Advanced Separation Technology for High-Purity Teicoplanin Single Components and Commercial Scalability
The pharmaceutical industry continuously seeks robust methodologies for isolating complex glycopeptide antibiotics with high fidelity, and the technology disclosed in patent CN102718843A represents a significant leap forward in the preparation of single teicoplanin components. Teicoplanin, a critical antimicrobial agent effective against Gram-positive bacteria including MRSA, exists naturally as a mixture of closely related structural analogs, which complicates its therapeutic application and toxicological study. Traditional separation techniques often struggle to resolve these analogs efficiently, leading to impure batches that fail to meet the stringent regulatory standards required for a reliable teicoplanin intermediate supplier. This patent introduces a novel chromatographic approach utilizing polymer microspheres that not only simplifies the workflow but also enhances the purity profile of the final active pharmaceutical ingredients. By integrating macroporous resin pretreatment with advanced polymer microsphere chromatography, the process achieves a level of resolution previously difficult to attain on a commercial scale, thereby addressing the critical needs of R&D directors focused on impurity control and supply chain heads concerned with production continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of teicoplanin components has relied heavily on solvent extraction and silica gel-based chromatography, methods that are fraught with significant operational inefficiencies and technical bottlenecks. Solvent extraction processes, such as those disclosed in European patent EP 0137506, involve the use of water-miscible organic solvents like acetone or acetonitrile in large volumes, creating substantial environmental burdens and safety hazards within the manufacturing facility. Furthermore, these conventional techniques often result in low product purity and require cumbersome steps such as repeated centrifugation and filtration, which drastically increase the lead time for high-purity antibiotics. Silica gel columns, while effective for analytical purposes, suffer from low recycling rates and high costs when applied to preparative scales, making them economically unviable for cost reduction in antibiotic manufacturing. The inability to precisely control the ratio of individual components like TA2-1 through TA2-5 in these traditional workflows often leads to batch-to-batch variability, posing a severe risk to the quality consistency required by global regulatory bodies.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data leverages a synergistic combination of macroporous adsorption resins and polymer microsphere chromatography to overcome these historical limitations. This method begins with a strategic pretreatment of the fermentation liquor where pH adjustment facilitates the release of intracellular teicoplanin, significantly boosting the initial yield before purification even begins. The subsequent use of macroporous resins for decolorization and adsorption effectively removes bulk impurities such as polysaccharides and pigments without the excessive solvent consumption associated with liquid-liquid extraction. The core innovation lies in the final chromatographic step where polymer microspheres serve as the stationary phase, allowing for the simultaneous separation of all six single components with high resolution. This streamlined technology not only simplifies the overall process flow but also ensures that the commercial scale-up of complex glycopeptides is feasible, offering a pathway to produce high-purity teicoplanin components that meet the exacting standards of modern pharmaceutical development.
Mechanistic Insights into Polymer Microsphere Chromatography
The success of this separation strategy hinges on the precise physicochemical interactions between the teicoplanin analogs and the polymer microsphere stationary phase under gradient elution conditions. Teicoplanin components differ subtly in the structure of their acyl side chains, which influences their hydrophobicity and interaction strength with the chromatography media. By employing a gradient elution system that transitions from lower to higher concentrations of aqueous organic solvents such as methanol or ethanol, the method exploits these minute differences in polarity to achieve baseline separation. The polymer microspheres, specifically types like PS25-300 or PS30RPC-300, provide a rigid and porous structure that minimizes swelling and shrinking during solvent changes, ensuring stable flow dynamics and consistent retention times. This stability is crucial for maintaining the integrity of the separation profile over long production runs, allowing for the collection of distinct fractions corresponding to TA2-1, TA2-2, TA2-3, TA2-4, TA2-5, and TA3-1 with minimal cross-contamination.
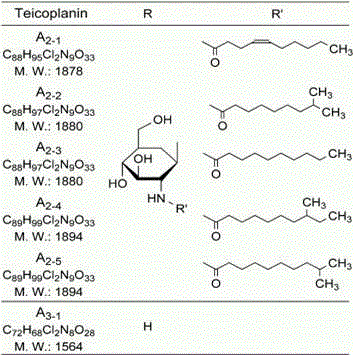
Understanding the structural nuances of these components is vital for R&D directors aiming to optimize the impurity profile of the final drug substance. As illustrated in the structural diagram, the variations in the R and R' groups among the six components dictate their biological activity and pharmacokinetic properties. The chromatographic method described ensures that each of these distinct molecular entities is isolated with a purity exceeding 95.0%, as confirmed by high-performance liquid chromatography (HPLC) analysis. This high level of purity is achieved through a multi-stage collection process where specific eluents are gathered based on real-time HPLC detection, allowing for the targeted isolation of minor components that might otherwise be lost in a crude mixture. Furthermore, the method includes a recycling step for mixed fractions, where components like TA2-2 and TA2-3 are re-injected for secondary separation, maximizing the overall recovery rate and ensuring that no valuable material is wasted during the purification process.
