Advanced Synthetic Route For Novel Quinolinone Derivatives Enabling Commercial Scale Production
The global pharmaceutical landscape is continuously evolving to address the escalating prevalence of metabolic disorders, with diabetes mellitus representing one of the most critical public health challenges of the twenty-first century. In response to the urgent need for more effective therapeutic agents with improved safety profiles, recent intellectual property developments have highlighted promising new chemical entities. Specifically, the technical disclosure found in patent CN103880844A introduces a novel quinolinone compound characterized by a unique structural formula that demonstrates significant potential in regulating plasma glucose levels. This innovation is not merely a theoretical exercise but represents a tangible advancement in the field of antidiabetic medication, offering a mechanism that promotes glucose excretion through urine while mitigating the risks of hypoglycemia often associated with older drug classes. For stakeholders in the fine chemical and pharmaceutical sectors, understanding the synthetic accessibility and commercial viability of such compounds is paramount to securing a competitive edge in the supply chain. As a leading entity in the industry, we recognize that the transition from laboratory bench to commercial reactor requires a synthesis pathway that balances high purity with operational efficiency, ensuring that life-saving medications can reach patients without unnecessary delays or prohibitive costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex heterocyclic systems like quinolinones has been plagued by significant technical hurdles that hinder large-scale production and drive up manufacturing costs. Traditional routes often rely on harsh reaction conditions, including extreme temperatures or the use of highly corrosive reagents that pose safety risks and complicate waste management protocols in an industrial setting. Furthermore, conventional methodologies frequently suffer from poor regioselectivity, leading to the formation of multiple isomeric by-products that are chemically similar to the target molecule and notoriously difficult to separate during the purification phase. This lack of selectivity not only depresses the overall yield of the active pharmaceutical ingredient but also necessitates extensive downstream processing, such as repeated recrystallization or preparative chromatography, which consumes vast amounts of solvents and time. Additionally, many older synthetic strategies depend on scarce or expensive starting materials that are subject to volatile market pricing, creating instability in the supply chain and making long-term procurement planning difficult for pharmaceutical manufacturers who require consistent quality and availability.
The Novel Approach
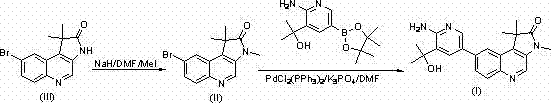
In stark contrast to these legacy challenges, the methodology outlined in the referenced patent presents a streamlined and robust synthetic strategy that effectively circumvents the bottlenecks of traditional chemistry. By leveraging modern transition metal catalysis, specifically the Suzuki-Miyaura cross-coupling reaction, this novel approach enables the construction of the carbon-carbon bond essential for the quinolinone scaffold under remarkably mild and controlled conditions. The use of a palladium catalyst system allows for high tolerance of functional groups, meaning that sensitive moieties within the molecule remain intact throughout the synthesis, thereby preserving the biological activity of the final product. This route significantly simplifies the purification process, as the reaction proceeds with high specificity to generate the desired isomer with minimal contamination from side products. Moreover, the selection of reagents and solvents is optimized for industrial applicability, utilizing materials that are readily available in bulk quantities and compatible with standard manufacturing equipment, thus facilitating a smoother transition from pilot plant studies to full-scale commercial production.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
The core of this synthetic breakthrough lies in the sophisticated application of palladium-catalyzed cross-coupling chemistry, which serves as the pivotal step in assembling the complex molecular architecture of the target quinolinone derivative. The reaction mechanism involves the oxidative addition of a palladium zero species into the carbon-bromine bond of the intermediate, followed by transmetallation with the organoboron reagent and subsequent reductive elimination to forge the new carbon-carbon linkage. This catalytic cycle is highly efficient, allowing for the use of catalytic amounts of the expensive metal rather than stoichiometric quantities, which aligns with green chemistry principles and reduces the heavy metal load in the final product. The choice of ligand and base is critical in this process, as they modulate the electronic and steric environment around the palladium center to accelerate the reaction rate and enhance selectivity. By carefully controlling parameters such as temperature and solvent polarity, chemists can fine-tune the reaction kinetics to maximize conversion while minimizing the degradation of sensitive functional groups, ensuring a high-quality output suitable for pharmaceutical applications.
Beyond the primary coupling event, the control of impurities is a critical aspect of this synthesis that directly impacts the safety and efficacy of the resulting medication. The patent details a meticulous purification protocol involving column chromatography with specific solvent ratios, designed to remove trace amounts of unreacted starting materials, catalyst residues, and any minor by-products formed during the reaction. This rigorous attention to detail in the downstream processing ensures that the final compound meets stringent purity specifications required by regulatory bodies for clinical use. The stability of the intermediate species is also managed through precise temperature control and inert atmosphere conditions, preventing oxidation or hydrolysis that could compromise the integrity of the molecule. Such comprehensive impurity profiling and control strategies are essential for establishing a reliable manufacturing process that can consistently deliver high-purity material, thereby reducing the risk of batch failures and ensuring patient safety throughout the product lifecycle.
