Advanced Cobalt-Catalyzed Asymmetric Hydrogenation for Scalable Chiral Alkyl Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient and cost-effective routes to access chiral building blocks, which are essential for the synthesis of bioactive molecules. Patent CN110655456B introduces a groundbreaking methodology for the preparation of chiral alkyl compounds through the asymmetric catalytic hydrogenation of trisubstituted olefins. Unlike traditional methods that rely heavily on scarce and expensive noble metals such as rhodium, ruthenium, or iridium, this invention utilizes a cobalt-based catalytic system. The core innovation lies in the use of a specific iminopyridine-imidazoline (IIP) tridentate ligand coordinated with cobalt halides, which enables high enantioselectivity and conversion rates even when starting from E/Z mixed olefin substrates. This technological leap addresses critical pain points in process chemistry, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates while adhering to green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of non-functionalized trisubstituted olefins has been a formidable challenge in organic synthesis. Conventional protocols predominantly depend on precious metal catalysts, which not only inflate the production costs due to the volatility of metal prices but also introduce significant regulatory hurdles regarding residual metal limits in final drug substances. Furthermore, a major bottleneck in existing literature is the strict requirement for stereochemically pure starting materials. To achieve high optical purity in the product, manufacturers were previously forced to synthesize or separate single-configuration E or Z olefins, a process that often involves multi-step synthetic routes, chromatographic separations, and substantial waste generation. This reliance on geometrically pure substrates severely limits atom economy and complicates the supply chain for high-purity pharmaceutical intermediates, making large-scale production economically unviable for many candidates.
The Novel Approach
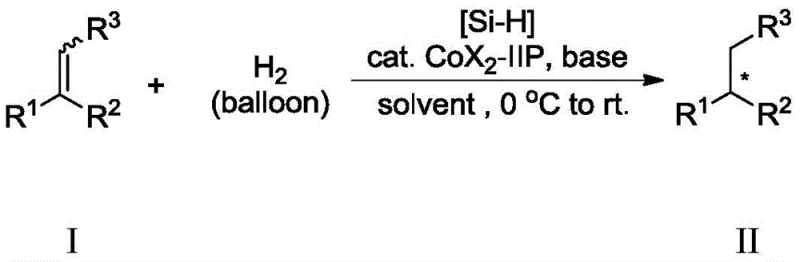
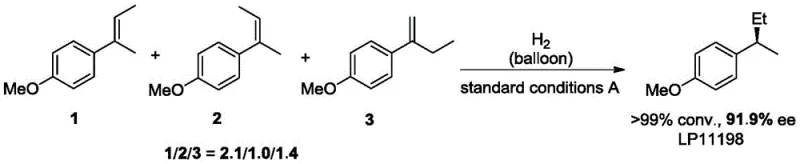
The methodology disclosed in CN110655456B fundamentally disrupts these limitations by demonstrating that geometric purity of the starting olefin is not a prerequisite for high stereoselectivity in the product. The novel cobalt-IIP catalyst system exhibits remarkable tolerance towards E/Z mixtures, allowing manufacturers to utilize crude or mixed olefin streams directly without energy-intensive purification steps. As illustrated in the reaction scope, the system successfully converts various trisubstituted olefins into chiral alkanes with conversions generally exceeding 99% and enantiomeric excess (ee) values ranging from 70% to 98%. This capability to process mixed isomers translates directly into cost reduction in pharmaceutical intermediate manufacturing by simplifying the upstream synthesis and eliminating separation losses. The use of atmospheric hydrogen and mild temperatures further enhances the safety profile and operational simplicity of the process.
Mechanistic Insights into Co-IIP Catalyzed Asymmetric Hydrogenation
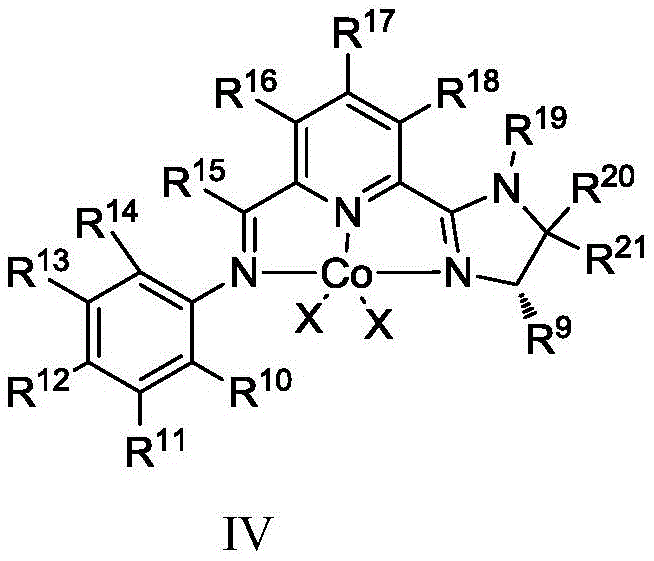
The success of this transformation hinges on the unique electronic and steric properties of the chiral CoX2-IIP complex. The catalyst features a tridentate nitrogen-containing ligand framework that creates a well-defined chiral pocket around the cobalt center. This specific coordination geometry is crucial for differentiating the prochiral faces of the trisubstituted olefin during the hydrometalation step. The presence of the imidazoline moiety, combined with the iminopyridine backbone, provides rigid structural control that ensures consistent stereochemical outcomes regardless of the E/Z ratio of the input material. The mechanism likely involves the in situ generation of a cobalt-hydride species facilitated by the silane co-catalyst and the reducing agent, which then undergoes migratory insertion with the olefin substrate. The subsequent protonolysis or sigma-bond metathesis releases the chiral alkane product and regenerates the active catalytic species.
