Commercializing Advanced Naphthalimide Derivatives for Robust pH Sensing Applications
Commercializing Advanced Naphthalimide Derivatives for Robust pH Sensing Applications
The landscape of fluorescent sensing materials is undergoing a significant transformation driven by the demand for higher selectivity and environmental stability in complex media. Patent CN102775348B introduces a groundbreaking naphthalimide derivative designed specifically to function as a highly reliable pH fluorescent molecular switch. This innovation addresses the critical limitations of traditional probes which often suffer from signal interference due to the presence of ubiquitous metal ions in aqueous solutions. The core of this technology lies in a unique tri-branched molecular architecture that incorporates three 1,8-naphthalimide fluorophores linked through a central amine core. 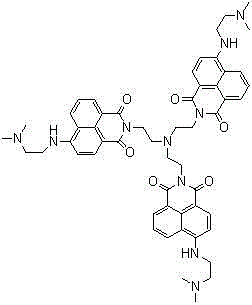 . This structural complexity allows for sophisticated photophysical behaviors, specifically an 'OFF-ON-OFF' switching mechanism that provides a distinct advantage over monotonic sensors. For R&D directors and procurement specialists in the fine chemical sector, this represents a viable pathway to developing next-generation diagnostic tools and smart materials that maintain fidelity in harsh industrial or biological environments.
. This structural complexity allows for sophisticated photophysical behaviors, specifically an 'OFF-ON-OFF' switching mechanism that provides a distinct advantage over monotonic sensors. For R&D directors and procurement specialists in the fine chemical sector, this represents a viable pathway to developing next-generation diagnostic tools and smart materials that maintain fidelity in harsh industrial or biological environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of pH-sensitive fluorescent probes has been plagued by the issue of cross-sensitivity, where the presence of background metal ions drastically alters the fluorescence signal. Traditional 1,8-naphthalimide derivatives, while possessing high quantum yields and good photostability, typically exhibit a monotonic decrease or increase in fluorescence intensity relative to pH changes. This 'acid-ON/alkali-OFF' or vice versa behavior is often insufficient for precise quantification in samples containing mixed ionic species. Furthermore, many existing synthesis routes rely on harsh conditions or expensive transition metal catalysts that introduce impurities difficult to remove, thereby compromising the purity required for high-end electronic or pharmaceutical applications. The reliance on single-fluorophore systems also limits the dynamic range and the ability to create ratiometric sensors that can self-calibrate against environmental fluctuations. These technical bottlenecks have historically increased the cost of quality control and reduced the reliability of sensor batches in commercial production settings.
The Novel Approach
The novel approach detailed in the patent data leverages a multi-step condensation strategy to create a sterically hindered, tri-branched structure that inherently rejects interference from common cations. By utilizing 4-bromo-1,8-naphthalic anhydride as the starting building block, the synthesis ensures a robust naphthalene core that is chemically stable under varying pH conditions. The introduction of substituted amino groups at the 4-position and the imide nitrogen creates a push-pull electronic system that is highly sensitive to protonation states but indifferent to metal coordination. This design results in a material that demonstrates a sharp, reversible fluorescence transition within a very narrow pH window, specifically around pH 5.05. Such precision is invaluable for applications requiring detection of slight acidity changes, such as in cellular imaging or monitoring industrial fermentation processes. The synthetic route avoids complex catalytic cycles, relying instead on thermal condensation in common solvents, which significantly simplifies the manufacturing process and enhances batch-to-batch consistency.
Mechanistic Insights into Tri-branched Naphthalimide Cyclization
The synthesis mechanism involves a sequential nucleophilic substitution that builds molecular complexity while maintaining high atom economy. The first stage involves the reaction of 4-bromo-1,8-naphthalic anhydride with tris(2-aminoethyl)amine in absolute ethanol. This step forms the central hub of the molecule, creating an intermediate known as NETB. The reaction conditions are mild, typically ranging from 40°C to 78°C, which prevents the degradation of the sensitive anhydride ring while ensuring complete conversion of the amine. 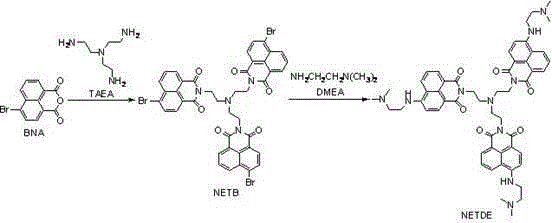 . The second stage involves reacting this intermediate with a large excess of N,N-dimethylethylenediamine. This excess acts both as a reactant and a solvent, driving the equilibrium towards the final product NETDE. The mechanism relies on the displacement of the bromine atom and the opening of any remaining anhydride functionalities to form stable imide bonds. This two-step process is highly efficient, yielding an orange solid that exhibits the desired photophysical properties without the need for chromatographic purification in the final step, merely requiring precipitation and washing.
. The second stage involves reacting this intermediate with a large excess of N,N-dimethylethylenediamine. This excess acts both as a reactant and a solvent, driving the equilibrium towards the final product NETDE. The mechanism relies on the displacement of the bromine atom and the opening of any remaining anhydride functionalities to form stable imide bonds. This two-step process is highly efficient, yielding an orange solid that exhibits the desired photophysical properties without the need for chromatographic purification in the final step, merely requiring precipitation and washing.