How to Synthesize Teicoplanin Single Components Efficiently
Implementing this synthesis route requires a disciplined approach to process parameters, particularly regarding pH control and solvent gradients, to ensure reproducible results across different batch sizes. The process initiates with the alkaline treatment of the fermentation broth to disrupt microbial cell walls, followed by neutralization to prepare the liquor for resin adsorption. Detailed standard operating procedures for the chromatographic separation involve precise loading concentrations and flow rates to prevent column overload and maintain resolution. For a comprehensive understanding of the specific operational parameters and equipment setup required to replicate this high-efficiency separation, please refer to the standardized synthesis steps outlined in the technical guide below.
- Pretreat teicoplanin fermentation liquor by adjusting pH to 11.0-13.0 with alkaline solution to disrupt cell walls, then neutralize filtrate to pH 7.0-7.5.
- Perform decolorization using macroporous decolorizing resin followed by adsorption and enrichment using macroporous adsorbent resin with gradient solvent desorption.
- Execute chromatographic separation on the crude extract using polymer microspheres with gradient elution to isolate six single components with purity greater than 95%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this polymer microsphere-based separation technology translates into tangible strategic advantages that extend beyond mere technical performance. The elimination of complex solvent extraction steps and the reduction in solvent volume directly contribute to a leaner manufacturing process, which inherently lowers the operational expenditure associated with waste disposal and raw material procurement. By utilizing robust macroporous resins that can be regenerated and reused, the process significantly reduces the consumption of consumables, leading to substantial cost savings over the lifecycle of the product. Moreover, the simplicity of the technology facilitates easier technology transfer and scale-up, reducing the risk of production delays and ensuring a more reliable teicoplanin intermediate supplier partnership. The ability to handle large sample quantities with consistent quality means that supply continuity can be maintained even during periods of high market demand, mitigating the risks associated with supply chain disruptions.
- Cost Reduction in Manufacturing: The process design inherently minimizes the use of expensive organic solvents and eliminates the need for high-cost preparative HPLC silica columns, which are often single-use or have limited lifespans. By replacing these with regenerable polymer microspheres and macroporous resins, the manufacturing cost per kilogram of active ingredient is drastically reduced. Additionally, the high recovery rate of the target components means that less starting fermentation broth is required to produce the same amount of final product, further optimizing the cost structure. This efficiency allows for competitive pricing strategies without compromising the margin, making it an attractive option for cost reduction in antibiotic manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the resin-based separation system ensures that production can be sustained over long periods without frequent column replacement or system downtime. The availability of the required macroporous resins and polymer microspheres from multiple global suppliers reduces the risk of raw material shortages, thereby enhancing the overall resilience of the supply chain. Furthermore, the simplified process flow reduces the number of potential failure points, leading to more predictable production schedules and shorter lead times for high-purity antibiotics. This reliability is critical for pharmaceutical companies that need to guarantee the availability of key intermediates for their downstream drug formulation processes.
- Scalability and Environmental Compliance: The technology is designed with scalability in mind, allowing for seamless transition from laboratory bench scale to multi-ton commercial production without significant process re-engineering. The reduction in solvent usage and the ability to recycle process materials align with increasingly stringent environmental regulations, reducing the carbon footprint of the manufacturing process. This environmental compliance not only mitigates regulatory risks but also enhances the corporate social responsibility profile of the supply chain. The ability to scale up complex glycopeptides efficiently ensures that the supply can grow in tandem with market demand, supporting the long-term strategic goals of global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this teicoplanin separation technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of adopting this method for industrial production. Understanding these details is essential for stakeholders evaluating the potential integration of this process into their existing manufacturing workflows to ensure alignment with quality and efficiency goals.
Q: What is the primary advantage of using polymer microspheres for teicoplanin separation?
A: The use of polymer microspheres allows for the simultaneous separation of all six single teicoplanin components (TA2-1 to TA3-1) from crude extract with high purity (>95%) and a total recovery rate exceeding 50%, significantly outperforming traditional silica gel methods.
Q: How does this method improve cost efficiency in antibiotic manufacturing?
A: This process eliminates the need for expensive preparative HPLC systems and reduces solvent consumption through efficient macroporous resin adsorption and desorption cycles, leading to substantial operational cost savings without compromising yield.
Q: Is this separation method suitable for large-scale industrial production?
A: Yes, the technology is designed for commercial scale-up, capable of handling large sample quantities with simple technology steps, ensuring consistent supply continuity and reduced lead time for high-purity antibiotics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Teicoplanin Single Components Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation antibiotics, and we possess the technical expertise to bring complex separation routes like the one described in CN102718843A to commercial fruition. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is smooth and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instruments to verify the identity and quality of every batch, guaranteeing that our teicoplanin components meet the highest global standards. Our commitment to technical excellence allows us to support our partners in overcoming the challenges associated with glycopeptide purification, delivering materials that facilitate faster drug development and regulatory approval.
We invite you to engage with our technical procurement team to discuss how our capabilities can align with your specific project requirements and supply chain objectives. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of partnering with us for your teicoplanin needs. We encourage you to reach out for specific COA data and route feasibility assessments, which will provide the concrete evidence needed to make informed sourcing decisions. Let us collaborate to optimize your supply chain and ensure the consistent availability of high-quality pharmaceutical intermediates for your critical projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
