Advanced Cobalt-Catalyzed Asymmetric Hydrogenation for Scalable Chiral Alkane Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient, cost-effective, and sustainable routes to access chiral building blocks. A significant breakthrough in this domain is documented in Chinese Patent CN110655456A, which discloses a novel method for preparing chiral alkyl compounds through the asymmetric catalytic hydrogenation of trisubstituted olefins. Unlike traditional approaches that rely heavily on scarce and expensive noble metals, this invention leverages an earth-abundant cobalt-based catalytic system. The core innovation lies in the use of a specific chiral iminopyridine imidazoline (IIP) ligand coordinated with cobalt halides, enabling the transformation of both single-configuration and, crucially, E/Z mixed trisubstituted olefins into high-value chiral alkanes. This technology addresses a long-standing challenge in organic synthesis where the separation of olefin isomers often constitutes a major bottleneck in terms of cost and yield. By accepting mixed feedstocks directly, the process streamlines the supply chain for critical pharmaceutical intermediates.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of non-functionalized trisubstituted olefins has been dominated by catalysts based on precious metals such as rhodium, ruthenium, and iridium. These systems, while effective, suffer from significant economic and logistical drawbacks due to the strategic scarcity and high volatility of these metal prices. Furthermore, conventional methodologies strictly require the starting olefin material to be of a single geometric configuration, either purely E or purely Z. In practical industrial settings, synthesizing or isolating a single isomer from a mixture often necessitates complex, multi-step purification processes or difficult chromatographic separations. This requirement not only inflates the cost of goods sold (COGS) but also generates substantial chemical waste, contradicting the principles of green chemistry. Previous attempts to use mixed isomers with titanium catalysts have yielded poor results, with low conversion rates and negligible enantioselectivity, leaving a gap in the market for a robust solution that can tolerate isomeric mixtures without compromising optical purity.
The Novel Approach
The methodology outlined in CN110655456A represents a paradigm shift by introducing a cobalt-based catalytic system that exhibits remarkable tolerance towards substrate geometry. The use of the chiral CoX2-IIP complex allows for the direct hydrogenation of E/Z mixed trisubstituted olefins, effectively bypassing the need for costly isomer separation steps. This approach utilizes atmospheric hydrogen as the hydrogen source, coupled with silane compounds like polymethylhydrosiloxane (PMHS) or diphenylsilane as co-catalysts, ensuring high atom economy. The reaction proceeds under mild conditions, typically at room temperature or even 0°C, which minimizes energy consumption and reduces the risk of thermal degradation for sensitive functional groups. By replacing noble metals with cobalt and accepting mixed feedstocks, this novel approach offers a dual advantage of drastic raw material cost reduction and simplified process engineering, making it highly attractive for the commercial scale-up of complex pharmaceutical intermediates.
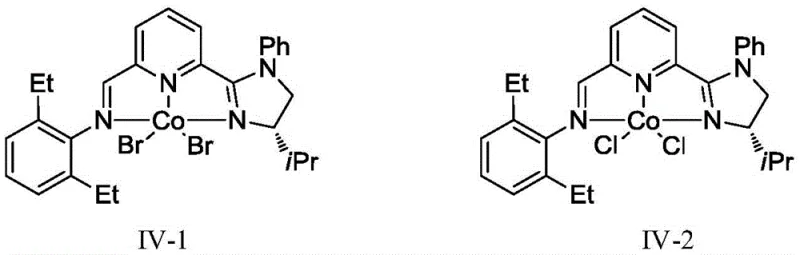
Mechanistic Insights into Co-IIP Catalyzed Asymmetric Hydrogenation
The heart of this transformative technology is the chiral cobalt catalyst, specifically the CoX2-IIP complex where IIP stands for iminopyridine imidazoline. This tridentate nitrogen-containing ligand creates a rigid chiral environment around the cobalt center, which is essential for inducing high enantioselectivity during the hydrometalation step. The catalyst structure, as exemplified by compounds IV-1 and IV-2, features specific substituents on the imidazoline and phenyl rings that fine-tune the steric and electronic properties of the active site. This precise tuning allows the catalyst to differentiate between the prochiral faces of the trisubstituted olefin effectively, even when the starting material is a mixture of isomers. The mechanism likely involves the formation of a cobalt-hydride species initiated by the reducing agent, such as sodium triethylborohydride, which then inserts into the olefin double bond. The subsequent sigma-bond metathesis with the silane co-catalyst regenerates the active hydride species and releases the chiral alkane product. This cycle operates with high turnover efficiency, achieving conversions generally exceeding 99 percent.