Impurity control in this synthesis is achieved through the strategic selection of solvents and precipitation techniques rather than expensive purification columns. The intermediate NETB is purified via silica gel column chromatography using a specific mobile phase ratio of methanol to chloroform, ensuring that unreacted anhydride or mono-substituted byproducts are removed before the final step. In the final conversion to NETDE, the addition of deionized water to the reaction mixture causes the product to precipitate out of the organic phase selectively. This phase separation technique effectively washes away soluble amine salts and polar byproducts. The resulting material shows consistent fluorescence spectra with minimal batch variation, a critical factor for supply chain managers who require predictable performance metrics. The structural integrity of the final molecule is confirmed by the absence of interfering peaks in spectroscopic analysis, indicating a clean reaction profile that minimizes downstream processing costs.
How to Synthesize Naphthalimide Derivative Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize yield and purity. The process begins with the dispersion of the anhydride in anhydrous ethanol, followed by the dropwise addition of the tri-amine to control exothermic heat. Detailed standard operating procedures regarding mixing speeds, heating ramps, and filtration protocols are essential for scaling this from gram to kilogram quantities. The following guide outlines the critical operational parameters derived from the patent examples to ensure successful replication in a pilot plant environment.
- Condense 4-bromo-1,8-naphthalic anhydride with tris(2-aminoethyl)amine in absolute ethanol at 40-78°C to form the intermediate NETB.
- React the purified NETB intermediate with excess N,N-dimethylethylenediamine at 75-95°C for 24-72 hours.
- Precipitate the final orange solid product by adding deionized water, followed by filtration and washing to obtain high-purity NETDE.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages due to its reliance on commodity chemicals and straightforward unit operations. The primary starting materials, such as 4-bromo-1,8-naphthalic anhydride and various ethylenediamines, are widely available in the global chemical market, reducing the risk of supply chain disruptions associated with exotic reagents. The elimination of transition metal catalysts means there is no need for expensive metal scavenging steps or rigorous testing for heavy metal residues, which significantly lowers the cost of goods sold. Furthermore, the use of ethanol and water as primary solvents aligns with green chemistry principles, reducing the environmental burden and costs associated with hazardous waste disposal. These factors combine to create a manufacturing process that is not only economically efficient but also resilient to regulatory changes regarding solvent usage and emissions.
- Cost Reduction in Manufacturing: The process achieves cost optimization by eliminating the need for precious metal catalysts and complex purification infrastructure. By utilizing a large excess of one reactant as the solvent, the process reduces the volume of auxiliary solvents required, leading to lower recovery and distillation costs. The high yield observed in the final precipitation step minimizes material loss, ensuring that the theoretical mass balance is closely met in practical production. Additionally, the mild reaction temperatures reduce energy consumption compared to high-pressure or cryogenic alternatives. These cumulative efficiencies translate into a competitive pricing structure for the final fluorescent probe, making it accessible for high-volume applications in consumer electronics or agricultural monitoring.
- Enhanced Supply Chain Reliability: The reliance on bulk chemical intermediates ensures that production can be scaled rapidly without waiting for specialized custom synthesis lead times. Since the reaction does not depend on air-sensitive reagents or inert atmosphere gloveboxes, it can be performed in standard stainless steel reactors available in most fine chemical facilities. This flexibility allows for multi-vendor sourcing of raw materials, mitigating the risk of single-source dependency. The robustness of the chemical structure also implies a long shelf life for the finished product, reducing inventory write-offs and allowing for strategic stockpiling during periods of low demand. This stability is crucial for maintaining continuous supply to downstream sensor manufacturers.
- Scalability and Environmental Compliance: The synthesis is inherently scalable because it avoids exothermic runaways and uses solvents with well-established recovery protocols. The wastewater generated is primarily aqueous with dissolved amines, which can be treated using standard biological or chemical oxidation methods common in industrial parks. The solid waste consists mainly of spent silica from the intermediate step, which can be managed through established hazardous waste channels or regenerated. By designing the process with environmental compliance in mind, manufacturers can avoid costly fines and expedite regulatory approvals for new production lines. This forward-thinking approach ensures long-term viability in markets with strict environmental, social, and governance (ESG) mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this naphthalimide derivative. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. They serve to clarify the operational boundaries and performance expectations for potential partners and technical evaluators.
Q: Does this naphthalimide derivative suffer from metal ion interference?
A: No, unlike conventional probes, this derivative exhibits switching behavior that is not significantly affected by coexisting metal ions such as Na+, K+, Ca2+, and transition metals, ensuring high selectivity for hydrogen ions.
Q: What is the fluorescence response profile of this material?
A: The material displays a unique 'OFF-ON-OFF' switching phenomenon within a narrow pH range, specifically showing a sharp increase in fluorescence intensity between pH 4.12 and 5.05, followed by a decrease, allowing for precise ratiometric sensing.
Q: Are the raw materials for this synthesis commercially scalable?
A: Yes, the synthesis utilizes readily available commodity chemicals such as 4-bromo-1,8-naphthalic anhydride and simple amines, avoiding rare catalysts and facilitating large-scale manufacturing without supply chain bottlenecks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthalimide Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating laboratory breakthroughs into commercially viable products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless. We understand that for applications like pH sensing, stringent purity specifications are non-negotiable, which is why our rigorous QC labs employ advanced spectroscopic and chromatographic methods to verify every batch. We are committed to delivering high-purity naphthalimide derivatives that meet the exacting standards of the electronic and specialty chemical industries.
We invite you to collaborate with us to optimize your supply chain for advanced fluorescent materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for your next project. By partnering with us, you gain access to a supply chain that prioritizes quality, consistency, and technical support.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
