Advanced Continuous Flow Synthesis of 3-Chloro-4-Oxoacetic Acid Amyl Ester for Industrial Scale-Up
Advanced Continuous Flow Synthesis of 3-Chloro-4-Oxoacetic Acid Amyl Ester for Industrial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking more efficient, safer, and scalable methods for producing critical intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN112979461A, which details a fully continuous flow preparation method for 3-chloro-4-oxoacetic acid amyl ester. This compound, often referred to as amyl 3-chloro-4-oxoacetate, serves as a pivotal building block in the synthesis of Vitamin B1 and finds extensive applications across medicine, pesticide, and dye manufacturing sectors. The structural integrity and functional groups of this molecule, as depicted in the chemical structure below, require precise control during synthesis to avoid degradation and ensure high purity standards required by global regulatory bodies.
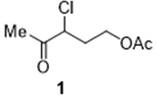
The innovation presented in this patent shifts the paradigm from traditional batch processing to a sophisticated micro-reaction system. By leveraging microchannel technology, the process achieves superior mass and heat transfer, which is critical for exothermic reactions involving hazardous reagents like chlorine gas. For R&D directors and process engineers, this represents a move towards intensified chemistry that not only enhances reaction kinetics but also drastically reduces the environmental footprint. The ability to produce high-purity 3-chloro-4-oxoacetic acid amyl ester with reduced impurity profiles makes this technology highly attractive for reliable pharmaceutical intermediates supplier networks aiming to meet stringent quality specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amyl 3-chloro-4-oxoacetate has relied on batch kettle methodologies that suffer from inherent inefficiencies and safety concerns. Early literature, such as the work by Karaulova et al. in 1967, described processes yielding only 71% using acetic anhydride in aqueous acetic acid, while later methods by Hawksley and others utilizing sulfuryl chloride struggled to exceed yields of 85%. These traditional approaches often necessitate the use of toxic catalysts like pyridine or harsh conditions involving sulfuric acid, which complicate downstream purification and waste treatment. Furthermore, batch chlorination reactions are notoriously difficult to control thermally; the accumulation of heat in large vessels can lead to side reactions, reduced selectivity, and potential safety hazards associated with handling large volumes of chlorine gas. The reaction times in these batch processes typically span 4 to 6 hours or even days, resulting in low space-time yields and high energy consumption, which are significant bottlenecks for cost reduction in vitamin B1 intermediate manufacturing.
The Novel Approach
In stark contrast, the novel approach outlined in patent CN112979461A utilizes a fully continuous flow micro-reaction system that fundamentally transforms the production landscape. This method integrates a micro-mixer, micro-channel reactor, and continuous extraction separator into a seamless workflow. The process begins with the simultaneous conveyance of acetylbutyrolactone and chlorine gas into a microchannel reactor, where rapid mixing and efficient heat dissipation allow for precise chlorination. The intermediate is then immediately subjected to continuous acylation with a mixture of glacial acetic acid, hydrochloric acid, and water. This transition eliminates the need for expensive and hazardous acetic anhydride, replacing it with cheaper and safer acetic acid. The continuous nature of the process reduces the total reaction time from days to approximately thirty minutes, dramatically improving throughput. For supply chain heads, this translates to enhanced supply chain reliability and the ability to respond quickly to market demands without the long lead times associated with batch campaign scheduling.
Mechanistic Insights into Micro-Channel Chlorination and Acylation
The core of this technological advancement lies in the precise control of reaction parameters within the micro-channel environment. The synthesis proceeds through a two-stage cascade reaction, starting with the chlorination of acetylbutyrolactone (Formula 3) to form the key intermediate, alpha-acetyl-alpha-chloro-gamma-butyrolactone (Formula 2). In a micro-reactor, the diffusion distance for molecules is microscopic, allowing for near-instantaneous mixing of the chlorine gas and the liquid substrate. This eliminates concentration gradients that typically plague batch reactors, ensuring that the chlorination occurs uniformly and minimizing over-chlorination or decomposition. The exothermic nature of the chlorination is managed effectively by the high surface-area-to-volume ratio of the microchannels, which facilitates rapid heat exchange with the cooling medium. This thermal control is crucial for maintaining the integrity of the sensitive lactone ring and preventing the formation of poly-chlorinated byproducts.
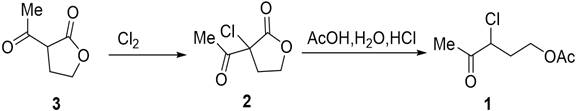
Following chlorination, the reaction mixture flows directly into the acylation stage without isolation, showcasing the power of telescoped synthesis. The intermediate reacts with the acetic acid mixture under controlled temperatures ranging from 50°C to 150°C within the microchannel reactor. The mechanism involves the acid-catalyzed ring opening of the lactone and subsequent esterification. In traditional batch systems, achieving the necessary activation energy often requires prolonged heating, which degrades the product. However, the pressurized environment of the flow system allows the reaction mixture to remain liquid at temperatures above the normal boiling point of the solvents, accelerating the reaction kinetics significantly. This results in a conversion rate of nearly 100% for the substrate and a product yield exceeding 90%, with purity levels reaching 96% or higher directly from the extractor. Such high purity reduces the burden on distillation columns, lowering energy costs and solvent usage, which is a key factor in commercial scale-up of complex fine chemicals.
