Advanced Dasatinib Synthesis: Scalable High-Purity API Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical oncology treatments, and the synthesis of Dasatinib stands as a prime example of this technological evolution. Patent CN109265455B discloses a preparation method that fundamentally reimagines the construction of this tyrosine kinase inhibitor, utilizing 2-bromothiazole-5-formic acid and 2-methyl-4-amino-6-chloropyrimidine as key starting materials. This innovative approach bypasses the cumbersome protection and deprotection sequences that have historically plagued the production of this vital API intermediate. By streamlining the reaction sequence into a more direct pathway, the technology ensures that the stability of raw materials and intermediate products remains optimal throughout the process. The result is a manufacturing protocol that not only enhances reaction activity and selectivity but also significantly reduces the generation of process wastewater, aligning with modern green chemistry standards. For stakeholders evaluating supply chain resilience, this patent represents a pivotal shift towards more sustainable and efficient production methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
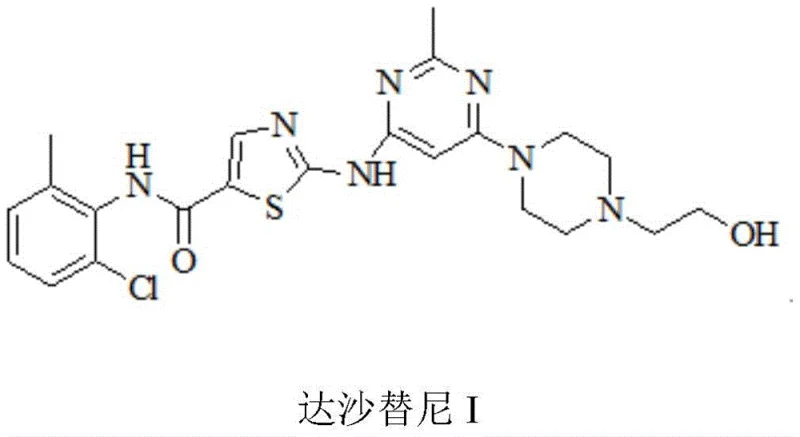
The Limitations of Conventional Methods
Historically, the synthesis of Dasatinib has been fraught with operational complexities that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional routes, such as those utilizing 2-chlorothiazole and 2-chloro-6-methylphenyl isocyanate, often require hazardous reagents like n-butyllithium and sodium hydride, which pose significant safety risks in large-scale environments. Furthermore, these legacy methods frequently involve multiple protection and deprotection steps, such as using p-methoxybenzyl chloride, which drastically increases the number of unit operations and overall processing time. The reliance on strong bases and unstable intermediates often leads to low reaction selectivity, resulting in the formation of disubstituted by-products that are difficult to remove. Consequently, extensive recrystallization and purification steps are necessary, which drive up costs and reduce the overall yield of the final product. The environmental footprint is also considerable, with large volumes of wastewater generated from the numerous washing and neutralization steps required to manage these aggressive chemical conditions.
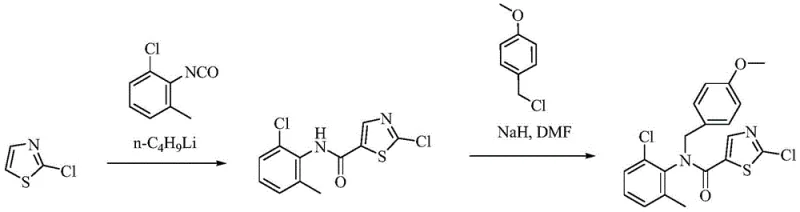
The Novel Approach
In stark contrast, the novel approach detailed in the patent data introduces a streamlined strategy that eliminates the need for dangerous reagents and complex protecting group manipulations. By leveraging the specific reactivity of 2-bromothiazole-5-formic acid, the process activates the 2-bromine atom through the electron-withdrawing effect of the 5-carboxylate, facilitating a smooth first substitution reaction with lower activity amines. This clever design allows for a continuous second substitution reaction with 4-(2-acetoxyl) ethylpiperazine, capitalizing on the stronger nucleophilic ability of the nitrogen atom to form the core structure efficiently. The subsequent steps involve classical acyl chlorination, amidation, and hydrolysis reactions that are well-understood and easy to control on an industrial scale. This reduction in process complexity translates directly to cost reduction in API manufacturing, as fewer steps mean lower labor, energy, and material consumption. Moreover, the high stability of the intermediates ensures that the reaction conditions are easy to realize, minimizing the risk of decomposition and ensuring consistent product quality.
