Revolutionizing Brivaracetam Production: Advanced Asymmetric Catalysis for Scalable API Intermediate Manufacturing
Revolutionizing Brivaracetam Production: Advanced Asymmetric Catalysis for Scalable API Intermediate Manufacturing
The pharmaceutical industry is constantly seeking more efficient pathways for the synthesis of third-generation antiepileptic drugs, particularly Brivaracetam, a high-affinity SV2A ligand approved by the FDA. A groundbreaking development detailed in patent CN114751849A introduces a highly efficient preparation method for the critical intermediate, (R)-4-propyl-dihydrofuran-2-one, which serves as the chiral backbone for the final active pharmaceutical ingredient. This innovation leverages a sophisticated asymmetric Baeyer-Villiger oxidation reaction, marking a significant departure from legacy synthetic strategies that rely heavily on inefficient chiral resolution. By employing specialized metal-ligand catalysts, this technology enables the direct construction of the chiral lactone ring with exceptional enantiomeric excess, thereby addressing long-standing challenges in purity and yield that have plagued previous manufacturing attempts.
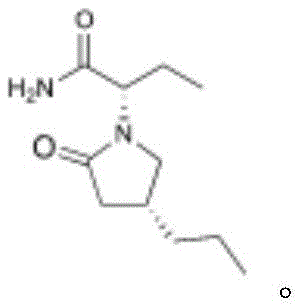
For R&D directors and process chemists, the structural integrity and optical purity of the intermediate are paramount, as they dictate the efficacy and safety profile of the final drug substance. The ability to access this key building block through a concise, catalytic asymmetric route represents a major technological leap forward in fine chemical synthesis. This report analyzes the technical merits of this novel approach, contrasting it with conventional methodologies to highlight its potential for transforming the supply chain dynamics of antiepileptic pharmaceutical intermediates. The integration of advanced organometallic catalysis not only streamlines the synthetic sequence but also aligns with modern green chemistry principles by minimizing waste generation and hazardous reagent usage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Brivaracetam has been hindered by reliance on racemic synthesis followed by chiral resolution, a process inherently flawed by poor atom economy and high operational costs. Traditional routes, such as those disclosed in earlier patents like CN1882535A, necessitate the separation of optical isomers using preparative chiral chromatography, which inevitably results in the discard of at least fifty percent of the synthesized material as the unwanted enantiomer. Furthermore, the physical properties of these optical isomers are often strikingly similar, making the separation process technically demanding, slow, and limited by the loading capacity of expensive chromatographic columns. This bottleneck severely restricts batch processing capabilities, rendering large-scale production economically unviable due to the exorbitant costs associated with stationary phases and solvent consumption. Additionally, alternative prior art methods involving chiral inducing groups like oxazolidinones introduce further complexity, requiring multiple assembly and removal steps that elongate the synthetic timeline and increase the overall cost of goods.
The Novel Approach
In stark contrast, the methodology presented in patent CN114751849A circumvents these inefficiencies by employing a direct asymmetric Baeyer-Villiger oxidation of 3-propylcyclobutanone to generate the chiral lactone intermediate with high fidelity. This innovative strategy utilizes a tailored catalytic system comprising chiral ligands complexed with metals such as Scandium, Palladium, or Silver, which effectively differentiate between enantiotopic faces of the substrate during the oxidation event. By installing the chirality at the early stage of the synthesis through catalysis rather than separation, the process achieves theoretical atom economy close to one hundred percent regarding the carbon skeleton utilization, eliminating the massive waste stream associated with racemic resolution. The reaction conditions are notably milder and safer, avoiding the extreme cryogenic temperatures and hazardous reagents like dimethyl sulfide borane found in older literature, thus facilitating a more robust and scalable operation. This paradigm shift from resolution to asymmetric synthesis fundamentally alters the economic landscape of Brivaracetam production, offering a pathway that is both commercially attractive and environmentally sustainable.
