Advanced Continuous Flow Synthesis of 3-Chloro-4-Oxoacetic Acid Amyl Ester for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, safer, and cost-effective methods for producing critical intermediates. A significant breakthrough in this domain is documented in patent CN112979461B, which details a fully continuous flow preparation method for 3-chloro-4-oxoacetic acid amyl ester. This compound serves as a vital intermediate in the synthesis of Vitamin B1, finding extensive applications across medicine, agrochemicals, and dye manufacturing sectors. The traditional production methods have long been plagued by inefficiencies, safety concerns, and environmental burdens, creating a pressing need for technological innovation. The patented continuous flow micro-reaction system addresses these challenges by integrating micro-mixers, micro-channel reactors, and continuous extraction separators into a seamless production line. This approach not only drastically shortens reaction times but also enhances product yield and purity while minimizing energy consumption. For global procurement teams and R&D directors, understanding the implications of this technology is crucial for securing a reliable supply chain of high-purity pharmaceutical intermediates. The shift from batch to continuous processing represents a paradigm shift in how complex organic molecules are manufactured at scale.
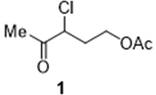
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-chloro-4-oxoacetic acid amyl ester has relied on traditional batch kettle reactors, which suffer from inherent limitations in heat and mass transfer. Previous literature and patent backgrounds indicate that conventional methods often utilize acetic anhydride and toxic catalysts like pyridine, leading to complex post-reaction purification steps and significant waste generation. For instance, earlier processes reported yields ranging from 71% to 86%, with reaction times extending up to 4-6 hours per batch. These batch processes are not only time-consuming but also pose significant safety risks due to the accumulation of reactive intermediates and the handling of hazardous chlorine gas in large volumes. The inability to precisely control reaction parameters in a large vessel often results in inconsistent product quality and the formation of unwanted by-products. Furthermore, the use of stoichiometric amounts of expensive reagents like acetic anhydride drives up the raw material costs, making the final product less competitive in the global market. The environmental footprint of these batch processes is also considerable, requiring extensive treatment of acidic and organic waste streams before disposal.
The Novel Approach
In stark contrast, the novel continuous flow method outlined in the patent leverages the superior physics of micro-reactor technology to overcome these traditional bottlenecks. By utilizing a micro-reaction system composed of sequentially connected micro-mixers and micro-channel reactors, the process achieves a level of precision unattainable in batch systems. The continuous chlorination of acetylbutyrolactone with chlorine gas is performed in a controlled micro-environment, ensuring complete conversion of the substrate while minimizing the residence time of hazardous intermediates. Subsequently, the acylation step is carried out using a mixture of glacial acetic acid, hydrochloric acid, and water, eliminating the need for expensive acetic anhydride. This substitution alone represents a significant cost reduction in fine chemical manufacturing. The continuous nature of the process allows for a space-time yield that is magnitudes higher than batch processing, effectively reducing the reactor volume required for the same output. The integration of continuous quenching and extraction further streamlines the workflow, enabling the direct isolation of the product with high purity. This holistic approach to process design ensures that the production of this critical intermediate is not only more efficient but also inherently safer and more environmentally sustainable.
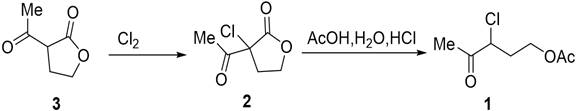
Mechanistic Insights into Continuous Flow Chlorination and Acylation
The core of this technological advancement lies in the precise control of reaction kinetics and thermodynamics within the micro-channels. In the first stage, acetylbutyrolactone reacts with chlorine gas to form alpha-acetyl-alpha-chloro-gamma-butyrolactone. In a micro-channel reactor, the high surface-to-volume ratio facilitates rapid heat dissipation, which is critical for exothermic chlorination reactions. The patent specifies that the temperature in the micro mixer can be controlled within the range of -20°C to 50°C, with a preferred range of 0°C to 50°C, ensuring that the reaction proceeds without thermal runaway. The residence time in the micro-channel reactor is remarkably short, typically between 0.1 to 30 minutes, yet it achieves 100% conversion of the substrate. This efficiency is due to the enhanced mixing at the micro-scale, which eliminates concentration gradients that often lead to side reactions in batch reactors. The molar ratio of acetylbutyrolactone to chlorine is tightly controlled, typically between 1:1 and 1:3, preventing the formation of poly-chlorinated by-products. This level of control is essential for maintaining the integrity of the molecular structure and ensuring the downstream acylation proceeds smoothly.
