Revolutionizing Clofarabine Production: Advanced Synthesis for Commercial Scale-Up
Revolutionizing Clofarabine Production: Advanced Synthesis for Commercial Scale-Up
The pharmaceutical landscape for treating intractable or recurrent acute lymphoblastic leukemia is constantly evolving, driven by the need for more efficient and cost-effective production of critical nucleoside analogues like clofarabine. Patent CN101555267A introduces a transformative synthesis method for 6-amino-2-chloro-9-(2-deoxy-2-fluoro-beta-D-ribofuranosyl)-9H-purine that addresses longstanding inefficiencies in existing manufacturing protocols. This technical breakthrough offers a robust pathway that combines mild reaction conditions with high yield potential, directly responding to the industry's demand for a reliable pharmaceutical intermediates supplier capable of delivering consistent quality. By streamlining the synthetic sequence and optimizing reagent selection, this innovation not only enhances the chemical feasibility of the process but also lays the groundwork for substantial cost reduction in API manufacturing, ensuring that life-saving medications remain accessible through a stable and scalable supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
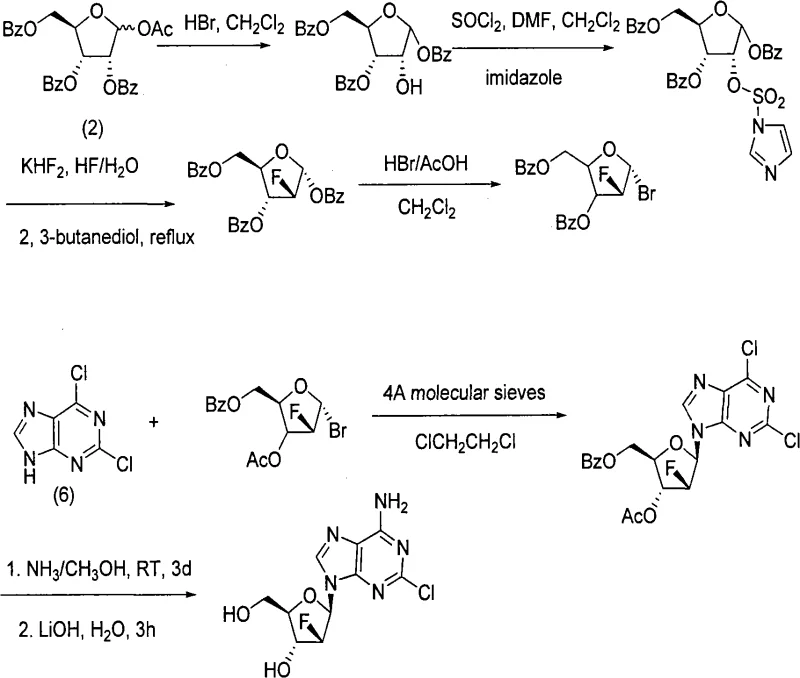
Historically, the production of clofarabine has been hindered by complex synthetic routes that involve excessive reaction steps and the utilization of prohibitively expensive reagents, creating significant bottlenecks for commercial scale-up of complex pharmaceutical intermediates. Traditional pathways, such as those starting with 1-O-ethanoyl-2,3,5-three-O-benzoyl-D-ribofuranose and 2,6-dichloropurine, often suffer from low overall yields and tedious operational procedures that are ill-suited for large-scale industrial adaptation. These legacy methods frequently require harsh reaction conditions that complicate impurity control and increase the burden on downstream purification processes, thereby inflating production costs and extending lead times. Furthermore, the reliance on specific, costly catalysts in older routes limits the flexibility of procurement managers who are tasked with optimizing supply chain reliability and reducing lead time for high-purity active pharmaceutical ingredients without compromising on quality standards.
