Revolutionizing Perampanel Production: A Deep Dive into Green Synthesis and Commercial Scalability
The pharmaceutical landscape for epilepsy treatment has been significantly shaped by the introduction of Perampanel, a non-competitive AMPA receptor antagonist. As the demand for this critical active pharmaceutical ingredient continues to grow globally, the efficiency and sustainability of its synthesis become paramount for supply chain stability. Patent CN110028442B discloses a groundbreaking preparation method that addresses the longstanding inefficiencies of conventional routes. This technical insight report analyzes the novel synthetic pathway, highlighting its potential to redefine cost structures and operational reliability for reliable pharmaceutical intermediates supplier networks. By shifting away from precious metal dependency, this technology offers a robust framework for cost reduction in pharmaceutical intermediates manufacturing. The following analysis details the chemical mechanisms and commercial implications of this innovation.
The Limitations of Conventional Methods vs. The Novel Approach
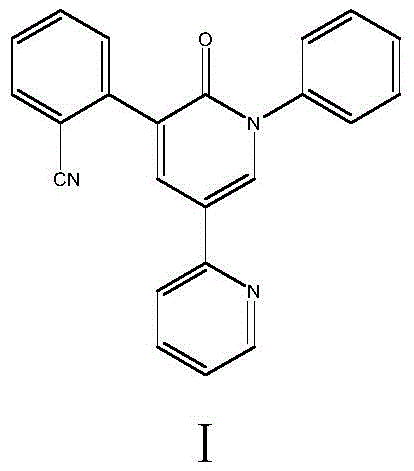
The Limitations of Conventional Methods
Historically, the synthesis of Perampanel has relied heavily on complex multi-step routes involving transition metal catalysis. As detailed in prior art such as CN103980188A and US2007014640, traditional pathways often utilize expensive starting materials like 2,5-dibromopyridine or 2-chloro-5-acetylpyridine. These routes necessitate the use of palladium acetate and copper acetate catalysts for coupling reactions, which introduces significant challenges. The reliance on precious metals not only inflates raw material costs but also creates a critical bottleneck in purification, as removing heavy metal residues to meet pharmaceutical standards requires additional, costly processing steps. Furthermore, the total yields of these conventional methods are often suboptimal, ranging typically between 26% and 42%, which severely impacts the overall atom economy. The generation of substantial waste water and the use of toxic reagents like organotin compounds in some variants further exacerbate environmental compliance issues, making these routes less viable for modern green manufacturing standards.
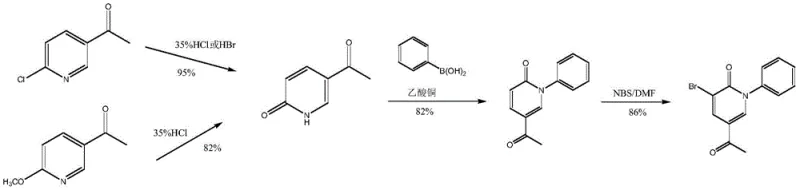
The Novel Approach
In stark contrast, the method disclosed in patent CN110028442B presents a streamlined alternative that circumvents the need for precious metal catalysts entirely. This novel approach utilizes readily available and inexpensive raw materials, such as pyridine-2-acetonitrile, formaldehyde, and aniline, to construct the core scaffold. The process is characterized by its operational simplicity, potentially allowing for a 'one-pot' synthesis strategy that minimizes the handling of intermediates. By eliminating the multiple coupling steps that require palladium or copper, the new route drastically reduces the risk of heavy metal contamination, thereby simplifying the downstream purification process. The reaction specificity is notably high, ensuring that side reactions are minimized, which directly translates to higher crude purity and reduced waste generation. This shift represents a fundamental improvement in process chemistry, aligning with the industry's drive towards more sustainable and economically efficient commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Amidation and Cyclization
The core of this innovative synthesis lies in a carefully orchestrated sequence of amidation, condensation, and cyclization reactions. The process begins with the formation of a key intermediate, compound IV, through the amidation of a formula II compound (such as 3-anilino-2-(pyridin-2-yl) propionitrile) with a formula III compound (such as 2-cyanophenylacetate). This step is conducted under controlled thermal conditions, typically between 70°C and 140°C, often in solvents like toluene or DMF, or even under solvent-free conditions to enhance green chemistry metrics. The subsequent transformation involves the condensation of compound IV with a methylene reagent, such as N,N-dimethylformamide dimethyl acetal, in the presence of an organic base catalyst like DBU or DMAP. This step constructs the necessary carbon framework for the final ring closure. The final cyclization is achieved by treating the condensed intermediate with an alkaline reagent, which facilitates the removal of hydrogen cyanide or hydrogen chloride to form the target pyridone ring structure of Perampanel. This mechanistic pathway is robust and avoids the sensitive organometallic steps found in older methods.