Impurity control is another critical aspect where this catalytic system excels. Traditional noble metal catalysts often leave behind trace metal residues that are difficult to remove and strictly regulated in final drug substances. The use of cobalt, while still requiring removal, is generally more manageable and less toxic than rhodium or palladium residues. Moreover, the high chemoselectivity of the Co-IIP system ensures that other sensitive functional groups present on the substrate, such as esters, ethers, halogens, or heterocycles, remain intact during the hydrogenation process. The patent data demonstrates excellent functional group tolerance across a wide range of substrates, including those with electron-donating and electron-withdrawing groups. This robustness minimizes the formation of side products and simplifies the downstream purification process, leading to higher overall yields and purity profiles that meet stringent pharmaceutical standards.
How to Synthesize Chiral Alkyl Compounds Efficiently
The synthesis protocol described in the patent provides a straightforward and reproducible pathway for generating optically active alkanes. The process begins with the preparation of the catalyst, which involves mixing the chiral IIP ligand with anhydrous cobalt bromide or chloride in a dry solvent like tetrahydrofuran under an inert atmosphere. Once the catalyst is formed, it is employed in the hydrogenation reaction alongside the olefin substrate and a silane co-catalyst. The reaction is initiated by the addition of a reducing agent and maintained under a hydrogen atmosphere. Detailed standardized synthesis steps for implementing this technology in a laboratory or pilot plant setting are provided in the guide below.
- Prepare the reaction system by charging a dry vessel with the chiral CoX2-IIP complex catalyst (either CoBr2-IIP or CoCl2-IIP) and the trisubstituted olefin substrate.
- Add the silane co-catalyst (PMHS or Ph2SiH2) and organic solvent (preferably toluene), then introduce atmospheric hydrogen via a balloon.
- Initiate the reaction by adding a reducing agent such as sodium triethylborohydride, stir at room temperature or 0°C for 24 hours, and isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cobalt-catalyzed hydrogenation technology offers compelling strategic benefits that extend beyond simple chemical efficiency. The primary advantage lies in the significant reduction of raw material costs associated with the catalyst itself. By substituting expensive, strategically constrained noble metals like rhodium and ruthenium with abundant cobalt, manufacturers can insulate their production costs from the volatile fluctuations of the precious metals market. Furthermore, the ability to utilize E/Z mixed olefin feedstocks eliminates the need for purchasing or synthesizing expensive single-isomer starting materials. This flexibility allows supply chain teams to source cheaper, crude olefin mixtures directly from upstream suppliers, thereby reducing the overall cost of goods and shortening the lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of noble metals and the avoidance of isomer separation steps result in substantial cost savings throughout the manufacturing lifecycle. Without the need for complex chromatographic purification of starting materials or expensive metal scavengers for product cleanup, the operational expenditure is drastically lowered. The use of atmospheric hydrogen and mild temperatures further reduces energy costs and infrastructure requirements, making the process economically viable for large-scale production.
- Enhanced Supply Chain Reliability: Relying on earth-abundant cobalt rather than scarce precious metals mitigates supply risk. Cobalt is widely available and not subject to the same geopolitical constraints as platinum group metals. Additionally, the tolerance for mixed feedstock means that supply chain disruptions affecting specific isomer availability are less likely to halt production. This robustness ensures a more stable and continuous supply of critical chiral intermediates for downstream drug manufacturing.
- Scalability and Environmental Compliance: The process is inherently scalable due to its mild operating conditions and simple workup procedures. The high atom economy and reduced waste generation align with modern environmental regulations and sustainability goals. By minimizing the use of hazardous reagents and avoiding the generation of heavy metal waste streams associated with noble metals, facilities can achieve easier regulatory compliance and reduce the environmental footprint of their chemical operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and reliability for potential partners.
Q: Can this method utilize E/Z mixed olefin feedstocks directly?
A: Yes, a key innovation of patent CN110655456A is the ability to use E/Z mixed trisubstituted olefins without prior separation, achieving high enantioselectivity (70-98% ee) regardless of the isomeric ratio.
Q: What are the advantages of using Cobalt over traditional Noble Metal catalysts?
A: Cobalt is an earth-abundant, inexpensive metal compared to Rhodium, Ruthenium, or Iridium. This switch significantly reduces raw material costs and eliminates concerns regarding heavy metal toxicity in final pharmaceutical products.
Q: What represent the typical reaction conditions for this hydrogenation process?
A: The process operates under mild conditions, typically at room temperature or 0°C using atmospheric hydrogen pressure. It utilizes silanes as co-catalysts and simple workup procedures like column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alkyl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the cobalt-catalyzed asymmetric hydrogenation technology described in CN110655456A for the production of high-value chiral intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral alkyl compounds delivered meets the highest quality standards required by global pharmaceutical clients.
We invite you to collaborate with us to leverage this cost-effective and sustainable synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this cobalt-based method can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a reliable supply of high-purity chiral building blocks for your drug development pipeline.
