Advanced Pravastatin Purification: Scaling High-Purity API Production Efficiently
Advanced Pravastatin Purification: Scaling High-Purity API Production Efficiently
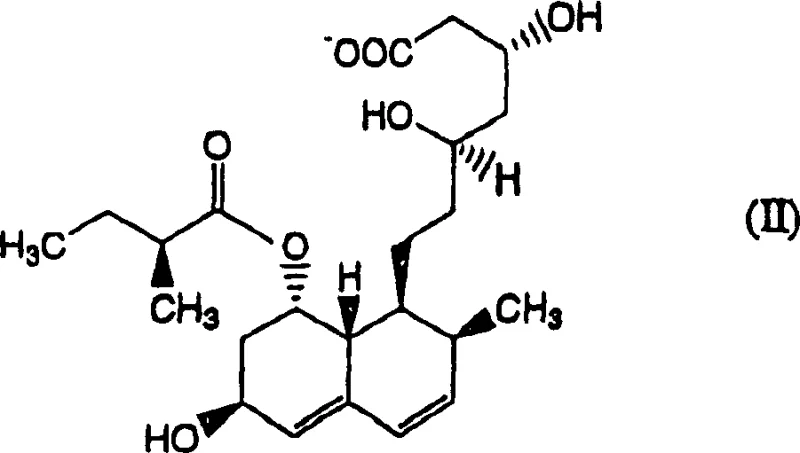
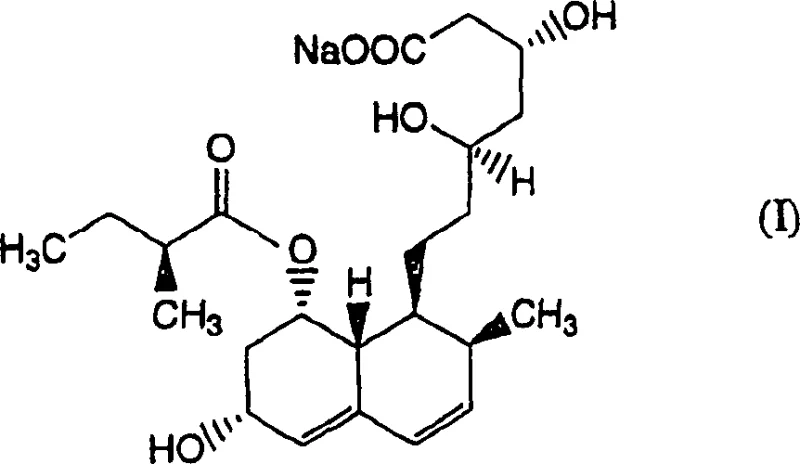
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients, particularly for statins like Pravastatin Sodium which are administered chronically for hyperlipidemia management. A pivotal advancement in this domain is detailed in patent CN1868996A, which outlines a robust method for isolating and purifying Pravastatin or its pharmacologically acceptable salts. This technology addresses the critical challenge of removing specific structural analogues, notably Compound (I), an optical isomer that is notoriously difficult to separate using conventional chromatography. By leveraging a combination of specialized organic solvent extraction and controlled chemical decomposition, this process achieves purity levels exceeding 99.5%, ensuring safety and efficacy for end patients while offering a streamlined pathway for manufacturers.
For procurement managers and supply chain directors, the implications of adopting this purification technology extend far beyond mere regulatory compliance. The shift away from high-performance liquid chromatography (HPLC) towards scalable solvent extraction and crystallization represents a significant opportunity for cost reduction in API manufacturing. Traditional methods often rely on expensive stationary phases and large volumes of solvents that are difficult to recover, creating bottlenecks in production throughput. In contrast, the methodology described in the patent utilizes common industrial solvents like n-butyl acetate and n-propyl acetate, which are more cost-effective and easier to manage in large-scale reactors. This transition not only lowers the direct cost of goods sold but also enhances the reliability of the supply chain by reducing dependency on complex, maintenance-intensive purification equipment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Pravastatin generated via two-stage fermentation has been plagued by the presence of persistent impurities that share nearly identical physicochemical properties with the target molecule. Conventional approaches often resort to ethyl acetate extraction followed by rigorous chromatographic separation to achieve acceptable purity levels. However, ethyl acetate lacks the selectivity required to effectively discriminate between Pravastatin and its close analogues, leading to co-extraction of impurities that necessitate downstream removal. Furthermore, reliance on chromatography introduces significant operational inefficiencies, including high solvent consumption, lengthy cycle times, and difficulties in scaling up from laboratory to commercial production. These factors collectively contribute to elevated manufacturing costs and extended lead times, posing challenges for meeting the growing global demand for cardiovascular medications.
The Novel Approach
The innovative strategy presented in the patent data fundamentally rethinks the purification workflow by optimizing the extraction solvent and introducing a chemical decomposition step. Instead of ethyl acetate, the process employs organic solvents represented by the formula CH3CO2R, where R is an alkyl group with three or more carbon atoms, such as n-propyl or n-butyl. This subtle change in solvent structure dramatically improves the partition coefficient, favoring the extraction of Pravastatin while leaving many impurities behind. Additionally, the method incorporates a targeted decomposition step using mineral acids or bases to chemically degrade specific stubborn impurities like Compound (I) into harmless byproducts. This dual-pronged approach eliminates the need for preparative HPLC, simplifying the process flow and making it inherently more suitable for continuous, large-scale industrial operations.
