Advanced Semi-Synthesis Of Taxane Intermediates For Commercial Scale-Up Of Complex Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and scalable methods for producing high-value anticancer agents, and patent CN1942458A presents a significant advancement in the semi-synthesis of taxane intermediates. This intellectual property details innovative pathways for converting cephalomannine and baccatin III intermediates into potent antineoplastic agents such as paclitaxel and docetaxel. For R&D Directors and Procurement Managers, understanding the nuances of this technology is critical for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent quality demands. The patent outlines specific chemical transformations that bypass traditional bottlenecks, offering a strategic advantage in the competitive landscape of API manufacturing. By leveraging naturally abundant taxanes like cephalomannine found in Taxus species needles, this method reduces reliance on scarce bark resources, thereby enhancing supply chain continuity. The technical depth provided in this document serves as a foundation for evaluating the feasibility of integrating these novel routes into existing production frameworks, ensuring both economic viability and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional semi-synthetic routes for producing docetaxel and paclitaxel have historically relied heavily on 10-deacetylbaccatin III (10-DAB) as the primary starting material, which presents significant logistical and economic challenges. The extraction of 10-DAB often requires substantial quantities of plant biomass, leading to environmental concerns and fluctuating raw material costs that impact cost reduction in API manufacturing. Furthermore, the esterification of the C-13 side chain onto the baccatin core frequently involves complex protection and deprotection strategies that increase the number of synthetic steps and reduce overall yield. These multi-step processes often generate substantial chemical waste and require expensive reagents, creating bottlenecks for supply chain heads aiming for reducing lead time for high-purity pharmaceutical intermediates. The reliance on specific natural sources also introduces variability in raw material quality, complicating the consistency required for commercial scale-up of complex pharmaceutical intermediates. Consequently, there is an urgent need for alternative pathways that utilize more accessible precursors while maintaining high stereochemical control.
The Novel Approach
The methodology described in CN1942458A introduces a transformative approach by utilizing cephalomannine as a versatile precursor for generating taxane intermediates through aziridine and epoxide analogs. This strategy effectively modifies the existing side chain of cephalomannine rather than constructing it from scratch, significantly streamlining the synthesis process. By converting the double bond in the cephalomannine side chain into reactive intermediates like aziridines, the process allows for precise manipulation of the C-13 position to match the structural requirements of paclitaxel and docetaxel. This innovation not only simplifies the synthetic route but also enhances the atom economy of the reaction, contributing to substantial cost savings in production. The ability to derive high-purity pharmaceutical intermediates from cephalomannine, which is often more readily available than 10-DAB in certain Taxus species, provides a strategic buffer against supply chain disruptions. For procurement teams, this represents a viable pathway to diversify sourcing strategies and mitigate risks associated with single-source raw material dependencies.
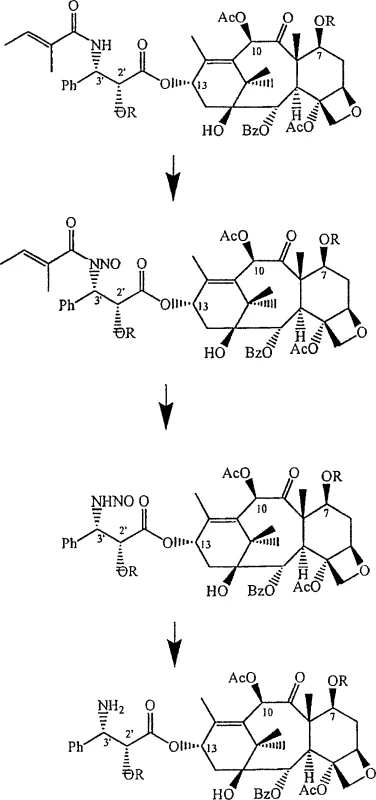
Mechanistic Insights into Cephalomannine Aziridine Conversion
The core chemical innovation lies in the precise transformation of the cephalomannine side chain, specifically targeting the olefinic bond for functionalization to create a primary amine taxane intermediate. The process involves converting cephalomannine into aziridine analogs using reagents such as chloramine-T in the presence of copper triflate, which facilitates the formation of the three-membered nitrogen-containing ring with high stereoselectivity. This aziridine intermediate serves as a pivotal junction, allowing for subsequent ring-opening reactions that install the necessary amine functionality at the C-3' position. The mechanistic pathway ensures that the stereochemistry at the chiral centers is preserved or controlled, which is paramount for the biological activity of the final anticancer agents. Detailed analysis of the reaction conditions reveals that maintaining anhydrous environments and specific temperature controls is essential to prevent side reactions that could compromise the purity of the intermediates. This level of mechanistic control is crucial for R&D teams focused on impurity profiling and ensuring that the final API meets rigorous pharmacopeial standards.
Furthermore, the patent describes alternative pathways involving the formation of epoxide analogs followed by conversion to azidoalcohol analogs, offering flexibility in process optimization. In this route, cephalomannine is treated with MCPBA to form an epoxide, which is then reacted with sodium azide to open the ring and introduce the nitrogen functionality. This multi-step sequence allows for fine-tuning of reaction parameters to maximize yield and minimize byproduct formation. The subsequent reduction of the azido group to a primary amine using Raney-nickel or other reducing agents completes the transformation of the side chain. Understanding these mechanistic details enables technical teams to troubleshoot potential scale-up issues and optimize reaction kinetics for industrial applications. The robustness of these chemical transformations underscores the potential for this technology to serve as a backbone for reliable pharmaceutical intermediates supplier operations, ensuring consistent quality across large production batches.