The Novel Approach
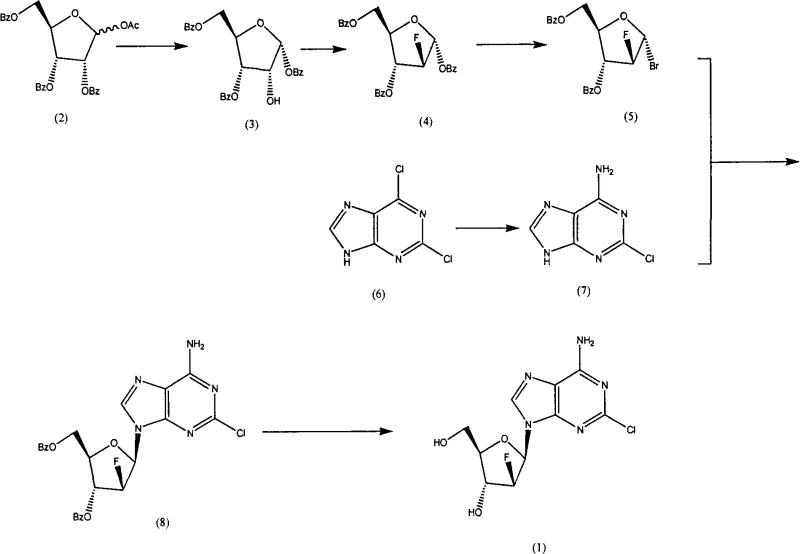
In stark contrast to the cumbersome legacy protocols, the novel approach detailed in the patent data presents a streamlined synthesis strategy that prioritizes operational simplicity and economic efficiency. This method employs a direct sequence involving crystallization, fluoridation, bromination, ammonia reaction, selection reaction, and methanol reaction, which collectively reduce the number of unit operations required to reach the final target molecule. By utilizing readily available starting materials and optimizing solvent systems such as methylene dichloride and tertiary amyl alcohol, the new route eliminates several high-cost barriers associated with previous technologies. This strategic shift not only facilitates easier amplification from laboratory to plant scale but also ensures that the process remains robust against variability, providing a solid foundation for cost reduction in electronic chemical manufacturing and related high-value sectors that demand precision and consistency.
Mechanistic Insights into DAST-Mediated Fluorination and Glycosylation
The core of this technological advancement lies in the precise execution of the fluorination and glycosylation steps, which are critical for establishing the stereochemical integrity of the nucleoside analogue. The utilization of DAST (Diethylaminosulfur trifluoride) as a fluorinating agent allows for the efficient conversion of the hydroxyl group to a fluoro substituent under controlled temperatures ranging from -20°C to 80°C, ensuring high selectivity and minimizing side reactions. Following this, the bromination step using hydrogen bromide in acetic acid generates a reactive glycosyl donor that is primed for coupling, a process that is meticulously managed to prevent degradation of the sensitive sugar moiety. This careful orchestration of reaction conditions demonstrates a deep understanding of organic synthesis mechanics, enabling the production of high-purity OLED material precursors and similar complex molecules where structural fidelity is paramount for biological activity.
Furthermore, the coupling reaction between the activated sugar donor and the purine base is facilitated by strong bases like potassium tert-butoxide in non-protic solvents, which promotes the formation of the desired beta-anomer with high stereoselectivity. The subsequent deprotection using sodium methylate in methanol is conducted under mild conditions to remove benzoyl protecting groups without affecting the fluorine atom or the purine ring structure. This mechanistic precision is essential for maintaining a clean impurity profile, which is a key concern for R&D Directors focusing on the purity and impurity spectrum of drug substances. By controlling these critical parameters, the process ensures that the final product meets stringent regulatory requirements, thereby enhancing the commercial viability of the synthesis route for global pharmaceutical markets.
How to Synthesize Clofarabine Efficiently
Implementing this advanced synthesis route requires a systematic approach that adheres strictly to the optimized reaction conditions and solvent specifications outlined in the patent documentation to ensure maximum efficiency and yield. The process begins with the preparation of the ribose intermediate, followed by sequential fluorination and bromination to activate the sugar for coupling with the purine base. Each step is designed to be operationally simple, allowing for easy monitoring and control within a standard chemical manufacturing facility equipped with appropriate safety measures for handling reactive reagents. For a comprehensive breakdown of the specific temperatures, reaction times, and workup procedures required to replicate this success, please refer to the standardized synthesis guide provided below, which details the exact protocol for achieving industrial-grade results.
- Deprotect 1-O-acetyl-2,3,5-tri-O-benzoyl-D-ribofuranose using HBr in methylene dichloride to obtain the hydroxyl intermediate.