From an impurity control perspective, the high selectivity of the Co-IIP system minimizes the formation of regioisomers and over-reduced byproducts, which are common issues in non-selective hydrogenations. The tolerance for diverse functional groups—including ethers, halogens, esters, and heterocycles—means that late-stage functionalization strategies can be employed without protecting group manipulation. This robustness is vital for R&D teams aiming to streamline synthetic routes for drug candidates. By avoiding the use of toxic transition metals like palladium or platinum, the process also simplifies the downstream purification workflow, reducing the burden on quality control laboratories to meet stringent heavy metal specifications. The ability to operate at 0°C to room temperature further prevents thermal degradation of sensitive intermediates, ensuring the integrity of the final fine chemical intermediates.
How to Synthesize Chiral Alkyl Compounds Efficiently
The synthesis protocol outlined in the patent provides a straightforward procedure accessible to standard laboratory setups. The process typically involves charging a reaction vessel with the chiral cobalt catalyst, the olefin substrate, and a silane additive in a solvent such as toluene. A reducing agent, preferably sodium triethylborohydride, is added to activate the system, followed by the introduction of hydrogen gas at atmospheric pressure. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction vessel with the chiral CoX2-IIP catalyst (e.g., CoBr2-IIP or CoCl2-IIP) and the trisubstituted olefin substrate in an organic solvent like toluene.
- Add the silane co-catalyst (PMHS or Ph2SiH2) and the reducing agent (such as sodium triethylborohydride) under an inert atmosphere.
- Introduce atmospheric hydrogen via a balloon and stir the mixture at 0°C to room temperature for 24 to 48 hours to achieve high conversion and enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cobalt-catalyzed technology offers transformative economic and logistical benefits. The shift from precious metals to earth-abundant cobalt drastically reduces the raw material cost base, insulating production budgets from the volatility associated with rhodium and iridium markets. Moreover, the elimination of the need for geometrically pure olefin feedstocks simplifies the sourcing strategy, allowing manufacturers to purchase cheaper, mixed-isomer raw materials from bulk chemical suppliers. This flexibility enhances supply chain resilience by broadening the vendor base and reducing dependency on specialized custom synthesis providers who charge premiums for isomer separation services.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by multiple factors beyond just the catalyst cost. By removing the requirement for E/Z separation, the overall step count in the synthetic route is effectively reduced, leading to significant savings in labor, solvent consumption, and waste disposal. The high atom economy of the hydrogenation reaction ensures that the majority of the starting mass is incorporated into the final product, minimizing material loss. Additionally, the use of atmospheric hydrogen eliminates the need for expensive high-pressure reactor equipment, lowering capital expenditure requirements for plant upgrades. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS) for the final chiral alkyl intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for pharmaceutical manufacturing, and this process improves reliability by utilizing widely available reagents. Cobalt salts and silane reducing agents are commodity chemicals with stable global supply chains, unlike specialized chiral phosphine ligands which may have limited production capacity. The mild reaction conditions (0°C to room temperature) also reduce the risk of batch failures due to thermal runaway or equipment malfunction, ensuring consistent delivery schedules. Furthermore, the robustness of the catalyst across a wide substrate scope means that a single catalytic system can be platformed for multiple products, simplifying inventory management and reducing the complexity of the chemical supply chain.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the simplicity of the operation and the absence of hazardous high-pressure conditions. The environmental profile is significantly improved by replacing toxic noble metals with less hazardous cobalt, which eases the burden of wastewater treatment and regulatory compliance regarding heavy metal discharge. The high conversion rates (>99%) mean that unreacted starting material recycling loops are often unnecessary, further reducing the environmental footprint. This alignment with green chemistry principles supports corporate sustainability goals and facilitates smoother regulatory approvals for new drug applications that rely on these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and claims within patent CN110655456B, providing a reliable basis for feasibility assessments.
Q: Can this method handle E/Z mixed olefin substrates?
A: Yes, a key innovation of patent CN110655456B is the ability to use E/Z mixed trisubstituted olefins directly without prior separation, maintaining high enantioselectivity (70%-98% ee).
Q: What are the advantages of using Cobalt over Noble Metals?
A: Cobalt is an earth-abundant, inexpensive metal compared to Rhodium, Ruthenium, or Iridium. This significantly reduces raw material costs and eliminates concerns regarding heavy metal toxicity in pharmaceutical products.
Q: What are the typical reaction conditions for this hydrogenation?
A: The reaction operates under mild conditions, typically using atmospheric hydrogen pressure (balloon), temperatures ranging from 0°C to room temperature, and common organic solvents like toluene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alkyl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies in driving down costs and accelerating time-to-market for our clients. Our technical team has thoroughly analyzed the Co-IIP hydrogenation pathway and is fully equipped to translate this academic innovation into industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity observed in the lab is maintained at the plant scale. Our facility is outfitted with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical integrity of every batch of chiral alkyl intermediates we produce.
We invite potential partners to engage with our technical procurement team to discuss how this cobalt-catalyzed route can be integrated into your specific supply chain. Whether you require a Customized Cost-Saving Analysis comparing this method to your current noble metal processes or need specific COA data for validation purposes, we are ready to provide comprehensive support. Let us collaborate to optimize your route feasibility assessments and secure a sustainable, cost-effective supply of these critical building blocks for your next-generation therapeutics.
