Advanced Synthesis of Lactosyl Thiourea Heterocycles for Commercial Antineoplastic Drug Development
The pharmaceutical industry is constantly seeking novel scaffolds that combine high biological activity with manageable synthetic complexity. Patent CN101215304B introduces a groundbreaking class of lactosyl thiourea heterocyclic compounds that demonstrate significant potential in antineoplastic applications. This technology leverages the inherent biological recognition properties of sugar moieties to enhance the therapeutic profile of thiourea derivatives. By integrating a lactose backbone with heterocyclic bases such as adenine or benzimidazole, the invention creates a unique chemical space that targets human lung and liver cancer cell lines with notable efficacy. For R&D directors and procurement managers, this patent represents a viable pathway for developing next-generation anticancer intermediates. The synthesis avoids the use of scarce or prohibitively expensive reagents, focusing instead on robust, scalable chemistry that aligns with modern green manufacturing principles. As a reliable pharmaceutical intermediates supplier, understanding the nuances of this six-step sequence is crucial for evaluating its commercial viability and integration into existing production pipelines.
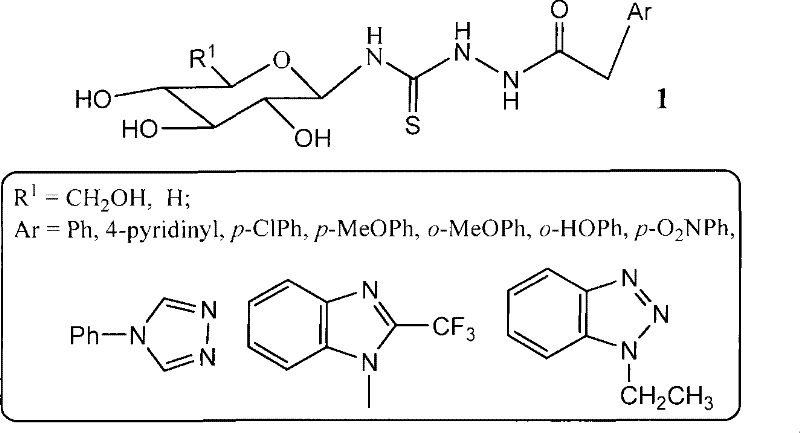
The limitations of conventional methods for synthesizing glycosyl thiourea derivatives often stem from the instability of glycosyl donors and the harsh conditions required for thiourea bond formation. Traditional approaches frequently rely on protected sugar halides that are prone to elimination side reactions, leading to complex impurity profiles that are difficult to purge. Furthermore, many existing routes utilize toxic heavy metal catalysts or require cryogenic conditions that are energy-intensive and difficult to maintain on a large scale. These factors contribute to high production costs and extended lead times, creating bottlenecks for supply chain heads who need consistent volume. In contrast, the novel approach detailed in this patent utilizes a peracetylated lactose derivative that is remarkably stable and easy to handle. The substitution of the anomeric bromine with a thiocyanate group proceeds under reflux conditions in xylene, eliminating the need for sensitive low-temperature control. This shift in reaction engineering drastically simplifies the operational requirements, making the process more robust against batch-to-batch variations. The subsequent coupling with heterocyclic acethydrazides is performed in polar aprotic solvents like DMF, ensuring high solubility of reactants and driving the reaction to completion without excessive forcing conditions.