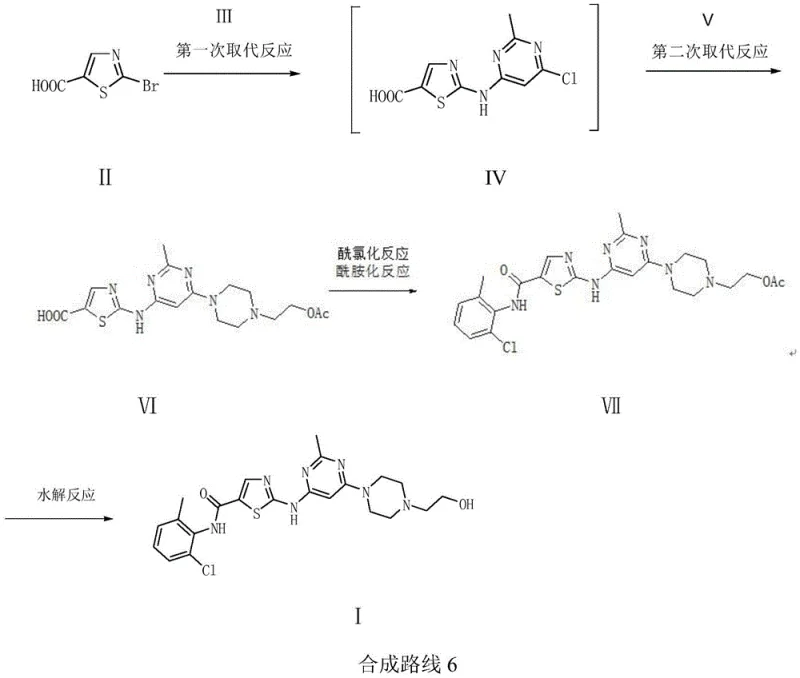
Mechanistic Insights into Substitution and Amidation Reactions
The core of this technological breakthrough lies in the precise manipulation of nucleophilic substitution mechanisms to construct the thiazole-pyrimidine scaffold with high fidelity. The initial reaction between 2-bromothiazole-5-carboxylic acid and 2-methyl-4-amino-6-chloropyrimidine is driven by the activation of the bromine leaving group, which is enhanced by the adjacent carboxylic acid moiety. This electronic effect lowers the energy barrier for the nucleophilic attack by the amino group of the pyrimidine, allowing the reaction to proceed smoothly at moderate temperatures between 80 to 110°C. The use of an acid-binding agent, such as potassium carbonate, further facilitates this process by neutralizing the generated acid, pushing the equilibrium towards the desired product. Following this, the second substitution with the piperazine derivative exploits the high nucleophilicity of the piperazine nitrogen, ensuring that the reaction proceeds with high specificity and minimal side reactions. This sequential substitution strategy avoids the formation of disubstituted by-products that are common in other routes, thereby simplifying the downstream purification process significantly.
Impurity control is inherently built into the reaction design through the use of an acetyl protecting group on the hydroxyethyl side chain of the piperazine ring. This strategic protection prevents the hydroxyl group from participating in unwanted side reactions during the subsequent acyl chlorination and amidation steps. The acyl chlorination of the carboxylic acid intermediate is performed using reagents like thionyl chloride, converting the acid into a highly reactive acyl chloride which then readily undergoes amidation with 2-chloro-6-methylaniline. The final hydrolysis step removes the acetyl group under mild basic conditions, revealing the free hydroxyl group essential for the biological activity of Dasatinib. This careful orchestration of functional group transformations ensures that the final product has less impurities and high purity, often reaching liquid phase purity levels of 99.9%. The robustness of this mechanism provides a reliable foundation for producing high-purity pharmaceutical intermediates that meet stringent regulatory requirements.
How to Synthesize Dasatinib Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and stoichiometry to maximize yield and purity. The process begins with the substitution reactions in a solvent like N,N-dimethylformamide, where temperature control is critical to ensure complete conversion without degrading the sensitive intermediates. Detailed standardized synthesis steps are essential for reproducibility, particularly during the acyl chlorination and amidation phases where moisture control is paramount. The final hydrolysis step must be monitored closely to ensure complete deprotection while avoiding over-hydrolysis of the amide bond. Adhering to the specific molar ratios and temperature ranges outlined in the patent data is crucial for achieving the reported high yields and purity profiles. The following guide outlines the critical operational parameters for successful execution.