Following chlorination, the intermediate undergoes acylation in a second micro-reaction system. The mechanism involves the ring-opening of the butyrolactone moiety and subsequent esterification with acetic acid. The patent highlights that this step is performed at temperatures between 50°C and 150°C, with a residence time of 10 to 80 minutes. The use of a micro-mixer ensures that the intermediate is instantly mixed with the acetic acid solution, initiating the reaction uniformly throughout the fluid stream. The continuous flow setup allows for the precise adjustment of the molar ratio between the intermediate and acetic acid, typically maintained between 1:2 and 1:8, to drive the equilibrium towards the product. The subsequent quenching with an inorganic base, such as sodium carbonate or sodium hydroxide, neutralizes the acidic components and stops the reaction instantly. This immediate quenching prevents the degradation of the product, which is a common issue in batch processes where cooling takes time. The final continuous extraction step utilizes the immiscibility of the organic product and the aqueous phase to separate the target molecule efficiently, resulting in a product purity of over 96% directly from the reaction stream.
How to Synthesize 3-Chloro-4-Oxoacetic Acid Amyl Ester Efficiently
Implementing this synthesis route requires a specialized setup that integrates fluid dynamics with chemical engineering principles. The process begins with the precise metering of acetylbutyrolactone and chlorine gas into the first micro-channel reactor. Operators must ensure that the flow rates are calibrated to maintain the desired molar ratio, as deviations can impact the conversion efficiency. The reaction mixture is then transferred to a buffer tank before entering the acylation stage, where it is mixed with the acid solution. Temperature control is paramount throughout the system, with heating or cooling modules applied to the micro-channels to maintain the optimal reaction window. The final separation stage involves a continuous extraction separator, which can be a plate-type or membrane-based unit, to isolate the organic phase. The organic solvent, such as methylene dichloride or ethyl acetate, is chosen based on its extraction efficiency and ease of removal. The entire system is designed to be automated, reducing the need for manual intervention and minimizing the risk of human error. For detailed standard operating procedures and equipment specifications, please refer to the technical guide below.
- Simultaneously convey acetylbutyrolactone and chlorine gas into a microchannel reactor for continuous chlorination at controlled temperatures between 0°C and 50°C to form the alpha-acetyl-alpha-chloro-gamma-butyrolactone intermediate.
- Mix the chlorinated intermediate with a solution of glacial acetic acid, hydrochloric acid, and water in a micromixer, then pass through a second microchannel reactor at 50°C to 150°C for continuous acylation.
- Quench the reaction mixture with an inorganic base solution in a third micromixer, followed by continuous extraction and separation using an organic solvent to isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this continuous flow technology translates into tangible strategic advantages beyond mere technical metrics. The shift from batch to continuous processing fundamentally alters the cost structure of manufacturing this intermediate. By eliminating the need for expensive reagents like acetic anhydride and toxic catalysts, the raw material costs are significantly reduced. Furthermore, the high space-time yield means that the same production capacity can be achieved with much smaller equipment footprints, lowering capital expenditure and facility maintenance costs. The automation of the process reduces labor intensity and the number of operators required, contributing to substantial cost savings in production. From a supply chain reliability perspective, the continuous nature of the process ensures a steady output of product, reducing the lead time for high-purity pharmaceutical intermediates. Unlike batch processes that produce in discrete lots with downtime between runs, the flow system can operate continuously for extended periods, ensuring supply continuity for downstream manufacturers. This reliability is crucial for companies managing just-in-time inventory systems.
