Advanced Synthesis of Tetrabenazine Intermediate: A Cost-Effective Route for High-Purity API Manufacturing
The pharmaceutical landscape for treating neurodegenerative disorders continues to evolve, with Tetrabenazine (marketed as Xenazine) remaining a cornerstone therapy for managing chorea associated with Huntington's disease. As a reversible inhibitor of vesicular monoamine transporter 2 (VMAT2), Tetrabenazine effectively depletes presynaptic dopamine, yet its complex synthesis presents significant challenges for generic manufacturers and supply chain stakeholders. The critical bottleneck lies in the efficient production of its key precursor, 6,7-dimethoxy-3,4-dihydroisoquinoline hydrochloride. Recent intellectual property developments, specifically patent CN111960999B, disclose a transformative synthetic methodology that addresses long-standing issues regarding yield, purity, and environmental safety. This technical insight report analyzes the proprietary route disclosed in the patent, highlighting its potential to redefine the cost structure and reliability of the global supply chain for this vital CNS intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
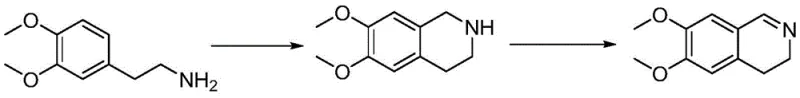
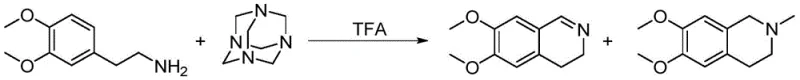
Historically, the industrial synthesis of 6,7-dimethoxy-3,4-dihydroisoquinoline has been plagued by inefficient chemistries that compromise both economic viability and operational safety. Early literature describes routes relying on noble metal catalysts, which not only inflate raw material costs but also introduce the risk of heavy metal contamination, necessitating expensive purification steps to meet regulatory limits. Another prevalent method involves the use of hexamethylenetetramine (urotropine) in trifluoroacetic acid (TFA); while effective, this approach suffers from the generation of approximately 10% N-methylated impurities, which are structurally similar and notoriously difficult to separate, thereby jeopardizing the final API quality.
Furthermore, the most common industrial practice employs aggressive chlorinating agents such as phosphorus oxychloride (POCl3), phosphorus pentachloride, or thionyl chloride for the cyclization step. These reagents are highly corrosive and release substantial quantities of hydrogen chloride gas, creating severe corrosion issues for reactor vessels and requiring sophisticated scrubbing systems. The reaction mixtures often turn dark and sticky due to polymerization side reactions, resulting in yields as low as 50% and complicating downstream isolation. Additionally, the quenching of these exothermic reactions poses significant safety hazards, while alternative reagents like oxalyl chloride release toxic carbon monoxide, presenting unacceptable environmental liabilities for modern green manufacturing facilities.
The Novel Approach
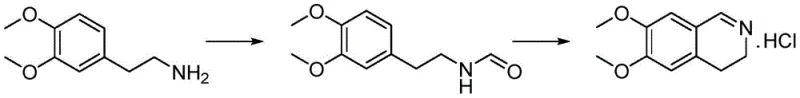
In stark contrast to these hazardous and low-yielding traditions, the method disclosed in CN111960999B introduces a refined two-step sequence that prioritizes atom economy and operational simplicity. The process initiates with a mild aminolysis reaction between 3,4-dimethoxyphenethylamine and ethyl formate to generate the formamide intermediate, avoiding the need for exotic reagents. The subsequent cyclization utilizes phthaloyl chloride as the dehydrating agent, a strategic choice that fundamentally alters the reaction profile. Unlike liquid or gaseous byproducts generated by traditional reagents, the reaction with phthaloyl chloride produces phthalic anhydride, a solid byproduct that precipitates or remains easily separable, drastically simplifying the workup procedure.
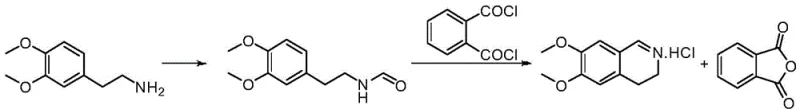
This innovative pathway operates under significantly milder thermal conditions, typically between 20°C and 30°C for the cyclization step, which minimizes thermal degradation and the formation of colored polymeric impurities. The result is a crystalline product that can be purified to exceptional standards through simple recrystallization, achieving purity levels exceeding 99.50% with single impurities controlled below 0.1%. By eliminating the need for noble metals and avoiding the generation of sticky tars or toxic gases, this method represents a paradigm shift towards safer, more sustainable, and higher-yielding manufacturing of this critical pharmaceutical building block.