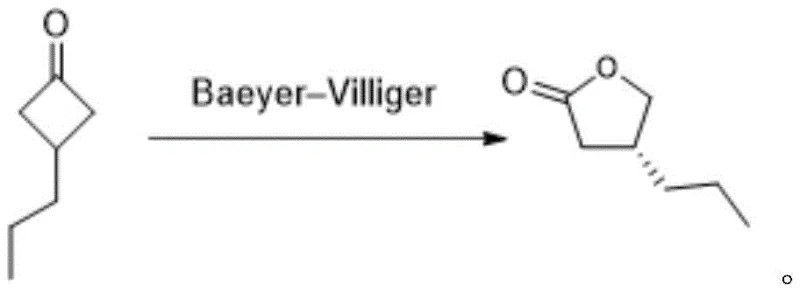
Mechanistic Insights into Metal-Catalyzed Asymmetric Baeyer-Villiger Oxidation
The success of this synthetic route hinges on the precise design of the catalytic species, which orchestrates the transfer of oxygen to the ketone substrate with high stereocontrol. The catalyst system typically involves a chiral ligand, such as the phosphoric acid derivatives or the bis-oxazoline type ligands described in the patent formulas, coordinated to a Lewis acidic metal center like Scandium triflate. Upon activation, this metal-ligand complex binds to the carbonyl oxygen of the 3-propylcyclobutanone, increasing its electrophilicity and organizing the transition state geometry to favor the formation of the (R)-enantiomer. The oxidizing agent, whether it be m-chloroperoxybenzoic acid or urea-hydrogen peroxide, then attacks the activated carbonyl in a concerted manner, guided by the steric bulk of the chiral ligand to ensure that the migration of the alkyl group occurs with retention of configuration and high enantioselectivity. This mechanistic precision allows for the achievement of enantiomeric excess values exceeding 99%, a level of purity that is difficult to attain through post-synthetic purification methods.
Furthermore, the choice of solvent and reaction temperature plays a critical role in modulating the activity and selectivity of the catalytic cycle. The patent data indicates that solvents such as ethyl acetate, tetrahydrofuran, or chloroform provide an optimal medium for solubilizing both the organic substrate and the metal catalyst while maintaining the stability of the peroxy-species involved. Operating at controlled low temperatures, typically ranging from -40°C to -20°C, helps to suppress background non-catalyzed reactions that would lead to racemic product formation, thereby locking in the chiral information provided by the catalyst. The robustness of this system is evidenced by its tolerance to various oxidizing agents and its ability to function with low catalyst loadings, suggesting a turnover number that is favorable for industrial application. Understanding these mechanistic nuances is essential for process engineers aiming to translate this laboratory-scale breakthrough into a GMP-compliant manufacturing process.
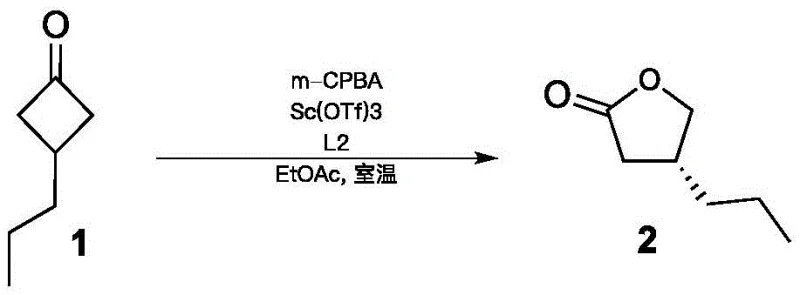
How to Synthesize (R)-4-propyl-dihydrofuran-2-one Efficiently
The practical implementation of this synthesis begins with the preparation of the achiral precursor, 3-propylcyclobutanone, which can be efficiently accessed from readily available commodity chemicals like 1-pentene and trichloroacetyl chloride via a zinc-mediated cycloaddition. Once the ketone substrate is secured, the critical asymmetric oxidation step is performed by pre-forming the catalyst complex in situ or using a pre-isolated catalyst species, ensuring consistent performance across batches. The reaction mixture is carefully monitored to prevent over-oxidation or degradation of the sensitive lactone product, followed by a straightforward workup involving quenching, extraction, and purification to isolate the high-purity intermediate. This intermediate is then coupled with L-2-aminobutanamide in a final amidation step to furnish the target Brivaracetam molecule, completing the synthesis in a remarkably short sequence that minimizes unit operations. For detailed operational parameters, stoichiometry, and specific purification protocols, please refer to the standardized synthesis guide below.
- Preparation of 3-propylcyclobutanone precursor via reaction of 1-pentene with trichloroacetyl chloride in the presence of zinc-copper couple and phosphorus oxychloride.
- Execution of asymmetric Baeyer-Villiger oxidation using a chiral metal catalyst complex (e.g., Scandium or Palladium based) and an oxidizing agent like m-CPBA or hydrogen peroxide at controlled low temperatures.
