Advanced Chromatographic Purification of Clofarabine for Commercial API Production
Advanced Chromatographic Purification of Clofarabine for Commercial API Production
The pharmaceutical industry constantly faces the challenge of delivering high-purity active pharmaceutical ingredients (APIs), particularly for potent oncology treatments where impurity profiles are strictly regulated. Patent CN101475621B introduces a groundbreaking method for purifying Clofarabine, a second-generation nucleoside analogue used in the treatment of acute leukemia, specifically addressing the persistent issue of stereoisomeric contamination. This technology leverages reverse-phase macroporous adsorption resin chromatography to achieve purity levels exceeding 99.8 percent, a critical benchmark for regulatory approval and patient safety. As a leading entity in fine chemical manufacturing, we recognize that the ability to separate structurally similar isomers is often the bottleneck in API commercialization. This patent provides a robust, scalable solution that transforms a complex purification challenge into a streamlined industrial process, ensuring a reliable clofarabine supplier can meet the rigorous demands of global pharmaceutical markets.
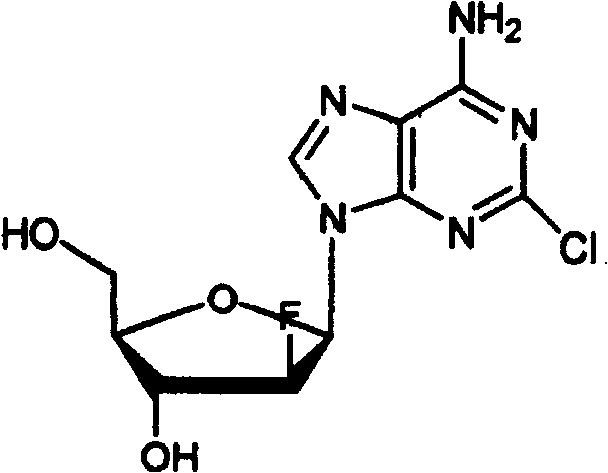
Clofarabine represents a significant advancement in cancer therapy, yet its synthesis inherently produces a challenging mixture of stereoisomers. The structural complexity of this molecule, characterized by a chlorinated purine base attached to a fluorinated arabinose sugar, creates unique separation difficulties. The presence of the alpha-configurational isomer, a byproduct of the glycosylation reaction, poses a severe risk to product quality if not effectively removed. Traditional purification strategies often fail to distinguish between these closely related molecules, leading to suboptimal yields or insufficient purity. The methodology outlined in CN101475621B overcomes these limitations by exploiting subtle polarity differences through advanced chromatographic techniques, offering a pathway to high-purity clofarabine that is both economically and technically superior to previous methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Clofarabine, as described in earlier patents like WO03011877, involves a condensation reaction between a protected ribofuranose derivative and 2-chloroadenine. This reaction inevitably generates a mixture containing both the desired beta-configuration product and the undesired alpha-configurational isomer. Following deprotection, the resulting crude product contains significant amounts of the alpha-anomer impurity. The fundamental problem lies in the fact that Clofarabine and its alpha-configurational isomer exhibit extremely approaching physicochemical properties. Their solubility profiles, melting points, and crystallization behaviors are so similar that conventional purification processes, such as standard recrystallization, are largely ineffective at removing the alpha isomer. Even with optimized synthetic conditions attempting to suppress the formation of the alpha isomer, content levels often remain above 2 percent, and in many standard operations, they can exceed 10 percent. This level of impurity is unacceptable for a human medicine intended for pediatric leukemia treatment, necessitating a more sophisticated separation strategy.