- Perform first and second substitution reactions using 2-bromothiazole-5-carboxylic acid and 2-methyl-4-amino-6-chloropyrimidine to form the core thiazole-pyrimidine structure.
- Execute acylchlorination followed by amidation with 2-chloro-6-methylaniline to construct the amide bond essential for kinase inhibition activity.
- Conduct final hydrolysis reaction to remove the acetyl protecting group, yielding the final Dasatinib active pharmaceutical ingredient with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers substantial strategic benefits that extend beyond simple chemical efficiency. The elimination of expensive and hazardous reagents like n-butyllithium and sodium hydride drastically simplifies the safety protocols required for manufacturing, reducing the operational burden on facility management. Furthermore, the shortened process flow means that the production cycle time is significantly compressed, allowing for faster response to market demand fluctuations. The use of cheap and easily obtained raw materials ensures that the supply chain is less vulnerable to raw material price volatility, providing greater cost predictability for long-term contracts. Additionally, the green and environment-friendly nature of the process, with small production amounts of process wastewater, reduces the costs associated with waste treatment and environmental compliance. These factors combine to create a manufacturing profile that is not only economically attractive but also resilient and sustainable.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis route directly contributes to significant cost savings by reducing the number of unit operations and the consumption of expensive reagents. By avoiding complex protection and deprotection sequences, the process minimizes the loss of material at each step, leading to higher overall yields and better atom economy. The use of common solvents and bases further lowers the material costs, while the high selectivity reduces the need for costly purification steps like chromatography. This efficiency translates into a lower cost of goods sold, allowing for more competitive pricing in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially available starting materials ensures a robust supply chain that is less prone to disruptions. Unlike routes that depend on specialized or unstable intermediates, this method uses compounds that can be sourced from multiple suppliers, reducing the risk of single-source dependency. The simplicity of the operation also means that the technology can be transferred more easily between manufacturing sites, providing flexibility in production planning. This reliability is crucial for maintaining continuous supply to downstream customers, especially in the critical oncology therapeutic market where interruptions can have severe consequences.
- Scalability and Environmental Compliance: The process is designed with industrial production in mind, featuring reaction conditions that are easy to realize and control on a large scale. The absence of hazardous reagents and the generation of minimal wastewater make it easier to comply with increasingly strict environmental regulations. This environmental compliance reduces the risk of regulatory fines and shutdowns, ensuring long-term operational continuity. Furthermore, the high yield and purity reduce the waste generated per unit of product, aligning with corporate sustainability goals and enhancing the brand reputation of the manufacturer as a responsible partner.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial viability of this Dasatinib synthesis method. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation. Understanding these details is crucial for making informed decisions about sourcing and manufacturing strategies. The data provided reflects the performance capabilities of the route under optimized conditions, offering a realistic view of its potential impact on your supply chain.
Q: What are the primary advantages of the CN109265455B synthesis route?
A: The route described in patent CN109265455B offers significant advantages including cheap and easily obtained raw materials, a short process flow, and safe, simple operations. It minimizes wastewater production, making it green and environment-friendly, while achieving high reaction selectivity and yield with fewer impurities compared to conventional methods.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the method is highly beneficial for industrial production. The reaction conditions are easy to realize, and the stability of raw materials and intermediates is proper. The process avoids complex protection and deprotection steps found in other routes, facilitating easier commercial scale-up of complex pharmaceutical intermediates.
Q: What purity levels can be expected from this manufacturing process?
A: The prepared Dasatinib exhibits less impurities and high purity, with liquid phase purity reaching up to 99.9% in examples. The high selectivity of the substitution reactions and the specific functional group reactions contribute to this exceptional purity profile, meeting stringent quality specifications for API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dasatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who can translate complex patent technologies into reliable commercial supply. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Dasatinib intermediate meets the highest quality standards. Our expertise in handling complex chemical transformations allows us to optimize the novel route described in CN109265455B for maximum efficiency and cost-effectiveness. By partnering with us, you gain access to a supply chain that is both robust and responsive to your specific needs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project requirements. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this more efficient manufacturing method. Our team is ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us help you secure a reliable supply of high-quality Dasatinib intermediates that will drive the success of your pharmaceutical products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