- Final coupling of the resulting (R)-4-propyl-dihydrofuran-2-one with L-2-aminobutanamide in a polar aprotic solvent system to yield high-purity Brivaracetam.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this asymmetric catalytic route offers transformative benefits that directly impact the bottom line and operational resilience of pharmaceutical manufacturing. By eliminating the need for chiral chromatography, companies can avoid the substantial capital expenditure and recurring costs associated with purchasing and regenerating expensive chiral stationary phases, which are often subject to supply constraints and price volatility. The drastic improvement in atom economy means that less raw material is required to produce the same amount of active ingredient, leading to significant reductions in the cost of goods sold and a smaller environmental footprint due to reduced solvent and waste disposal requirements. Moreover, the simplified process flow reduces the number of processing steps and hold times, allowing for faster throughput and shorter lead times for delivering high-purity pharmaceutical intermediates to downstream formulation sites. This efficiency gain is crucial for maintaining a competitive edge in the generic drug market where margin pressure is intense and speed to market is a key differentiator.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps removes a major cost driver, as chromatographic separations are notoriously expensive due to low throughput and high solvent consumption. By shifting to a catalytic asymmetric synthesis, the process achieves near-quantitative utilization of the carbon skeleton, meaning that the cost of starting materials is amortized over a much higher yield of useful product. Additionally, the avoidance of specialized chiral auxiliaries that require installation and removal further simplifies the bill of materials, reducing the complexity of inventory management and lowering the overall procurement spend on exotic reagents. These cumulative savings create a substantial cost advantage that can be passed on to customers or reinvested into further process optimization.
- Enhanced Supply Chain Reliability: The reliance on commodity starting materials such as 1-pentene and common oxidizing agents ensures a stable and diversified supply base, mitigating the risk of disruptions caused by single-source dependencies on specialized chiral pool materials. The robustness of the catalytic system allows for flexible manufacturing schedules, as the reaction is less sensitive to minor variations in conditions compared to delicate enzymatic or resolution processes. This reliability translates into more predictable delivery timelines for API manufacturers, enabling them to better plan their production cycles and meet market demand without the fear of unexpected yield drops or purification bottlenecks. Consequently, supply chain managers can operate with lower safety stock levels, freeing up working capital and improving overall logistics efficiency.
- Scalability and Environmental Compliance: The transition from batch chromatography to continuous or large-batch catalytic reactions significantly enhances the scalability of the process, allowing for seamless expansion from pilot plant to commercial tonnage production without fundamental changes to the chemistry. The use of safer oxidants and the reduction of hazardous waste streams align with increasingly stringent environmental regulations, reducing the liability and permitting hurdles associated with chemical manufacturing. This green chemistry profile not only improves the corporate sustainability metrics but also future-proofs the manufacturing site against evolving regulatory landscapes regarding solvent emissions and waste treatment. Such compliance is increasingly becoming a prerequisite for qualifying as a supplier to major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Brivaracetam synthesis pathway, derived from the specific embodiments and comparative data provided in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this asymmetric catalytic technology for large-scale API production. Stakeholders are encouraged to review these points to understand how this method resolves specific pain points associated with legacy manufacturing routes.
Q: How does this new method improve upon traditional chiral resolution techniques?
A: Traditional methods often involve synthesizing a racemic mixture followed by chiral chromatography, which discards at least 50% of the material and requires expensive columns. This patented asymmetric catalysis route directly synthesizes the desired (R)-enantiomer with up to 99.8% ee, drastically improving atom economy and eliminating the need for wasteful separation processes.
Q: What are the safety advantages of this catalytic system compared to prior art?
A: Previous synthetic routes often utilized hazardous reagents such as dimethyl sulfide borane or required extremely low temperatures (-70°C) that are energy-intensive and difficult to maintain on scale. This method employs milder reaction conditions (e.g., -40°C to room temperature) and safer oxidizing agents, significantly reducing operational risks and environmental hazards associated with explosive peroxides or toxic byproducts.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. By removing the bottleneck of chiral column chromatography and utilizing robust metal-ligand catalysts that function effectively at moderate concentrations, the route supports continuous processing and large-batch manufacturing, ensuring a reliable supply chain for high-volume API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the asymmetric Baeyer-Villiger oxidation described in CN114751849A can be successfully translated from the laboratory to the manufacturing floor. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation for chiral and impurity profiling. Our infrastructure is designed to handle complex catalytic reactions safely and efficiently, providing our partners with a secure and reliable source for their key building blocks.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain to drive down costs and improve quality. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance your operational efficiency and market position.
