Advanced Synthesis of Naphthalimide Derivative NETDE for High-Performance pH Sensing Applications
The landscape of functional electronic materials is constantly evolving, driven by the demand for highly sensitive and selective sensing mechanisms. Patent CN102775348A introduces a significant advancement in this field with the synthesis of a novel naphthalimide derivative, specifically designed to function as a robust pH fluorescent molecular switch. This technology addresses critical limitations in current sensing materials by offering a unique fluorescence response profile that is immune to interference from common metal ions. For R&D directors and procurement specialists in the electronic chemical sector, understanding the underlying chemistry of this derivative is essential for integrating high-performance sensing capabilities into next-generation devices. The patent outlines a precise two-step synthetic pathway that balances high yield with operational simplicity, making it a viable candidate for commercial adoption. By leveraging this specific molecular architecture, manufacturers can achieve superior detection limits in complex aqueous environments, a capability that is increasingly vital for applications ranging from biological imaging to environmental monitoring systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional pH fluorescent probes often suffer from significant drawbacks that limit their utility in complex industrial and scientific applications. Many existing naphthalimide-based sensors exhibit a monotonic decrease in fluorescence intensity as pH increases, which can lead to ambiguity in signal interpretation, particularly in dynamic environments where precise threshold detection is required. Furthermore, a pervasive issue in the field is the susceptibility of these probes to interference from coexisting metal ions such as copper, zinc, or iron, which can quench fluorescence or cause false positives. This lack of selectivity necessitates extensive sample purification or the use of masking agents, adding layers of complexity and cost to the analytical process. Additionally, conventional synthesis routes for high-performance fluorophores frequently rely on harsh reaction conditions or expensive catalysts that are difficult to remove, posing challenges for both environmental compliance and final product purity. These limitations create a bottleneck for supply chain managers seeking reliable, high-volume sources of sensing materials that do not require extensive downstream processing.
The Novel Approach
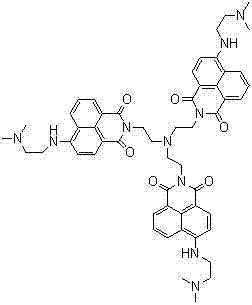
The methodology described in patent CN102775348A represents a paradigm shift by introducing a tri-branched naphthalimide structure that demonstrates a distinct 'OFF-ON-OFF' switching behavior. This non-monotonic response allows for precise identification of specific pH windows, significantly enhancing the reliability of data acquisition in critical applications. The synthetic strategy employs a stepwise condensation and substitution process that avoids the use of transition metal catalysts, thereby eliminating the risk of heavy metal contamination in the final product. This metal-free approach not only simplifies the purification workflow but also aligns with stringent environmental regulations regarding waste disposal and product safety. By utilizing readily available starting materials like 4-bromo-1,8-naphthalic anhydride and tris(2-aminoethyl)amine, the process ensures a stable supply chain foundation. The resulting derivative maintains its fluorescence switching characteristics even in the presence of high concentrations of interfering ions, providing a level of robustness that is essential for deployment in real-world industrial settings where sample matrices are rarely pure.
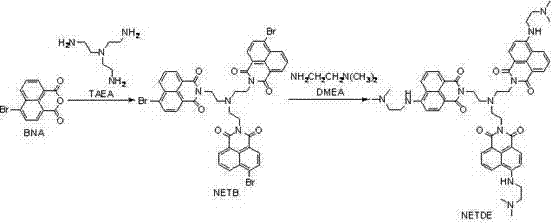
Mechanistic Insights into Naphthalimide Fluorophore Construction
The core of this technology lies in the strategic construction of the naphthalimide fluorophore, which serves as the signal transducer in the molecular switch. The synthesis begins with the nucleophilic attack of tris(2-aminoethyl)amine on the anhydride group of the naphthalene derivative, forming an imide linkage that is crucial for the molecule's photophysical properties. This reaction is carefully controlled in absolute ethanol at moderate temperatures to ensure complete conversion while minimizing side reactions that could lead to polymeric by-products. The resulting intermediate acts as a scaffold, holding three fluorophore units in a specific spatial arrangement that facilitates intramolecular interactions. This tri-branched architecture is key to the enhanced fluorescence quantum yield and the specific Stokes shift observed in the final product. For technical teams, understanding this mechanistic pathway is vital for troubleshooting potential scale-up issues, as the stoichiometry and solvent choice directly influence the crystallinity and purity of the intermediate. The precise control over the imide formation ensures that the electronic structure of the naphthalene core remains intact, preserving its ability to absorb and emit light efficiently.
Following the formation of the scaffold, the final substitution step introduces the pH-sensitive amine groups at the 4-position of the naphthalimide ring. This modification is critical as it alters the electron-donating capability of the system in response to protonation and deprotonation events. The reaction with N,N-dimethylethylenediamine is conducted under solvent-free or high-concentration conditions to drive the equilibrium towards the product, achieving high conversion rates without the need for excessive solvent recovery. The mechanism involves a nucleophilic aromatic substitution where the bromine atom is displaced by the amine, a process that is facilitated by the electron-withdrawing nature of the imide group. This specific substitution pattern creates the 'OFF-ON-OFF' profile by modulating the internal charge transfer (ICT) state of the molecule. From a quality control perspective, this step requires careful monitoring to ensure that all bromine sites are substituted, as incomplete reaction would lead to a mixture of products with varying fluorescence properties. The robust nature of this chemical transformation ensures that the final derivative possesses the structural integrity needed for long-term stability in sensing applications.