Mechanistic Insights into Solvent Selectivity and Impurity Decomposition
Understanding the chemical basis of this purification method is crucial for R&D directors evaluating its feasibility for technology transfer. The core of the innovation lies in the differential solubility and chemical stability of Pravastatin versus its impurities under specific conditions. Compound (I), the primary target for removal, is an optical isomer that differs slightly in stereochemistry but significantly in stability under acidic or basic conditions. By adjusting the pH of the reaction mixture to between 2 and 5 using mineral acids like phosphoric acid, or alternatively to between 10 and 14 using mineral alkalis like sodium hydroxide, the process induces selective hydrolysis or degradation of the impurity. This chemical transformation renders the impurity either water-soluble or easily separable, allowing the intact Pravastatin molecule to remain in the desired phase.
Furthermore, the choice of solvent plays a pivotal role in the initial bulk separation. Solvents with longer alkyl chains, such as n-butyl acetate, exhibit different polarity and hydrogen bonding capabilities compared to ethyl acetate. This difference alters the solvation shell around the Pravastatin molecule, enhancing its solubility in the organic phase while reducing the solubility of polar impurities. The synergy between this selective extraction and the subsequent chemical decomposition creates a powerful purification cascade. For technical teams, this means that purity specifications can be met with fewer processing steps, reducing the cumulative yield loss typically associated with multi-step purification trains. The result is a process that is not only chemically elegant but also operationally robust, capable of consistently delivering high-purity material batch after batch.
How to Synthesize Pravastatin Sodium Efficiently
Implementing this purification protocol requires precise control over reaction parameters to maximize yield and purity. The process begins with the fermentation broth, which is first clarified to remove biomass before undergoing pH adjustment. The critical extraction step follows, where the choice of solvent and the number of extraction stages are optimized to recover the maximum amount of product. Subsequent treatment with acid or base must be carefully monitored to ensure complete decomposition of the target impurity without affecting the stability of the Pravastatin lactone ring or side chain. Detailed standardized synthesis steps see the guide below.
- Adjust the fermentation broth pH and extract using organic solvents with alkyl groups of 3 or more carbons, such as n-butyl acetate.
- Treat the extract with mineral acid at pH 2-5 or mineral alkali at pH 10-14 to decompose specific impurities like Compound (I).
- Isolate the purified Pravastatin sodium through crystallization, achieving purity levels exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this purification technology offers substantial strategic advantages for organizations looking to optimize their supply chain for cardiovascular APIs. The elimination of chromatographic purification steps translates directly into reduced capital expenditure and lower operating costs, as there is no longer a need for expensive resin columns or the associated solvent recovery systems designed for high-dilution processes. Moreover, the use of common industrial solvents like n-butyl acetate simplifies procurement logistics, as these materials are widely available and less subject to supply volatility compared to specialized chromatographic grades. This stability ensures consistent production schedules and reduces the risk of delays caused by raw material shortages, thereby enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The process significantly lowers manufacturing costs by removing the need for high-performance liquid chromatography, which is both solvent-intensive and time-consuming. By utilizing standard extraction and crystallization unit operations, facilities can achieve higher throughput with existing infrastructure. The selective decomposition of impurities reduces the number of recycling loops required, further saving on energy and utility consumption. Additionally, the improved selectivity of the solvents minimizes product loss during purification, leading to better overall mass balance and yield efficiency.
- Enhanced Supply Chain Reliability: Relying on widely available solvents such as n-propyl acetate and n-butyl acetate mitigates the risk of supply chain disruptions associated with specialty chemicals. The simplified process flow reduces the complexity of manufacturing, making it easier to qualify multiple production sites or contract manufacturing organizations. This flexibility allows procurement teams to diversify their supply base, ensuring continuity of supply even in the face of regional disruptions. The robustness of the method also means faster turnaround times from production to quality release, enabling quicker response to market demand fluctuations.
- Scalability and Environmental Compliance: The technology is inherently designed for scale-up, utilizing standard reactor configurations that are common in the fine chemical industry. This facilitates a smoother transition from pilot plant to commercial production without the engineering challenges often posed by chromatographic scaling. From an environmental standpoint, reducing solvent usage and eliminating silica gel waste contributes to a greener manufacturing footprint. The ability to recycle solvents more efficiently in a liquid-liquid extraction system compared to chromatography further aligns with sustainability goals and regulatory expectations for waste reduction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. They are derived from the specific advantages and operational parameters detailed in the patent documentation, providing clarity for stakeholders evaluating the process for potential adoption or sourcing. Understanding these nuances is essential for making informed decisions about integrating this method into existing production workflows or selecting a qualified supplier.
Q: How does this process remove the difficult optical isomer impurity?
A: The process utilizes specific mineral acid or alkali treatments that selectively decompose the structural analogue impurity (Compound I) without degrading the target Pravastatin molecule.
Q: Why are n-propyl or n-butyl acetate preferred over ethyl acetate?
A: Solvents with alkyl chains of 3 or more carbons demonstrate superior selectivity in extraction, significantly reducing the co-extraction of impurities compared to traditional ethyl acetate methods.
Q: Is this purification method suitable for industrial scale-up?
A: Yes, by eliminating the need for complex chromatography and using standard solvent extraction and crystallization unit operations, the method is highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pravastatin Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and scalability in the production of life-saving medications like Pravastatin. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle complex purification challenges, utilizing advanced solvent extraction and crystallization techniques to meet stringent purity specifications. With rigorous QC labs and a commitment to quality, we deliver high-purity Pravastatin Sodium that adheres to the highest international standards, providing you with a secure and reliable source for your pharmaceutical formulations.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of advanced purification technologies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Contact us today to request specific COA data and route feasibility assessments, and discover how our expertise can support your long-term business goals in the competitive cardiovascular therapeutic market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
