Advanced Metal-Free Synthesis of 3-Hydroxymethyl-4-Phenyl-3,4-Dihydroquinolinones for Scalable Pharmaceutical Manufacturing
Introduction to Patent CN112125843B: A Breakthrough in Heterocyclic Synthesis
The pharmaceutical industry constantly seeks robust, scalable, and environmentally benign pathways for constructing complex heterocyclic scaffolds, particularly those found in bioactive natural products and synthetic drugs. Patent CN112125843B introduces a significant advancement in this domain by disclosing a novel preparation method for 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone compounds. This technology leverages potassium monopersulfate (commonly known as Oxone) as a dual-function reagent to promote a tandem epoxidation-intramolecular Friedel-Crafts alkylation sequence. Unlike traditional methods that often rely on expensive transition metals or harsh acidic conditions, this approach utilizes readily available N-methyl-N-aryl-2-phenylacrylamides as starting materials in an acetonitrile medium. The process is characterized by its mild reaction conditions, operational simplicity, and exceptional functional group tolerance, making it highly attractive for the commercial scale-up of complex pharmaceutical intermediates. By eliminating the need for external catalysts and additives, this method addresses critical pain points regarding metal contamination and waste generation, offering a greener alternative for the synthesis of dihydroquinolinone derivatives which are pivotal in drug discovery.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dihydroquinolinone derivatives has often necessitated the use of transition metal catalysts such as palladium, copper, or rhodium, which introduce significant challenges in a GMP manufacturing environment. The presence of residual heavy metals in the final active pharmaceutical ingredient (API) requires rigorous and costly purification steps, including scavenging resins or repeated recrystallizations, to meet stringent regulatory limits. Furthermore, many conventional cyclization protocols demand strong Brønsted or Lewis acids to activate the electrophile, which can lead to poor chemoselectivity and degradation of sensitive functional groups on the substrate. These harsh conditions often limit the substrate scope, preventing the incorporation of diverse substituents that might be crucial for structure-activity relationship (SAR) studies. Additionally, multi-step sequences involving separate epoxidation and cyclization stages increase the overall process time, reduce atom economy, and generate substantial chemical waste, thereby inflating the cost of goods sold (COGS) and complicating supply chain logistics for large-scale production.
The Novel Approach
The methodology described in CN112125843B represents a paradigm shift by employing a metal-free, acid-additive-free strategy that streamlines the synthesis into a single pot. By utilizing potassium monopersulfate, the process achieves a tandem transformation where the oxidant simultaneously facilitates the initial epoxidation of the alkene moiety and, through its acidic byproduct (potassium bisulfate), promotes the subsequent intramolecular Friedel-Crafts cyclization. This dual functionality eliminates the need for separate reagents and simplifies the workup procedure significantly. The reaction proceeds efficiently in acetonitrile at moderate temperatures, demonstrating excellent compatibility with various substituents including alkyl, alkoxy, and halogen groups on the aromatic ring. This broad substrate scope allows medicinal chemists to rapidly access a diverse library of analogues without redesigning the synthetic route for each variant. The high yields achieved under these mild conditions, coupled with the absence of toxic metal residues, position this technology as a superior choice for cost reduction in pharmaceutical intermediate manufacturing and ensures a cleaner impurity profile for downstream processing.
Mechanistic Insights into Oxone-Promoted Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction initiates with the oxidation of the electron-rich alkene in the N-methyl-N-aryl-2-phenylacrylamide by the peroxymonosulfate anion, generating an epoxide intermediate and potassium bisulfate. This epoxidation step is highly selective and occurs under thermal conditions without the need for photochemical activation or metal catalysis. The generated potassium bisulfate then acts as an in situ proton source, activating the epoxide ring towards nucleophilic attack. This activation triggers an intramolecular Friedel-Crafts alkylation where the pendant aromatic ring attacks the activated epoxide carbon, forming the new C-C bond and closing the dihydroquinolinone ring system. The final step involves a dehydroaromatization or proton transfer to yield the stable 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone product. This cascade mechanism is elegant in its efficiency, as it avoids the isolation of unstable intermediates and minimizes side reactions typically associated with stepwise syntheses.
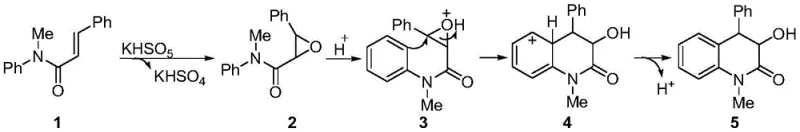
From an impurity control perspective, the mild nature of the Oxone-mediated system offers distinct advantages over aggressive acid-catalyzed routes. The absence of strong exogenous acids reduces the risk of polymerization or decomposition of the acrylamide starting material, which is a common issue in traditional Friedel-Crafts reactions. Furthermore, the specific requirement for acetonitrile as the solvent suggests a unique solvation environment that stabilizes the transition states involved in the cyclization, as evidenced by the complete lack of reactivity in other polar and non-polar solvents like DMF, DCM, or toluene. This solvent specificity acts as a built-in quality control parameter; deviation from the specified medium results in no reaction rather than the formation of complex, hard-to-remove byproducts. Consequently, the crude reaction mixture is cleaner, facilitating easier purification via standard silica gel chromatography and ensuring that the final product meets high-purity specifications required for pharmaceutical applications without extensive downstream processing.