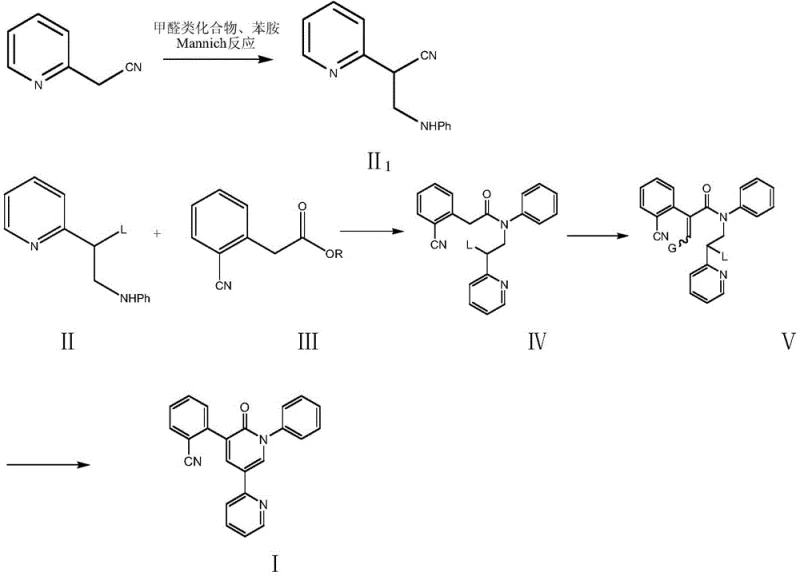
Impurity control is a critical aspect of this mechanism, particularly for R&D teams focused on regulatory compliance. The high selectivity of the unit reactions ensures that by-products are minimized at each stage. For instance, the amidation step is optimized to prevent hydrolysis of the nitrile or ester groups, which are common side reactions in similar chemistries. The use of specific bases and controlled temperatures during the cyclization step prevents the formation of polymeric by-products or isomeric impurities that could complicate crystallization. The patent data indicates that gas phase and liquid phase purities can consistently reach levels as high as 99.8% to 99.9%. This level of control is achieved without the need for extensive chromatographic purification, which is often required in metal-catalyzed routes to remove catalyst ligands and metal salts. The result is a high-purity pharmaceutical intermediates stream that is ready for final formulation with minimal additional processing, significantly reducing the technical burden on quality control laboratories.
How to Synthesize Perampanel Efficiently
Implementing this synthesis route requires precise adherence to the reaction parameters outlined in the patent to maximize yield and safety. The process is designed to be flexible, allowing for variations in solvent systems and reagents while maintaining high efficiency. For R&D teams looking to adopt this technology, the key lies in optimizing the molar ratios of the methylene reagent and the base catalyst during the condensation phase. The ability to run the reaction in a 'one-pot' manner offers significant operational advantages, reducing the time and labor associated with isolating intermediate compound IV. Detailed standard operating procedures regarding temperature ramps, addition rates, and work-up protocols are essential for successful technology transfer. The following guide outlines the standardized synthesis steps derived from the patent data to ensure reproducibility and safety in a pilot or production environment.
- Perform amidation reaction between compound of formula II and compound of formula III in a solvent or under solvent-free conditions to prepare compound of formula IV.
- Condense compound IV with a methylene reagent in the presence of a catalyst and solvent to obtain compound V.