How to Synthesize Novel Quinolinone Derivatives Efficiently
Implementing this synthesis in a practical setting requires a clear understanding of the operational parameters and safety considerations associated with each step of the process. The procedure begins with the preparation of the key intermediate through an N-methylation reaction, which sets the stage for the subsequent coupling event by introducing the necessary structural features. Following this, the Suzuki coupling is executed under nitrogen protection to maintain the activity of the catalyst and prevent side reactions with atmospheric oxygen. The detailed standardized synthesis steps provided below outline the specific reagents, molar ratios, and thermal conditions required to replicate the high yields reported in the patent literature, serving as a foundational guide for process chemists looking to adopt this technology.
- Perform N-methylation of the bromo-quinolinone precursor using sodium hydride and methyl iodide in dimethylformamide at elevated temperatures to generate the key intermediate.
- Execute a Suzuki-Miyaura cross-coupling reaction between the bromo-intermediate and a pyridine boronic ester using a palladium catalyst and potassium phosphate base.
- Purify the final crude product via column chromatography using a petroleum ether and ethyl acetate solvent system to achieve pharmaceutical grade purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthetic route offers substantial benefits that extend far beyond the laboratory, directly influencing the bottom line and operational resilience of the supply chain. The elimination of complex multi-step sequences in favor of a concise two-step process drastically reduces the overall manufacturing timeline, allowing for faster turnaround times from order placement to delivery. This efficiency is compounded by the use of commodity chemicals and solvents that are widely sourced, mitigating the risk of supply disruptions caused by the scarcity of exotic reagents. Furthermore, the high yields achieved in each step minimize material waste, leading to a more sustainable production model that aligns with increasingly strict environmental regulations and corporate sustainability goals. For procurement managers, this translates into a more predictable cost structure and the ability to negotiate better terms with suppliers due to the standardized nature of the required inputs.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy of the Suzuki coupling reaction, which ensures that a maximum proportion of the starting materials ends up in the final product rather than being lost as waste. By avoiding the need for expensive protecting group strategies or harsh reagents that require specialized containment, the capital expenditure for manufacturing facilities is significantly lowered. The reduction in solvent consumption and energy usage, owing to the milder reaction temperatures and simplified workup procedures, further contributes to a decrease in the overall cost of goods sold. These cumulative savings allow pharmaceutical companies to allocate resources more effectively, potentially lowering the price point for the final medication or reinvesting in further research and development initiatives.
- Enhanced Supply Chain Reliability: The reliance on robust and well-understood chemical transformations enhances the reliability of the supply chain by reducing the likelihood of unexpected technical failures during production. Since the reagents and catalysts used are standard items in the fine chemical industry, sourcing them is straightforward, and there is a broad base of qualified vendors to choose from, preventing single-source dependency. The scalability of the process means that production volumes can be ramped up quickly to meet surges in demand without the need for extensive process re-engineering or new equipment installation. This flexibility is crucial for maintaining continuity of supply in a dynamic market environment where demand for antidiabetic medications is consistently growing.
- Scalability and Environmental Compliance: Scaling this synthesis from the gram scale to the tonnage scale is facilitated by the inherent safety and controllability of the reaction conditions, which do not involve hazardous exotherms or unstable intermediates. The waste stream generated is manageable and consists primarily of standard organic solvents and inorganic salts that can be treated using conventional wastewater treatment methods, ensuring compliance with environmental discharge standards. The reduced use of heavy metals, thanks to the catalytic nature of the palladium system, simplifies the disposal of spent catalysts and lowers the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the brand reputation of the manufacturer as a responsible and sustainable partner in the global healthcare ecosystem.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial implications of this novel quinolinone synthesis, providing clarity for decision-makers evaluating this technology for their portfolios. These answers are derived directly from the technical disclosures and experimental data presented in the patent documentation, ensuring accuracy and relevance for professional assessment. Understanding these details is essential for gauging the feasibility of integrating this compound into existing product lines or development pipelines.
Q: What is the primary advantage of the Suzuki coupling method described in CN103880844A?
A: The primary advantage lies in the high regioselectivity and yield (up to 87%) achieved under relatively mild thermal conditions, which minimizes the formation of difficult-to-remove side products compared to traditional nucleophilic substitution methods.
Q: Can this synthesis route be scaled for industrial manufacturing?
A: Yes, the route utilizes commercially available reagents such as dimethylformamide and standard palladium catalysts, and the reaction temperatures (80-100°C) are well within the operational range of standard stainless steel reactors used in fine chemical production.
Q: How does this compound contribute to diabetes treatment mechanisms?
A: The novel quinolinone compound functions by promoting urinary glucose excretion and effectively reducing plasma glucose levels, offering a potential therapeutic pathway for managing hyperglycemia without the severe hypoglycemic risks associated with sulfonylureas.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolinone Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure necessary to bring complex synthetic routes like this novel quinolinone derivative from concept to commercial reality. Our team of experienced chemists is adept at optimizing reaction conditions to ensure maximum efficiency and purity, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that consistency is key in the pharmaceutical industry, which is why our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch meets the highest international standards. By partnering with us, you gain access to a supply chain that is not only robust and reliable but also capable of adapting to your specific volume and quality requirements with agility and precision.
We invite you to explore the potential of this advanced synthesis for your next project and discuss how we can support your strategic goals through our customized manufacturing services. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific needs, where we can provide specific COA data and route feasibility assessments to demonstrate the value we bring to your organization. Let us help you navigate the complexities of fine chemical sourcing and secure a competitive advantage in the rapidly evolving market for antidiabetic therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