- Perform fluorination using DAST reagent followed by bromination with hydrogen bromide acetic acid solution to generate the glycosyl donor.
- Couple the glycosyl donor with 2-chloroadenine using potassium tert-butoxide in tertiary amyl alcohol, followed by methanolysis to yield clofarabine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this novel synthesis method offers profound benefits that extend beyond mere chemical efficiency, directly addressing the pain points of cost management and supply continuity. By eliminating the need for exotic or prohibitively expensive catalysts and simplifying the overall workflow, manufacturers can achieve significant cost savings in raw material acquisition and processing overhead. This efficiency translates into a more competitive pricing structure for the final intermediate, allowing procurement managers to negotiate better terms and secure a more stable supply of critical materials. Moreover, the robustness of the process reduces the risk of batch failures, which is a crucial factor for supply chain heads who are responsible for maintaining uninterrupted production schedules and meeting strict delivery commitments to downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The streamlined nature of the new route inherently lowers the cost of goods sold by reducing the number of isolation and purification steps required, which in turn minimizes solvent consumption and waste disposal expenses. The avoidance of expensive reagents found in conventional methods further contributes to a leaner cost structure, enabling substantial financial optimization without sacrificing product quality. This economic advantage is particularly valuable in a market where margin pressure is high, allowing companies to reinvest savings into R&D or capacity expansion while maintaining healthy profitability levels across their portfolio of specialty chemicals.
- Enhanced Supply Chain Reliability: The use of common, commercially available solvents and reagents ensures that the supply chain is less vulnerable to disruptions caused by the scarcity of niche chemicals. This accessibility means that production can be sustained even during periods of market volatility, providing a reliable pharmaceutical intermediates supplier with the agility to respond to sudden spikes in demand. Additionally, the simplified operational requirements reduce the dependency on highly specialized labor, making it easier to scale production capacity rapidly when needed to support global clinical trials or commercial launches of new therapies.
- Scalability and Environmental Compliance: The mild reaction conditions and high yields associated with this process facilitate a smoother transition from pilot scale to full commercial production, reducing the technical risks typically associated with scaling up complex organic syntheses. Furthermore, the reduction in waste generation and the use of less hazardous materials align with increasingly stringent environmental regulations, helping companies meet their sustainability goals and avoid potential compliance penalties. This alignment with green chemistry principles not only protects the environment but also enhances the corporate reputation of manufacturers as responsible stewards of chemical safety and ecological balance.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis innovation, we have compiled a set of frequently asked questions that address common concerns regarding implementation and performance. These insights are derived directly from the patent specifications and are intended to provide clear, actionable information for stakeholders evaluating the feasibility of adopting this technology. Understanding these details is crucial for making informed decisions about process integration and supply chain strategy, ensuring that all parties are aligned on the capabilities and limitations of the new manufacturing route.
Q: What are the primary advantages of this new clofarabine synthesis route over conventional methods?
A: The novel route described in patent CN101555267A significantly simplifies the operational procedure by utilizing milder reaction conditions and avoiding expensive reagents found in traditional pathways, leading to easier industrial amplification and reduced production costs.
Q: How does this method impact the purity profile of the final nucleoside analogue?
A: By optimizing crystallization steps and employing specific solvent systems like methylene dichloride and normal heptane, the process achieves high purity levels, with embodiments demonstrating purity exceeding 99.6%, which is critical for pharmaceutical applications.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly states that the method is adapted for industrial production due to its simple synthesis procedure, easy operation, and high yield, making it ideal for reliable pharmaceutical intermediates suppliers aiming for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clofarabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into tangible commercial successes, and we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this clofarabine synthesis to life. Our team of expert chemists and engineers is dedicated to optimizing every aspect of the manufacturing process to ensure stringent purity specifications and rigorous QC labs validate every batch against the highest international standards. We understand that consistency is key in the pharmaceutical industry, and our state-of-the-art facilities are designed to handle the specific nuances of nucleoside analogue synthesis, guaranteeing a supply of high-purity nucleoside analogues that your projects depend on for success.
We invite you to collaborate with us to explore how this advanced synthesis route can transform your supply chain dynamics and drive down your overall production costs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate the clear advantages of partnering with a leader in fine chemical manufacturing committed to your growth and operational excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