- Remove hydrogen cyanide or hydrogen chloride under the action of an alkaline reagent to form rings and prepare Perampanel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers compelling strategic advantages beyond mere technical feasibility. The primary benefit is the substantial reduction in raw material costs driven by the elimination of precious metal catalysts and expensive halogenated starting materials. Traditional routes often suffer from price volatility associated with palladium and specialized boronic acids; this new method relies on commodity chemicals that are widely available in the global market. This shift ensures greater price stability and reducing lead time for high-purity pharmaceutical intermediates by mitigating the risk of supply disruptions for niche reagents. Furthermore, the simplified process flow reduces the consumption of solvents and energy, contributing to lower overall manufacturing overheads. These factors combine to create a more resilient supply chain capable of meeting fluctuating market demands without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The economic impact of removing palladium and copper catalysts from the synthesis cannot be overstated. Precious metals represent a significant portion of the COGS (Cost of Goods Sold) in traditional API manufacturing, not only due to their purchase price but also due to the costs associated with their recovery and the validation of their removal. By utilizing a metal-free organic synthesis pathway, manufacturers can achieve significant cost savings. Additionally, the high atom economy and reduced waste generation lower the costs associated with waste treatment and disposal. The ability to potentially operate under solvent-free conditions or with recoverable solvents further enhances the economic efficiency, making the final product more competitive in the global marketplace without sacrificing quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for complex intermediates. The raw materials for this new method, such as pyridine-2-acetonitrile and aniline, are produced by multiple chemical manufacturers worldwide, ensuring a robust and diversified supply base. This reduces the risk of production stoppages due to raw material shortages. Moreover, the simplified process reduces the number of unit operations, which decreases the likelihood of equipment failure or process deviations that can delay batch release. For supply chain planners, this means more predictable lead times and the ability to maintain lower safety stock levels while still ensuring uninterrupted supply to downstream formulation partners.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces new challenges regarding heat transfer and mixing, but this method is designed with scalability in mind. The reaction conditions are moderate, and the exothermic profiles are manageable, facilitating safe transfer from laboratory to pilot and commercial scales. From an environmental perspective, the reduction in heavy metal waste and organic solvent usage aligns with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden and the risk of fines or shutdowns due to environmental violations. The 'green' nature of the process also enhances the corporate sustainability profile, which is becoming a key criterion for procurement decisions in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is crucial for stakeholders evaluating its adoption. The following questions address common concerns regarding process safety, scalability, and quality assurance. These answers are derived directly from the experimental data and technical disclosures within the patent documentation. They serve to clarify the operational realities of the technology and provide confidence to decision-makers considering this route for their supply chain. By addressing these key points, we aim to facilitate a smoother evaluation process for technical and commercial teams alike.
Q: How does this new method compare to traditional palladium-catalyzed routes?
A: Unlike traditional routes that rely on expensive palladium and copper catalysts with complex purification needs, this novel method utilizes cheap and easily available raw materials. It eliminates the need for precious metal catalysis, significantly reducing heavy metal residue risks and overall production costs while maintaining high selectivity and yield.
Q: Is this synthesis process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It features simple and convenient operation, potentially allowing for a 'one-pot' procedure which minimizes intermediate isolation steps. The reaction conditions are easy to control, and the method generates less three wastes, making it environmentally compliant and economically efficient for industrial scale-up.
Q: What level of purity can be achieved with this preparation method?
A: The method demonstrates high reaction selectivity for related unit reactions, resulting in a final product with high yield and purity. Experimental data from the patent indicates gas phase and liquid phase purities reaching approximately 99.8% to 99.9%, ensuring the material meets stringent quality specifications for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perampanel Supplier
The technical potential of this novel synthesis route is immense, offering a pathway to more sustainable and cost-effective production of Perampanel. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. Our facility is equipped with rigorous QC labs and stringent purity specifications to ensure that every batch meets the highest international standards. We understand the complexities of transitioning a patent concept into a robust commercial process, and our team is dedicated to optimizing every step for maximum efficiency and yield. Partnering with us means gaining access to a supply chain that is both technically advanced and commercially reliable.
We invite you to explore how this innovative synthesis method can optimize your procurement strategy. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments. By collaborating early in the development cycle, we can ensure that the transition to this new supply source is seamless and beneficial for your long-term production goals. Let us help you secure a competitive advantage through superior chemistry and supply chain excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
