering to the standardized operating procedures is essential for maintaining the integrity of the chiral information throughout the multi-step sequence. The detailed standardized synthesis steps see the guide below.
- Perform asymmetric hydrogenation of the ketone precursor (Formula I) using a chiral ligand-metal catalyst complex to generate the chiral alcohol intermediate (Formula II) with high enantiomeric excess.
- Execute a base-mediated cyclization reaction on the chiral alcohol intermediate, followed by acidification and neutralization to isolate levo-nornicotine.
- Conduct N-methylation of levo-nornicotine using a formaldehyde-formic acid system, followed by extraction and distillation to obtain pure levo-nicotine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthetic route offers compelling advantages over traditional extraction methods, primarily centered around supply security and cost structure optimization. By decoupling production from agricultural cycles, manufacturers can guarantee continuous availability of levo-nicotine regardless of seasonal tobacco harvest fluctuations or geopolitical trade barriers affecting raw leaf exports. The synthetic pathway utilizes commodity chemicals that are widely available in the global market, reducing the risk of supply chain disruptions associated with single-source natural extracts. Furthermore, the elimination of the resolution step means that nearly all raw material carbon atoms are directed towards the desired product, significantly reducing the effective cost of goods sold. This efficiency translates into substantial cost savings for procurement managers who are tasked with managing budgets for high-value fine chemical intermediates without compromising on quality specifications.
- Cost Reduction in Manufacturing: The economic model of this process is superior because it avoids the inherent 50% material loss associated with chiral resolution of racemates. In traditional resolution, half of the synthesized material becomes waste or requires costly recycling, whereas this asymmetric method produces the target isomer directly. Additionally, the avoidance of cryogenic conditions (-78°C) and pyrophoric reagents like butyllithium reduces energy consumption and eliminates the need for specialized, expensive containment equipment. The use of common solvents like ethyl acetate and toluene further lowers operational expenditures compared to processes requiring exotic or highly regulated solvents. These factors combine to create a leaner manufacturing cost structure that allows for more competitive pricing in the marketplace.
- Enhanced Supply Chain Reliability: Supply chain heads benefit from the predictability of a fully synthetic route. Agricultural products are susceptible to pests, droughts, and regulatory changes regarding pesticide residues, all of which can cause sudden spikes in price or shortages. A chemical synthesis performed in a controlled reactor environment mitigates these external risks, ensuring reducing lead time for high-purity pharmaceutical intermediates. The scalability of the hydrogenation step is well-understood in the industry, allowing for seamless transition from pilot kilogram batches to multi-ton annual production capacities. This reliability is crucial for long-term contracts where consistent quality and delivery timelines are paramount for maintaining customer trust and production schedules.
- Scalability and Environmental Compliance: Environmental regulations are becoming increasingly stringent, particularly regarding waste disposal and solvent emissions. This process generates significantly less waste compared to resolution methods, as there is no mother liquor containing the unwanted enantiomer to dispose of. The catalytic nature of the key step means that metal loading can be optimized to minimize heavy metal residue in the final product, simplifying the purification process and ensuring compliance with strict ICH guidelines for elemental impurities. The overall E-factor of the process is improved, aligning with green chemistry principles and making it easier to obtain environmental permits for large-scale manufacturing facilities. This sustainability profile enhances the brand value of the final product in markets that prioritize eco-friendly sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of synthetic levo-nicotine. These answers are derived from the specific technical disclosures and advantageous effects outlined in the patent literature, providing clarity on the feasibility and benefits of this advanced manufacturing route. Understanding these details helps stakeholders make informed decisions about integrating this material into their supply chains.
Q: What is the optical purity achievable with this new synthesis method?
A: The patented asymmetric hydrogenation process consistently achieves an optical purity of up to 99.7% e.e., significantly surpassing traditional extraction methods which often struggle to exceed 95% purity without extensive purification.
Q: How does this method improve supply chain stability compared to plant extraction?
A: Unlike plant extraction which is subject to agricultural variables like climate and harvest cycles, this fully synthetic route utilizes readily available chemical raw materials, ensuring consistent production capacity and eliminating seasonal supply fluctuations.
Q: Does the process avoid the use of hazardous reagents found in prior art?
A: Yes, this method avoids the use of highly flammable organic lithium reagents and cryogenic conditions (-78°C) required in older synthetic routes, thereby enhancing operational safety and reducing specialized equipment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levo-Nicotine Supplier
The technical potential of this asymmetric hydrogenation route is immense, offering a clear path to high-purity materials that meet the rigorous demands of the pharmaceutical and fine chemical industries. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such sophisticated chemistry to the market. Our facility is equipped with state-of-the-art high-pressure hydrogenation reactors and stringent purity specifications are maintained through our rigorous QC labs, ensuring that every batch of levo-nicotine meets the 99.7% optical purity benchmark. We understand the critical nature of chiral intermediates in drug development and are committed to delivering materials that accelerate your research and production timelines without compromise.
We invite you to discuss how this optimized synthesis route can benefit your specific project requirements. Our team is prepared to provide a Customized Cost-Saving Analysis that compares this synthetic approach against your current sourcing methods. Please contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to secure your supply chain with a product that defines excellence in chiral synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