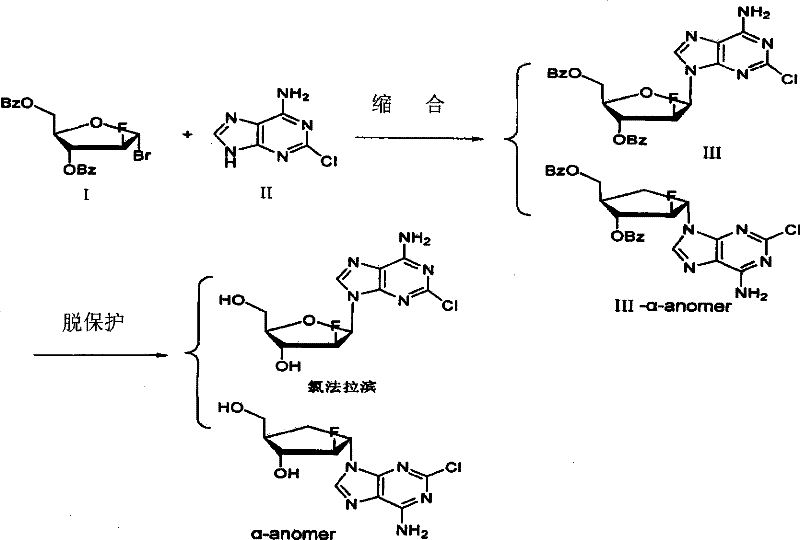
The Novel Approach
The novel approach detailed in this patent shifts the paradigm from trying to prevent impurity formation during synthesis to efficiently removing it post-synthesis using chromatography. Instead of relying on the subtle thermodynamic differences utilized in recrystallization, this method employs a kinetic and adsorption-based separation using a chromatographic column packed with inverse macroporous adsorption resin. By selecting specific resin types, such as MCI-gel CHP20P or MCI-gel CHP20SS, the process creates an environment where the polarity differences between the alpha and beta isomers are amplified. The method involves loading a dilute aqueous solution of the crude product onto the column and performing a gradient elution. Initially, pure water is used to wash out highly polar components, followed by a gradual increase in organic solvent concentration (methanol, ethanol, or acetone). This precise control over the mobile phase polarity allows the more polar alpha-configurational isomer to elute first, while the desired Clofarabine, being slightly less polar, is retained longer and elutes subsequently. This results in a clean separation that recrystallization simply cannot achieve, ensuring cost reduction in API manufacturing by maximizing the recovery of the valuable beta-isomer.
Mechanistic Insights into Reverse-Phase Macroporous Resin Chromatography
The core mechanism driving this purification success is the interaction between the solute molecules and the hydrophobic surface of the polystyrene-type macroporous adsorption resin. Unlike silica-based normal phase chromatography, reverse-phase chromatography relies on hydrophobic interactions. The resin beads, typically composed of a cross-linked polystyrene matrix, provide a non-polar stationary phase. When the aqueous crude solution is introduced, both the Clofarabine and its alpha-isomer adsorb onto the resin surface due to their hydrophobic purine rings. However, the stereochemistry at the anomeric center influences the overall three-dimensional shape and dipole moment of the molecule. The alpha-isomer, with its different spatial arrangement of the hydroxyl and fluorine groups, exhibits a slightly higher polarity compared to the beta-isomer (Clofarabine). In a reverse-phase system, higher polarity translates to weaker retention on the hydrophobic stationary phase. Consequently, as the eluent strength is increased through the gradient addition of organic solvents, the alpha-isomer desorbs and moves through the column faster. The beta-isomer requires a higher concentration of organic modifier to break its hydrophobic interactions with the resin, resulting in a later elution time. This differential retention time is the key to achieving the high resolution necessary for pharmaceutical grade purity.
Furthermore, the choice of macroporous resin is critical for managing impurity profiles and ensuring process robustness. The patent specifies the use of resins like MCI-gel CHP20P, which have specific particle size distributions (75-150 μm) and pore structures optimized for small molecule separation. These physical characteristics ensure efficient mass transfer and minimize band broadening, which is essential when processing large volumes of dilute solutions. The gradient elution profile, moving from pure water to mixtures with decreasing water ratios (e.g., 20:1 down to 20:4 water-to-organic), is meticulously designed to sharpen the separation peaks. By carefully monitoring the eluent via HPLC, operators can precisely cut the fractions, collecting only those segments where the Clofarabine content is greater than 99.8 percent and single impurity content is less than 0.1 percent. This mechanistic understanding allows for the commercial scale-up of complex nucleoside analogs, transforming a laboratory curiosity into a viable manufacturing process that guarantees reducing lead time for high-purity anticancer agents by avoiding multiple failed recrystallization attempts.