How to Synthesize 3-Chloro-4-Oxoacetic Acid Amyl Ester Efficiently
Implementing this continuous flow protocol requires a specialized setup comprising feed pumps, mass flow controllers for gases, and temperature-controlled micro-reactor modules. The process is designed to be automated, minimizing human intervention and ensuring consistent product quality batch after batch. Operators must carefully balance the molar ratios of chlorine to substrate, typically maintaining a ratio between 1:1 and 1:3 to ensure complete conversion while avoiding excess gas waste. The detailed standardized synthetic steps, including specific flow rates, residence times, and quenching protocols, are critical for replicating the high yields reported in the patent data. For technical teams looking to adopt this methodology, understanding the interplay between flow velocity and residence time is essential to optimize the space-time yield.
- Simultaneously convey acetylbutyrolactone and chlorine gas into a microchannel reactor for continuous chlorination to obtain alpha-acetyl-alpha-chloro-gamma-butyrolactone.
- Mix the chlorinated intermediate with a solution of glacial acetic acid, hydrochloric acid, and water in a micromixer, then react in a microchannel reactor for continuous acylation.
- Quench the reaction mixture with an inorganic base solution and perform continuous extraction and separation to isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the shift to this continuous flow technology offers compelling economic and operational benefits that go beyond simple yield improvements. The elimination of toxic catalysts like pyridine and the replacement of acetic anhydride with glacial acetic acid significantly lowers raw material costs and simplifies waste disposal procedures. Traditional batch processes often generate substantial amounts of hazardous waste that require expensive treatment; the continuous flow method's precision minimizes byproduct formation, leading to substantial cost savings in environmental compliance. Moreover, the intrinsic safety of handling chlorine gas in micro-channels, where the active inventory is mere milliliters rather than tons, reduces insurance premiums and safety infrastructure costs. This makes the process not only cheaper but also more resilient to regulatory scrutiny.
- Cost Reduction in Manufacturing: The continuous flow process drastically simplifies the production workflow by combining reaction and separation steps, which reduces labor intensity and equipment footprint. By avoiding the use of expensive acetic anhydride and toxic catalysts, the direct material costs are significantly lowered. Additionally, the high selectivity of the micro-reaction system minimizes the formation of impurities, which means less solvent and energy are required for purification distillation. These factors combine to create a leaner manufacturing process that offers significant cost reduction in vitamin B1 intermediate manufacturing without compromising on quality.
- Enhanced Supply Chain Reliability: The automation and continuous nature of this process ensure a steady stream of product, eliminating the stop-start cycles of batch production that often lead to supply bottlenecks. The ability to run the system for extended periods with minimal intervention enhances the reliability of supply for downstream API manufacturers. Furthermore, the process is robust against variations in raw material quality due to the precise control of reaction conditions, ensuring consistent output. This reliability is crucial for reducing lead time for high-purity agrochemical intermediates and pharmaceutical ingredients, allowing partners to maintain tighter inventory controls.
- Scalability and Environmental Compliance: Scaling this technology does not require building larger, more dangerous reactors; instead, it employs a numbering-up strategy where additional micro-reactor units are added in parallel. This modular approach allows for flexible capacity expansion that matches market demand, reducing capital expenditure risks. From an environmental perspective, the closed system prevents the release of volatile organic compounds and chlorine gas, aligning with green chemistry principles. The reduced energy consumption and waste generation make this process highly compliant with increasingly strict global environmental regulations, future-proofing the supply chain against regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous flow synthesis of 3-chloro-4-oxoacetic acid amyl ester. These insights are derived directly from the technical specifications and advantageous effects described in the patent literature, providing clarity on how this method outperforms legacy batch processes. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this technology into their existing production lines or sourcing strategies.
Q: How does the continuous flow method improve safety compared to batch chlorination?
A: The continuous flow method utilizes microchannel reactors with extremely small internal volumes, significantly reducing the online liquid holdup of hazardous chlorine gas and exothermic reaction mixtures. This intrinsic safety feature minimizes the risk of thermal runaway and gas leakage compared to large batch kettle reactors.
Q: What are the yield and purity advantages of this micro-reaction process?
A: According to patent data, the continuous flow process improves the product yield from approximately 70% in traditional batch methods to over 90%, while increasing purity from 95% to over 96% directly from the reaction, reducing the need for extensive downstream purification.
Q: Can this process be easily scaled for industrial production?
A: Yes, the process is designed for easy industrial scale-up through a multi-channel parallel amplification strategy. Instead of increasing reactor size, multiple micro-reactor units can be operated in parallel to increase capacity while maintaining the precise heat and mass transfer characteristics of the laboratory scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-4-Oxoacetic Acid Amyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous flow chemistry in the production of high-value intermediates like 3-chloro-4-oxoacetic acid amyl ester. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of micro-reactor technology are realized in full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced process analytical technology (PAT) to maintain stringent purity specifications, guaranteeing that every batch meets the exacting standards required for Vitamin B1 synthesis and other critical applications.
We invite procurement leaders and technical directors to collaborate with us to optimize their supply chains. By leveraging our expertise in flow chemistry, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project benefits from the highest efficiency and safety standards available in the industry today.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