Mechanistic Insights into Lactose-Based Thiourea Cyclization
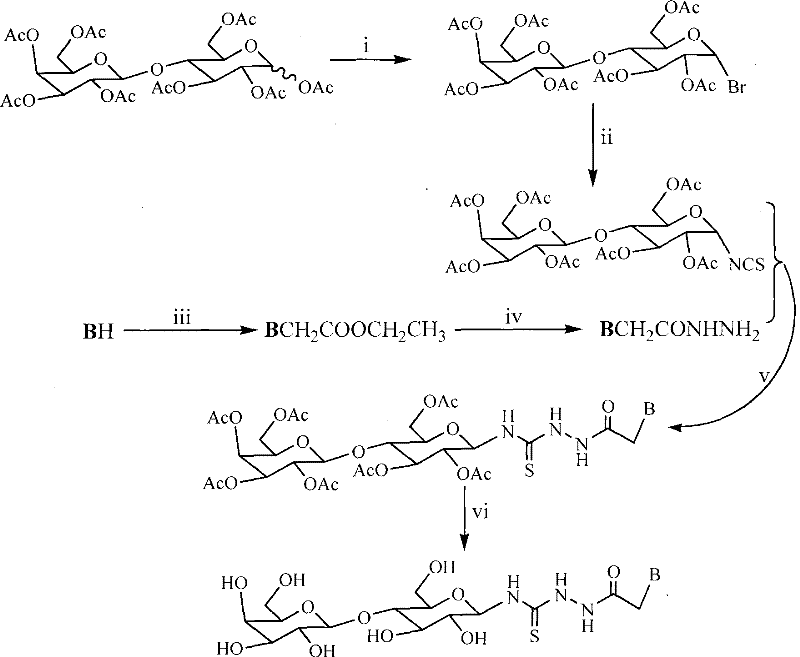
The core of this synthetic strategy lies in the precise manipulation of the lactose anomeric center to install the thiourea linkage without compromising the integrity of the disaccharide. The mechanism begins with the activation of peracetylated lactose using red phosphorus and bromine, generating a reactive glycosyl bromide intermediate. This species is then converted into a glycosyl isothiocyanate via nucleophilic substitution with lead thiocyanate. This specific transformation is critical because the isothiocyanate group serves as a highly electrophilic handle for the subsequent condensation with the heterocyclic hydrazide. The reaction proceeds through a nucleophilic attack by the hydrazide nitrogen on the carbon of the isothiocyanate group, forming the thiosemicarbazide backbone. This step is facilitated by the electron-withdrawing nature of the acetyl protecting groups, which stabilize the transition state. For R&D teams, understanding this electronic interplay is vital for optimizing reaction times and minimizing the formation of urea byproducts. The final deprotection step uses sodium methylate in methanol, which selectively cleaves the ester bonds while leaving the thiourea and glycosidic linkages intact. This chemoselectivity ensures that the final product retains the desired structural features necessary for biological activity.
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, particularly for oncology applications where safety margins are narrow. The described process incorporates multiple purification checkpoints, including recrystallization and column chromatography, to ensure high purity standards. The use of lead thiocyanate does introduce a heavy metal, but the subsequent workup involves filtration and washing steps that effectively reduce residual lead to acceptable levels. Moreover, the crystalline nature of the intermediates allows for efficient purification via recrystallization from mixed solvent systems like toluene and petroleum ether. This physical property is a significant advantage over amorphous intermediates that require extensive chromatographic purification, which is often a bottleneck in manufacturing. The final product is obtained as a white solid powder, indicating a high degree of crystallinity and purity. For quality control laboratories, this means that standard analytical techniques such as HPLC and NMR can be easily applied to verify identity and assay. The robust nature of the synthesis ensures that the impurity profile remains consistent across different batches, facilitating regulatory approval processes.
How to Synthesize Lactosyl Thiourea Heterocycles Efficiently
The synthesis of these complex molecules requires careful attention to reaction stoichiometry and temperature control to maximize yield and minimize side reactions. The process begins with the preparation of the glycosyl donor, followed by the independent synthesis of the heterocyclic acceptor. These two streams converge in the condensation step to form the protected thiosemicarbazide. Detailed standard operating procedures for each of the six steps are essential for reproducibility. Operators must be trained to handle reagents like bromine and sodium hydride with appropriate safety precautions. The reaction times vary depending on the specific heterocyclic base used, requiring real-time monitoring via thin-layer chromatography.
- Bromination of peracetylated lactose using red phosphorus and bromine water to form 1-bromo-2,3,6,2',3',4',6'-seven-O-ethanoyl-lactose.
- Conversion of the bromo-intermediate to isothiocyanate using lead thiocyanate in refluxing xylene solvent.
- Coupling of heterocyclic base with ethyl chloroacetate, followed by hydrazinolysis to form the acethydrazide intermediate.
- Condensation of the acethydrazide with the lactose isothiocyanate to form the protected thiosemicarbazide.