How to Synthesize Naphthalimide Derivative NETDE Efficiently
The synthesis of the NETDE derivative is a streamlined process designed for reproducibility and high yield, making it suitable for both laboratory research and commercial production. The procedure involves two distinct reaction stages, each optimized to maximize the purity of the intermediates and the final product. The initial condensation step requires precise temperature control and stoichiometric balance to form the tris-imide intermediate, which is then rigorously purified to remove any unreacted anhydride. This attention to detail in the early stages is crucial, as impurities carried forward can significantly impact the fluorescence performance of the final sensor. The subsequent substitution reaction leverages the reactivity of the intermediate to introduce the pH-sensitive moieties, completing the molecular assembly. For process engineers, this route offers a clear pathway to scale-up, as it relies on standard unit operations such as filtration, washing, and recrystallization. Detailed standardized synthesis steps see the guide below.
- Condense 4-bromo-1,8-naphthalic anhydride with tris(2-aminoethyl)amine in absolute ethanol at 60-78°C to form the intermediate NETB.
- Purify the intermediate NETB via silica gel column chromatography and hot acetone washing to ensure high purity before the next step.
- React the purified NETB with excess N,N-dimethylethylenediamine at 75-95°C for 24-72 hours to yield the final NETDE derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical performance. The elimination of expensive transition metal catalysts from the process significantly reduces the raw material costs and simplifies the supply chain logistics, as there is no need to source or recover precious metals. This metal-free synthesis also drastically reduces the environmental burden associated with heavy metal waste disposal, aligning with global sustainability goals and reducing regulatory compliance costs. The use of common solvents like ethanol and the ability to perform the final step under solvent-minimized conditions further contribute to cost reduction in specialty chemical manufacturing by lowering solvent purchase and recovery expenses. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-separate by-products, which enhances the overall throughput of the production line. These factors combine to create a manufacturing process that is not only economically efficient but also resilient to fluctuations in the availability of specialized reagents.
- Cost Reduction in Manufacturing: The synthetic pathway described in the patent avoids the use of costly palladium or other transition metal catalysts, which are often required for similar coupling reactions in fine chemical synthesis. By relying on nucleophilic substitution and condensation reactions using abundant amines and anhydrides, the direct material costs are significantly lowered. Additionally, the purification process utilizes standard silica gel chromatography and hot washing techniques, which are far less capital-intensive than high-pressure liquid chromatography or complex distillation setups often needed for less selective reactions. This streamlined approach allows for substantial cost savings in both operational expenditure and capital investment, making the final fluorescent probe more competitive in the market. The high yields reported in the patent embodiments further ensure that raw material utilization is optimized, minimizing waste and maximizing the output per batch.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as 4-bromo-1,8-naphthalic anhydride and various alkyl amines, are commodity chemicals with well-established global supply chains. This reduces the risk of supply disruptions that are common with proprietary or highly specialized reagents. The robustness of the reaction conditions, which tolerate moderate temperature variations and do not require inert atmospheres or ultra-dry conditions, means that production can be maintained consistently across different manufacturing sites. For supply chain planners, this translates to reduced lead time for high-purity electronic chemicals, as the process is less susceptible to delays caused by equipment failure or reagent scarcity. The stability of the intermediate and final products also allows for flexible inventory management, enabling manufacturers to stockpile key materials without significant degradation risks.
- Scalability and Environmental Compliance: The process is inherently scalable, moving from gram-scale laboratory synthesis to multi-ton commercial production without fundamental changes to the chemistry. The absence of toxic heavy metals simplifies the waste treatment process, as the effluent primarily consists of organic solvents and salts that can be managed through standard incineration or biological treatment methods. This ease of waste management ensures compliance with increasingly strict environmental regulations, avoiding potential fines or production stoppages. The energy requirements for the reaction are moderate, with temperatures ranging from 60°C to 95°C, which can be easily achieved using standard steam heating systems found in most chemical plants. This energy efficiency contributes to a lower carbon footprint for the manufacturing process, appealing to environmentally conscious clients and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this naphthalimide derivative. These answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this material into your specific product lines. The information covers aspects of synthesis, purification, and performance characteristics that are critical for decision-making.
Q: What is the primary advantage of the NETDE derivative over traditional pH probes?
A: The NETDE derivative exhibits a unique 'OFF-ON-OFF' fluorescence switching behavior within a narrow pH range, offering higher sensitivity and selectivity compared to monotonic probes, while remaining unaffected by coexisting metal ions.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the synthesis utilizes common solvents like ethanol and mild reaction temperatures (60-95°C), avoiding extreme conditions or expensive transition metal catalysts, which facilitates straightforward commercial scale-up.
Q: What are the purity specifications for the intermediate NETB?
A: The intermediate NETB is purified through silica gel column chromatography and hot acetone washing, ensuring the removal of unreacted anhydride and by-products before the final substitution step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthalimide Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into commercially viable products. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand that the integrity of the fluorescent switch mechanism depends on stringent purity specifications, which is why our rigorous QC labs employ advanced analytical techniques to verify every batch. Our commitment to quality ensures that the naphthalimide derivatives we supply meet the exacting standards required for high-performance electronic and sensing applications. By partnering with us, you gain access to a supply chain that is both robust and responsive, capable of meeting the dynamic demands of the global market.
We invite you to collaborate with us to optimize your supply chain for advanced functional materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to reach out to request specific COA data and route feasibility assessments to verify how our manufacturing capabilities align with your project goals. Let us help you secure a reliable source of high-quality naphthalimide derivatives that will drive the success of your next-generation sensing technologies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