How to Synthesize Clofarabine Efficiently
The implementation of this purification protocol requires careful attention to column preparation and elution parameters to ensure reproducibility and high yield. The process begins with the conditioning of the resin, which must be thoroughly soaked in ethanol to expand the polymer matrix and remove any manufacturing residues, followed by equilibration with water. The loading concentration of the crude Clofarabine solution is a critical parameter; maintaining it between 0.001 and 0.025 weight percent prevents column overload, which could compromise the resolution between the alpha and beta isomers. Once loaded, the gradient elution must be performed at a controlled flow rate, typically around 6 ml/min for laboratory columns, to allow sufficient time for the adsorption-desorption equilibrium to occur at each theoretical plate. Detailed standardized synthesis steps see the guide below for exact operational parameters regarding solvent ratios and fraction collection criteria.
- Prepare the chromatographic column by packing it with reverse-phase macroporous adsorption resin (such as MCI-gel CHP20P) that has been soaked in ethanol for over 10 hours and washed with pure water.
- Load the crude Clofarabine aqueous solution onto the column, ensuring the concentration is maintained between 0.001 and 0.025 weight percent to optimize separation efficiency.
- Perform gradient elution starting with pure water, followed by water-organic solvent mixtures with gradually decreasing water ratios, collecting fractions where purity exceeds 99.8%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chromatographic purification method offers substantial strategic benefits beyond mere technical compliance. The primary advantage lies in the dramatic improvement of process yield and material efficiency. Traditional methods that rely on repeated recrystallization to chase purity often result in significant material loss, driving up the cost of goods sold (COGS). In contrast, this chromatographic method boasts a yield reaching more than 90 percent, meaning that nearly all the synthesized material is converted into saleable, high-purity product. This efficiency directly translates to significant cost savings in raw material consumption and waste disposal. Furthermore, the solvents used in the process, such as ethanol and water, are inexpensive, readily available, and easily recyclable, further enhancing the economic viability of the process. The simplicity of the operation also reduces the need for highly specialized labor, lowering operational expenditures.
- Cost Reduction in Manufacturing: The elimination of inefficient recrystallization cycles and the high recovery rate of the active ingredient significantly lower the overall production cost. By avoiding the loss of valuable intermediates during purification, manufacturers can optimize their budget allocation. Additionally, the use of common, non-hazardous solvents reduces the costs associated with hazardous waste management and solvent procurement. The process does not require exotic catalysts or extreme conditions, which minimizes energy consumption and equipment wear, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: A robust purification process is the backbone of a reliable supply chain. Because this method consistently achieves purity levels above 99.8 percent regardless of minor fluctuations in the crude synthesis quality, it acts as a buffer against upstream variability. This reliability ensures that delivery schedules are met without the delays often caused by failed quality control batches. The scalability of the chromatographic process means that production can be ramped up from pilot scale to multi-ton commercial production without fundamental changes to the purification logic, ensuring continuity of supply for downstream drug formulation partners.
- Scalability and Environmental Compliance: The method is explicitly designed for industrial mass production, utilizing standard glass or stainless steel columns that are easy to scale. The environmental footprint is minimized through the use of water and alcohols, which are greener alternatives to chlorinated solvents often found in older purification protocols. The ability to recycle the mobile phase solvents further aligns with modern green chemistry principles and environmental regulations. This compliance reduces the regulatory burden on the supply chain team, facilitating smoother audits and faster market entry for the final pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the purification of Clofarabine using the methodology described in patent CN101475621B. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and advantages of this approach for potential manufacturing partners.
Q: Why is conventional recrystallization ineffective for purifying Clofarabine?
A: Conventional recrystallization is ineffective because Clofarabine and its alpha-configurational isomer possess extremely similar physicochemical properties, making them difficult to separate based on solubility differences alone.
Q: What purity levels can be achieved using this chromatographic method?
A: This method utilizing reverse-phase macroporous adsorption resin ensures that the purity of the Clofarabine product reaches more than 99.8 percent, with single impurity content less than 0.1%.
Q: Is this purification process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial mass production, featuring simple operation, non-toxic solvents that are recyclable, and high yields exceeding 90%, making it economically viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clofarabine Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from patent literature to commercial reality requires deep technical expertise and state-of-the-art facilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results of CN101475621B can be realized on an industrial scale. We are committed to delivering high-purity intermediates and APIs that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Our capability to implement complex chromatographic separations allows us to offer a reliable clofarabine supplier service that guarantees consistency, quality, and regulatory compliance for your oncology drug development programs.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By leveraging our technical prowess in nucleoside chemistry and purification, we can help you navigate the complexities of API production. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced purification capabilities can enhance your project's success and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