Mechanistic Insights into Phthaloyl Chloride-Mediated Cyclization
The core innovation of this synthetic strategy lies in the mechanistic role of phthaloyl chloride during the Bischler-Napieralski type cyclization. In traditional mechanisms using POCl3, the activation of the amide oxygen leads to an imidoyl chloride intermediate, but the concomitant release of HCl and phosphate species often promotes side reactions. In the patented route, phthaloyl chloride acts as a potent electrophilic activator that converts the formamide carbonyl into a highly reactive imidoyl chloride species in situ. The adjacent aromatic ring then undergoes intramolecular electrophilic substitution to close the dihydroisoquinoline ring. Crucially, the leaving group from the phthaloyl chloride moiety cyclizes internally to form phthalic anhydride.
This mechanistic pathway offers distinct advantages for impurity control. The formation of phthalic anhydride is thermodynamically favorable and results in a chemically distinct solid that does not co-elute with the basic isoquinoline product during acidic workup or filtration. This inherent orthogonality in physical properties ensures that the final product is free from the dark, tarry residues characteristic of phosphorus-based cyclizations. Furthermore, the mild acidity of the reaction environment prevents the over-oxidation of the electron-rich dimethoxy-substituted ring, a common failure mode in oxidative cyclization methods, thereby preserving the integrity of the aromatic system essential for downstream biological activity.
How to Synthesize 6,7-Dimethoxy-3,4-dihydroisoquinoline Hydrochloride Efficiently
The implementation of this novel synthetic route requires precise control over reaction parameters to maximize the benefits of yield and purity described in the patent data. The process is designed to be robust, utilizing commercially available starting materials and standard unit operations found in most multipurpose pharmaceutical plants. The following overview outlines the critical stages of the synthesis, emphasizing the conditions required to achieve the reported high-quality specifications suitable for cGMP production.
- Mix 3,4-dimethoxyphenethylamine with ethyl formate and heat to 60-65°C to form N-(3,4-dimethoxyphenethyl)formamide.
- Dissolve the crude formamide in dichloromethane and react with phthaloyl chloride at 20-30°C to induce ring closure.
- Filter the precipitated solid, recover phthalic anhydride from the mother liquor, and recrystallize the product using isopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN111960999B offers compelling strategic advantages beyond mere technical elegance. The shift away from noble metal catalysts and hazardous chlorinating agents directly translates into a more resilient and cost-effective supply chain. By utilizing commodity chemicals like ethyl formate and phthaloyl chloride, manufacturers can mitigate the volatility associated with specialized reagent sourcing, ensuring consistent production schedules and reducing the risk of supply disruptions caused by regulatory restrictions on hazardous materials.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts removes a significant line item from the bill of materials, while the avoidance of complex heavy metal scavenging steps reduces downstream processing costs. Additionally, the ability to recover and recycle phthalic anhydride from the mother liquor further enhances the overall material efficiency, driving down the cost per kilogram of the final intermediate significantly compared to legacy processes.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous reagents simplifies logistics and storage requirements. Unlike processes requiring strict anhydrous conditions or cryogenic temperatures, this method operates at near-ambient temperatures, reducing energy consumption and equipment stress. The robustness of the chemistry ensures high batch-to-batch consistency, minimizing the risk of failed batches that could disrupt the supply of the final API to patients.
- Scalability and Environmental Compliance: The generation of solid byproducts rather than noxious gases aligns perfectly with increasingly stringent environmental regulations. The simplified waste stream, primarily consisting of recoverable solvents and phthalic anhydride, reduces the burden on wastewater treatment facilities and lowers the cost of hazardous waste disposal. This environmental compatibility facilitates easier permitting for capacity expansion and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms traditional alternatives in real-world manufacturing scenarios.
Q: What are the advantages of using phthaloyl chloride over phosphorus oxychloride for this cyclization?
A: Phthaloyl chloride offers milder reaction conditions and generates phthalic anhydride as a solid byproduct, which is easily separated by filtration. In contrast, phosphorus oxychloride releases hazardous HCl gas, creates sticky dark impurities, and poses significant exothermic risks during quenching.
Q: How does this method improve the purity profile for cGMP manufacturing?
A: The novel route avoids the formation of N-methyl impurities common in urotropine-based methods and prevents over-oxidation byproducts seen in noble metal catalysis. The process consistently achieves purity levels exceeding 99.50% with single impurities below 0.1%.
Q: Is the solvent system scalable for industrial production?
A: Yes, the method utilizes common organic solvents like dichloromethane or acetonitrile, which are standard in industrial settings. Furthermore, the solvent and the phthalic anhydride byproduct can be efficiently recovered and recycled from the mother liquor, enhancing process sustainability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,7-Dimethoxy-3,4-dihydroisoquinoline Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN111960999B are fully realized in large-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of 6,7-dimethoxy-3,4-dihydroisoquinoline hydrochloride meets the exacting standards required for CNS drug development.
We invite pharmaceutical partners to leverage our technical proficiency to optimize their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of this novel chemistry can enhance your project's profitability and timeline.
