Advanced Oxytocin Synthesis: Revolutionizing API Manufacturing with One-Step Reverse Phase Chromatography
Advanced Oxytocin Synthesis: Revolutionizing API Manufacturing with One-Step Reverse Phase Chromatography
The pharmaceutical industry is constantly seeking more efficient pathways for the production of complex polypeptides, and the technology disclosed in patent CN106674332B represents a significant leap forward in the manufacturing of oxytocin. This groundbreaking preparation method shifts away from the cumbersome traditional solid-phase synthesis follow-ups, introducing a highly integrated approach that combines reverse phase cyclization, purification, and desalination into a single, seamless operation. By utilizing efficient liquid phase RP chromatography with specialized silica gel C18 fillers, this process addresses the critical pain points of low yield and bulky processing volumes that have long plagued peptide manufacturing. For R&D directors and supply chain leaders, this innovation signals a move towards more robust, scalable, and cost-effective production of this vital hormone, ensuring a more reliable supply of high-purity active pharmaceutical ingredients for global healthcare markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of oxytocin has relied heavily on solid-phase synthesis followed by cleavage to obtain the precursor crude product, which typically exists as a trifluoroacetate salt containing two free sulfhydryl groups. The conventional workflow necessitates a high-dilution cyclization step to facilitate the formation of the critical disulfide bond between the cysteine residues. This high-dilution requirement creates massive operational inefficiencies, as the sample concentration must be kept extremely low to prevent intermolecular polymerization, leading to excessively large reaction volumes. Consequently, the subsequent purification and salt-exchange steps become incredibly bulky and resource-intensive, creating a bottleneck that limits throughput and drives up the cost of goods sold. Furthermore, the separation of the cyclized product from linear impurities and salts often requires multiple discrete unit operations, increasing the risk of product degradation and yield loss during transfer and handling.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages the hydrophobic properties of the polypeptide backbone to achieve online cyclization directly on the chromatography column. Instead of diluting the precursor in a vast reactor, the crude product solution is loaded directly onto a preparative C18 column where the peptide is adsorbed onto the stationary phase. This adsorption effectively concentrates the reactants locally on the column surface, allowing the cyclization to proceed efficiently without the need for bulk high-dilution conditions. The method ingeniously integrates the oxidation step within the elution gradient by introducing a mobile phase containing hydrogen peroxide and a pH-adjusted buffer, which promotes the formation of the disulfide bond while simultaneously purifying the molecule. This consolidation of cyclization, purification, and desalination into one continuous flow process drastically simplifies the workflow, reduces solvent consumption, and enhances the overall stability of the sensitive peptide structure.
Mechanistic Insights into Reverse Phase Absorption Cyclization
The core of this technological breakthrough lies in the precise manipulation of the interaction between the oxytocin precursor and the reverse phase stationary phase. The precursor, characterized by the linear sequence Cys-Tyr-Ile-Gln-Asn-Cys-Pro-Leu-Gly-NH2, possesses significant hydrophobic character due to its amino acid side chains, which allows it to bind strongly to the C18 silica gel filler under high organic solvent conditions. When the precursor solution is loaded, the polypeptide is retained on the column while weakly bound acidic impurities and salts are washed away by the initial neutral mobile phase. This selective retention creates an ideal microenvironment for the subsequent chemical transformation, effectively isolating the target molecules from the bulk solution matrix.
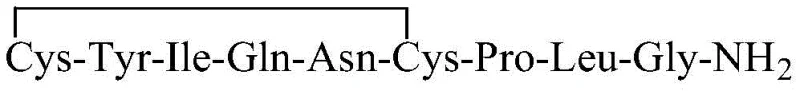
Following the initial wash, the introduction of the oxidative mobile phase triggers the critical mechanistic step: the formation of the intramolecular disulfide bond. The mobile phase, adjusted to a meta-alkaline pH range of 7.5 to 9.0 and supplemented with hydrogen peroxide, provides the necessary conditions for the oxidation of the two free sulfhydryl (-SH) groups on the cysteine residues. Because the peptide is anchored to the hydrophobic stationary phase, the effective local concentration of the thiol groups is sufficiently high to favor intramolecular cyclization over intermolecular dimerization or polymerization. As the gradient elution progresses, the newly formed cyclic oxytocin, now possessing a different hydrophobicity profile than the linear precursor, is eluted from the column. This simultaneous purification ensures that only the correctly folded and oxidized product is collected, while unreacted linear precursors and other byproducts remain retained or elute at different times, resulting in a final product with exceptional purity levels exceeding 99%.