How to Synthesize Taxane Intermediates Efficiently
The synthesis of taxane intermediates from cephalomannine involves a series of carefully controlled chemical reactions that transform the natural product into a versatile precursor for paclitaxel and docetaxel. The process begins with the activation of the side-chain double bond, followed by functional group interconversion to establish the required amine moiety. Detailed operational protocols emphasize the importance of solvent selection, such as using dry acetonitrile or THF, to maintain reaction integrity and prevent hydrolysis of sensitive intermediates. Temperature management is also critical, with specific steps requiring cooling to 0°C or heating to 65°C to drive reactions to completion without degrading the taxane core. The final steps involve deprotection and purification via column chromatography to isolate the high-purity pharmaceutical intermediates needed for downstream API synthesis. For detailed standardized synthesis steps, please refer to the guide below.
- Convert cephalomannine into aziridine analogs or epoxide analogs through specific chemical reactions involving chloramine-T or MCPBA.
- Hydrolyze the resulting analogs to obtain primary amine taxane intermediates using lithium hydroxide or formic acid under controlled conditions.
- Transform the primary amine taxane intermediates into final active agents like paclitaxel or docetaxel through esterification and deprotection steps.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the semi-synthetic routes described in this patent offers significant strategic benefits for procurement and supply chain management within the pharmaceutical sector. The ability to utilize cephalomannine, which can be extracted from the needles and leaves of Taxus species rather than the bark, aligns with sustainable sourcing initiatives and reduces the ecological impact of raw material harvesting. This shift in raw material sourcing enhances supply chain reliability by tapping into more renewable plant parts, thereby ensuring a more consistent flow of starting materials for commercial scale-up of complex pharmaceutical intermediates. Additionally, the streamlined synthetic pathway reduces the number of processing steps required to reach the key intermediate, which directly correlates to lower operational costs and reduced energy consumption. For procurement managers, this translates into a more cost-effective supply base that can withstand market volatility and raw material price fluctuations.
- Cost Reduction in Manufacturing: The novel process eliminates the need for complex total synthesis of the side chain, leveraging the existing structure of cephalomannine to significantly reduce reagent costs and processing time. By avoiding expensive chiral catalysts and multi-step protection schemes associated with traditional methods, the overall manufacturing cost is drastically simplified. This efficiency allows for substantial cost savings in production without compromising the quality of the high-purity pharmaceutical intermediates. The reduction in synthetic steps also minimizes waste generation, leading to lower disposal costs and a smaller environmental footprint, which is increasingly important for regulatory compliance and corporate sustainability goals.
- Enhanced Supply Chain Reliability: Diversifying the source of taxane precursors from bark-dependent 10-DAB to needle-derived cephalomannine mitigates the risk of supply shortages caused by seasonal variations or regulatory restrictions on tree harvesting. This flexibility ensures a more stable supply of raw materials, reducing lead time for high-purity pharmaceutical intermediates and enabling manufacturers to meet tight delivery schedules. The robustness of the chemical process also means that production can be scaled up more predictably, providing supply chain heads with greater confidence in long-term planning and inventory management. This reliability is crucial for maintaining continuous production lines for critical oncology medications.
- Scalability and Environmental Compliance: The reaction conditions described utilize common industrial solvents and reagents that are readily available and manageable on a large scale, facilitating the commercial scale-up of complex pharmaceutical intermediates. The process design inherently reduces the generation of hazardous byproducts, simplifying waste treatment and ensuring adherence to strict environmental regulations. This compliance reduces the risk of production shutdowns due to environmental violations and enhances the company's reputation as a responsible manufacturer. Furthermore, the scalability of the process allows for rapid response to increased market demand, ensuring that the supply of life-saving medications remains uninterrupted during global health crises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this semi-synthetic technology in industrial settings. These answers are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of the described methods for stakeholders involved in API production. Understanding these details is essential for making informed decisions about technology adoption and supplier selection. The responses highlight the practical aspects of the chemistry and the strategic benefits for the supply chain.
Q: What are the primary starting materials for this taxane semi-synthesis process?
A: The process primarily utilizes cephalomannine, which can be extracted from various Taxus species needles and leaves, offering a more sustainable alternative to bark extraction for 10-deacetylbaccatin III.
Q: How does the aziridine analog route improve impurity control?
A: By converting the side-chain double bond into an aziridine ring, the method allows for precise regioselective ring opening, minimizing side reactions and ensuring high-purity pharmaceutical intermediates.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the reaction conditions described, such as using standard solvents like acetonitrile and THF at moderate temperatures, are designed to be scalable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced synthesis technologies in delivering high-quality oncology intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like those described in CN1942458A can be successfully translated into industrial reality. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of taxane intermediates meets the highest international standards. Our infrastructure is designed to support the complex chemistry required for aziridine analog conversion, providing a secure and reliable foundation for your API manufacturing needs.
We invite you to collaborate with us to optimize your supply chain and achieve significant operational efficiencies through the adoption of these advanced synthetic routes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production requirements, demonstrating how these methods can enhance your bottom line. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate the potential of this technology for your portfolio. Partnering with us ensures access to cutting-edge chemical solutions and a dependable supply of high-purity pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