- Cost Reduction in Manufacturing: The process eliminates the use of costly acetic anhydride, replacing it with glacial acetic acid, which is significantly cheaper and more readily available. Additionally, the high conversion rates and selectivity minimize the loss of raw materials, ensuring that every kilogram of input contributes maximally to the final output. The reduction in waste generation also lowers the costs associated with waste treatment and disposal, further enhancing the economic viability of the process. The energy efficiency of micro-reactors, due to their superior heat transfer capabilities, results in lower utility costs compared to the heating and cooling demands of large batch kettles.
- Enhanced Supply Chain Reliability: The continuous flow system is inherently scalable through a multi-channel parallel amplification strategy, allowing manufacturers to ramp up production quickly in response to market demand without the long lead times associated with building new batch facilities. The robustness of the automated system reduces the risk of production delays caused by operational errors or equipment failures. This stability ensures that buyers can rely on consistent delivery schedules, which is essential for maintaining their own production timelines. The ability to produce high-purity material directly from the reactor reduces the need for extensive reprocessing, further shortening the overall manufacturing cycle time.
- Scalability and Environmental Compliance: The small reaction volume and closed system design significantly enhance process safety, reducing the risk of accidents and ensuring compliance with stringent environmental regulations. The efficient use of reagents and the minimization of by-products mean that the process generates less hazardous waste, simplifying the environmental compliance burden. The system's modularity allows for easy expansion, enabling manufacturers to scale production from 100 kgs to 100 MT annual commercial production seamlessly. This scalability ensures that the supply can grow alongside the demand, providing a long-term partnership opportunity for buyers seeking a stable source of critical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding the technical feasibility and commercial viability of this continuous flow synthesis method. These insights are derived directly from the patent data and are intended to provide clarity for technical and procurement decision-makers. Understanding these details is essential for evaluating the potential integration of this technology into your supply chain. The answers reflect the specific advantages of the micro-reactor system over traditional methods, highlighting its suitability for modern pharmaceutical manufacturing standards.
Q: How does the continuous flow method improve yield compared to traditional batch synthesis?
A: Traditional batch methods typically achieve yields between 70% and 85% due to side reactions and poor heat dissipation. The continuous flow micro-reactor system described in patent CN112979461B enhances mass and heat transfer, suppressing side reactions and consistently achieving yields exceeding 90% with purity levels above 96%.
Q: What are the safety advantages of using micro-channel reactors for chlorination?
A: Chlorination reactions are exothermic and involve hazardous gases. The micro-channel reactor system minimizes the online liquid holdup and reaction volume, significantly reducing the potential safety hazards associated with large-scale batch chlorination. Additionally, the system allows for precise control of chlorine dosage, preventing excessive gas accumulation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scale-up through a multi-channel parallel amplification strategy. The continuous nature of the flow system, combined with high space-time yield and automation, facilitates the transition from laboratory scale to commercial production volumes without the bottlenecks typical of batch processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-4-Oxoacetic Acid Amyl Ester Supplier
The technological potential of the continuous flow synthesis method for 3-chloro-4-oxoacetic acid amyl ester is immense, offering a pathway to more sustainable and efficient chemical manufacturing. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of this critical intermediate. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the highest industry standards. We understand the complexities involved in transitioning from laboratory scale to industrial production and have the expertise to navigate these challenges effectively. Our commitment to quality and reliability makes us a trusted partner for global pharmaceutical and fine chemical companies.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific needs. We encourage you to reach out to us to request specific COA data and route feasibility assessments for this compound. By partnering with us, you gain access to advanced manufacturing technologies and a dedicated team committed to your success. Let us help you secure a reliable supply of high-purity intermediates for your next project.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