How to Synthesize Oxytocin Efficiently
The implementation of this synthesis route requires precise control over chromatographic parameters to ensure consistent quality and yield. The process begins with the preparation of the precursor solution, typically dissolved in a low concentration of acetonitrile to facilitate loading onto the column. The operational protocol involves a carefully timed gradient elution sequence where the mobile phase composition shifts from loading conditions to oxidative conditions, and finally to high-strength elution to recover the product. This method eliminates the need for separate reactor vessels for cyclization and distinct columns for desalting, streamlining the entire manufacturing train. For detailed operational parameters and specific gradient profiles required to replicate this high-efficiency synthesis, please refer to the standardized guide below.
- Dissolve the oxytocin precursor crude product containing two free sulfhydryl groups in a 5% acetonitrile solution to form a 5g/L loading solution.
- Load the solution onto a C18 silica gel column and perform reverse phase cyclization by washing with neutral mobile phases to remove weakly bound acid ions.
- Elute using a mobile phase containing hydrogen peroxide and sodium hydroxide (pH 7.5-9.0) to promote disulfide bond formation, followed by gradient purification and desalination to collect the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this reverse phase chromatography method translates into tangible strategic advantages that go beyond mere technical elegance. The primary value driver is the drastic simplification of the manufacturing process, which directly impacts the cost structure and reliability of the supply chain. By collapsing three distinct processing steps—cyclization, purification, and desalination—into a single unit operation, manufacturers can significantly reduce the capital expenditure required for equipment and the operational expenditure associated with labor and facility usage. This leaner process architecture minimizes the physical footprint of the production line and reduces the complexity of validation and regulatory compliance, making it an attractive option for scaling up production to meet global demand.
- Cost Reduction in Manufacturing: The elimination of the high-dilution cyclization step is a major cost-saving mechanism, as it removes the need for massive reaction vessels and the corresponding vast quantities of solvents required to maintain low concentrations. In traditional processing, the recovery and recycling of these solvents represent a significant energy and cost burden; by avoiding this bulkiness, the new method substantially lowers utility costs and waste treatment expenses. Furthermore, the integration of desalination into the chromatographic run removes the need for separate ion-exchange or dialysis steps, further reducing material costs and processing time. These cumulative efficiencies lead to a more competitive cost base for the final API, allowing for better margin management in a price-sensitive pharmaceutical market.
- Enhanced Supply Chain Reliability: The continuous nature of this chromatographic process enhances supply chain resilience by reducing the number of potential failure points in the manufacturing workflow. Traditional batch processes with multiple transfer steps are prone to delays and yield losses at each handover point; conversely, this integrated flow system ensures a smoother, more predictable production cadence. The robustness of the C18 silica gel filler and the reproducibility of the gradient elution profile mean that batch-to-batch variability is minimized, ensuring a consistent supply of high-quality material. This reliability is crucial for maintaining uninterrupted production schedules for downstream formulation partners who depend on timely deliveries of critical hormones like oxytocin.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles and industrial expansion goals. The reduction in solvent volume directly correlates to a lower environmental impact, simplifying waste disposal and helping manufacturers meet increasingly stringent environmental regulations. Additionally, the process is inherently scalable; the use of preparative columns with dynamic axial compression allows for easy scale-up from laboratory pilot runs to multi-ton commercial production without fundamental changes to the chemistry. This scalability ensures that suppliers can rapidly respond to surges in market demand, securing the supply chain against shortages and positioning the manufacturer as a dependable long-term partner.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new preparation method is essential for stakeholders evaluating its potential impact on their supply chains. The following questions address common inquiries regarding the purity, scalability, and operational differences of this reverse phase technique compared to established industry standards. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a factual basis for decision-making.
Q: How does this reverse phase method improve upon traditional high-dilution cyclization?
A: Traditional methods require high-dilution conditions which result in bulky processing volumes and difficult purification. This novel method utilizes online reverse phase absorption to concentrate the sample on the column, allowing for cyclization, purification, and desalination in a single continuous step, significantly reducing solvent usage and processing time.
Q: What purity levels can be achieved with this chromatographic technique?
A: According to the patent data, this method consistently achieves HPLC purity levels exceeding 99.6%, effectively removing impurities and ensuring the final polypeptide meets stringent pharmaceutical standards without requiring multiple separate processing stages.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrialization and continuous production. By integrating cyclization and purification into one step using preparative columns (e.g., 50mm internal diameter), it eliminates the bottlenecks associated with batch-wise high-dilution reactions, making it highly scalable for commercial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxytocin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chromatographic techniques like the one described in CN106674332B for the production of high-value peptides. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial realities. Our facilities are equipped with state-of-the-art preparative chromatography systems and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of oxytocin we produce adheres to the highest international quality standards. We are committed to leveraging such cutting-edge technologies to deliver superior products that enhance the efficiency and reliability of our clients' pharmaceutical pipelines.
We invite procurement leaders and R&D directors to collaborate with us to explore how this optimized synthesis route can benefit your specific project requirements. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies of adopting this method for your supply chain. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your trusted partner in the commercialization of complex peptide APIs.