How to Synthesize 3-Hydroxymethyl-4-Phenyl-3,4-Dihydroquinolinone Efficiently
To implement this synthesis effectively, precise adherence to the optimized reaction parameters is essential. The protocol involves dissolving the N-methyl-N-aryl-2-phenylacrylamide substrate in acetonitrile to achieve a concentration between 0.1 and 0.5 mol/L, followed by the addition of 1.5 equivalents of potassium monopersulfate. The mixture is then heated to 90°C and stirred for approximately 24 hours. Monitoring the reaction progress via TLC is recommended to ensure complete conversion while avoiding prolonged exposure to heat which might degrade the product. Upon completion, the solvent is removed under reduced pressure, and the residue is purified using column chromatography with a petroleum ether and ethyl acetate gradient. This standardized approach ensures reproducibility and high yields across different batches. For detailed standard operating procedures and safety guidelines, please refer to the technical documentation below.
- Prepare the reaction mixture by combining N-methyl-N-aryl-2-phenylacrylamide substrate with 1.5 equivalents of potassium monopersulfate in acetonitrile solvent.
- Heat the reaction mixture to 90°C and stir for approximately 24 hours to facilitate the tandem epoxidation and intramolecular Friedel-Crafts alkylation.
- Upon completion, concentrate the mixture and purify the crude product using silica gel column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Oxone-mediated synthesis offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for precious metal catalysts like palladium or rhodium, companies can avoid the volatility associated with the pricing and availability of these critical minerals. Moreover, the removal of transition metals from the process flow negates the requirement for expensive metal scavengers and the associated validation testing for residual metals, leading to significant cost reduction in pharmaceutical intermediate manufacturing. The use of potassium monopersulfate, a commodity chemical widely used in water treatment and cleaning, ensures a stable and reliable supply chain with minimal risk of disruption. This stability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for API clients.
- Cost Reduction in Manufacturing: The economic impact of this metal-free protocol is profound. Traditional methods often incur hidden costs related to the disposal of heavy metal waste and the energy-intensive processes required to purge trace metals from the final product. By switching to an organic oxidant system, manufacturers can streamline their waste management protocols and reduce the environmental compliance burden. The high atom economy of the tandem reaction means less raw material is wasted in side reactions, directly improving the overall yield and lowering the cost per kilogram of the final intermediate. Additionally, the simplified workup procedure reduces labor hours and solvent consumption during purification, contributing to substantial cost savings in the overall production budget without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents. Potassium monopersulfate and acetonitrile are bulk chemicals produced by numerous global suppliers, reducing dependency on single-source vendors for specialized catalysts. This diversification of the supply base mitigates the risk of shortages and price spikes that can plague the fine chemical industry. Furthermore, the robustness of the reaction conditions—specifically the tolerance to various functional groups—means that the same process can be applied to a wide range of substrate derivatives without needing to qualify new reagents or adjust parameters significantly. This flexibility allows for rapid scale-up from pilot plant to commercial production, ensuring that lead times for high-purity pharmaceutical intermediates are minimized and customer demand can be met consistently.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles. The absence of toxic heavy metals simplifies the handling of reaction effluents and reduces the hazard classification of the waste streams. This facilitates easier permitting for manufacturing facilities and lowers the costs associated with hazardous waste disposal. The reaction operates at atmospheric pressure and moderate temperatures, reducing the energy footprint compared to high-pressure hydrogenation or cryogenic processes. These factors collectively enhance the scalability of the process, allowing for safe operation in large-scale reactors. The ability to produce complex heterocycles with a lower environmental impact is increasingly becoming a key differentiator in vendor selection for major pharmaceutical companies committed to sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature. Understanding these nuances is vital for process chemists looking to adapt this method for specific project needs. The answers provide clarity on solvent selection, stoichiometry, and reaction conditions to ensure successful execution.
Q: Why is acetonitrile critical for this Oxone-mediated cyclization?
A: Experimental data indicates that acetonitrile is the sole effective solvent for this transformation. Other common solvents like DCM, toluene, DMF, or THF result in no reaction, likely due to specific solvation effects required for the epoxidation-cyclization cascade.
Q: Does this method require transition metal catalysts or added acids?
A: No. A key advantage of this patent technology is that it operates without transition metal catalysts or external acid additives. The potassium bisulfate byproduct from Oxone serves as the necessary proton source for the cyclization step.
Q: What is the optimal temperature range for maximizing yield?
A: The optimal reaction temperature is 90°C. Lower temperatures (e.g., 80°C) lead to incomplete conversion and lower yields (~56%), while higher temperatures (e.g., 100°C) promote side reactions, reducing yield to ~69%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxymethyl-4-Phenyl-3,4-Dihydroquinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free oxidation technologies in modern drug synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112125843B are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle the specific requirements of Oxone-mediated reactions, including corrosion-resistant reactor setups and advanced purification suites. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone meets the highest international standards. Our commitment to quality assurance ensures that our clients receive intermediates that are ready for immediate use in downstream API synthesis, minimizing delays and maximizing efficiency in their development timelines.
We invite pharmaceutical partners to leverage our technical expertise to optimize their supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this green synthesis route can enhance your project's viability. Contact us today to discuss how we can support your next breakthrough in heterocyclic chemistry with reliable, scalable, and cost-effective manufacturing solutions.