- Deprotection using sodium methylate in methanol to yield the final high-purity lactosyl thiourea heterocyclic compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits for procurement managers focused on cost reduction in API manufacturing. The starting material, lactose, is an abundant and inexpensive commodity chemical, which provides a significant cost advantage over specialized sugar donors. The elimination of expensive transition metal catalysts further reduces the raw material bill, while also simplifying the waste treatment process. This aligns with the growing demand for sustainable chemistry practices in the pharmaceutical industry. For supply chain heads, the use of common industrial solvents such as methanol, xylene, and toluene ensures that raw material availability is not a constraint. These solvents are readily sourced from multiple suppliers, reducing the risk of supply disruptions. The scalability of the process is another key advantage, as the reaction conditions are compatible with standard stainless steel reactors used in fine chemical production. This means that technology transfer from lab to plant can be achieved with minimal equipment modification. The overall process efficiency translates to shorter production cycles and improved responsiveness to market demand.
- Cost Reduction in Manufacturing: The reliance on commodity chemicals like lactose and common solvents drastically lowers the direct material costs associated with production. By avoiding proprietary or scarce reagents, the manufacturing process becomes less susceptible to price volatility in the raw material market. The simplified purification workflow reduces the consumption of silica gel and eluents, which are often significant cost drivers in intermediate synthesis. Furthermore, the high yield of the condensation step minimizes material loss, ensuring that the maximum amount of starting material is converted into valuable product. These factors combine to create a highly cost-effective manufacturing model that supports competitive pricing strategies.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent output quality, which is critical for maintaining trust with downstream API manufacturers. The use of stable intermediates allows for the potential storage of key precursors, providing a buffer against unexpected demand spikes. This flexibility enables supply chain managers to optimize inventory levels and reduce carrying costs. Additionally, the process does not rely on specialized equipment or exotic conditions, meaning that production can be easily outsourced to multiple contract manufacturing organizations if needed. This diversification of manufacturing capacity mitigates the risk of single-point failures in the supply chain.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing unit operations that are well-understood in the chemical industry. The waste streams generated are primarily organic solvents and salts, which can be managed through standard recovery and treatment systems. The absence of persistent organic pollutants or highly toxic byproducts simplifies environmental compliance and reduces disposal costs. This environmental profile is increasingly important for companies aiming to meet corporate sustainability goals. The ability to scale from grams to tons without fundamental changes to the chemistry ensures a smooth transition from clinical supply to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these lactosyl thiourea derivatives. These answers are derived from the technical specifications and experimental data provided in the patent documentation. Understanding these details is essential for making informed decisions about process adoption and partnership.
Q: What are the key advantages of this lactose-based synthetic route?
A: The process utilizes readily available lactose as a starting material, significantly reducing raw material costs compared to complex sugar donors. The six-step sequence avoids expensive transition metal catalysts, simplifying purification and reducing heavy metal contamination risks in the final API intermediate.
Q: How does this method improve solubility for drug formulation?
A: By introducing the glycosyl moiety directly into the thiourea heterocyclic structure, the resulting compounds exhibit significantly enhanced water solubility compared to their non-glycosylated analogs. This physicochemical property is critical for improving bioavailability in antineoplastic drug formulations.
Q: Is this process scalable for industrial production?
A: Yes, the reaction conditions utilize common industrial solvents like xylene, toluene, and methanol, and avoid extreme cryogenic temperatures. The use of standard unit operations such as reflux, filtration, and recrystallization ensures that the process is highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lactosyl Thiourea Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the complexities of glycosylation chemistry and heterocyclic synthesis, ensuring that your projects are executed with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest industry standards. Our commitment to quality and reliability makes us the ideal partner for developing high-purity antineoplastic intermediates. We understand the critical nature of oncology drug supply chains and are dedicated to providing uninterrupted service.
We invite you to discuss your specific requirements with our technical procurement team to explore how we can support your development goals. Request a Customized Cost-Saving Analysis to understand the economic benefits of partnering with us. We are ready to provide specific COA data and route feasibility assessments to accelerate your project timeline. Let us help you optimize your supply chain for the next generation of cancer therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
