Optimizing Loteprednol Etabonate Intermediate Production for Global Ophthalmic Markets
Optimizing Loteprednol Etabonate Intermediate Production for Global Ophthalmic Markets
The pharmaceutical landscape for ophthalmic anti-inflammatory agents is continuously evolving, driven by the demand for safer, more effective corticosteroids with reduced systemic side effects. A pivotal development in this sector is the synthesis of Loteprednol Etabonate, a 'soft drug' glucocorticoid designed for rapid metabolic deactivation. Central to the manufacturing of this high-value API is the efficient production of its key precursor, the 17-beta carboxylic acid intermediate. Recent intellectual property, specifically patent CN113105518A, discloses a groundbreaking preparation method that addresses longstanding inefficiencies in the supply chain. This technical insight report analyzes the novel synthetic pathway, highlighting its potential to redefine cost structures and purity standards for global manufacturers seeking a reliable loteprednol etabonate intermediate supplier.
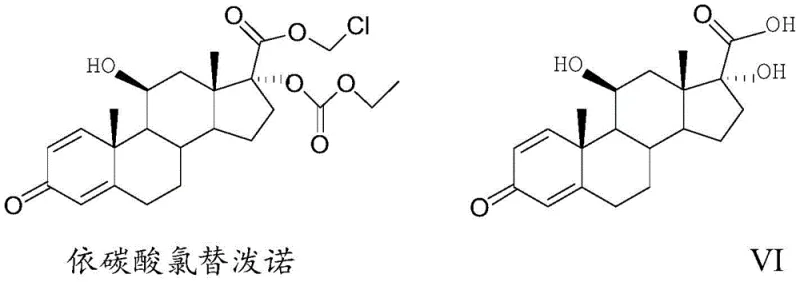
The strategic importance of this intermediate cannot be overstated, as it serves as the foundational scaffold for the final esterification into the active drug substance. Traditional manufacturing routes have often been plagued by high costs and complex purification requirements, creating bottlenecks for procurement teams. The methodology outlined in the patent leverages 11-alpha-hydroxy-ADD as a starting material, offering a distinct economic advantage over legacy routes that rely on more expensive substrates. By optimizing reaction conditions across five critical transformation steps, this process delivers a robust solution for cost reduction in ophthalmic steroid manufacturing, ensuring a stable supply of high-quality intermediates for downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the 17-beta carboxylic acid moiety found in loteprednol etabonate has relied heavily on prednisolone as the primary starting material. This conventional approach typically necessitates a periodate oxidation step to generate the carboxylic acid functionality at the C17 position. While chemically feasible, this route presents significant drawbacks for industrial application. Firstly, prednisolone is a high-cost raw material, which inherently inflates the Bill of Materials (BOM) for the final API. Secondly, the use of periodate oxidants introduces safety hazards and environmental concerns related to heavy metal waste and hazardous byproduct management. Furthermore, the older processes described in prior art, such as those referenced in CN111072743A, often suffer from lengthy synthetic sequences involving numerous auxiliary materials. These complex routes frequently result in lower overall yields and purity levels hovering around 97%, necessitating additional, costly purification steps to meet the stringent quality specifications required for ophthalmic formulations.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN113105518A represents a paradigm shift in process efficiency. By utilizing 11-alpha-hydroxy-ADD, a commercially available and cost-effective substrate, the new method bypasses the need for expensive prednisolone derivatives. The synthetic strategy employs a streamlined sequence of dehydration, halogenation, reduction, cyanation, and hydrolysis. This logical progression of functional group transformations allows for precise control over stereochemistry and regioselectivity without the need for exotic reagents. The result is a process that is not only shorter and simpler to operate but also delivers superior product quality. The elimination of hazardous oxidation steps and the use of standard organic solvents contribute to a greener manufacturing profile, aligning with modern sustainability goals while simultaneously enhancing the economic viability of producing high-purity 17-beta carboxylic acid.
Mechanistic Insights into the Five-Step Synthetic Cascade
The core of this technological advancement lies in the meticulous optimization of each reaction step to maximize yield and minimize impurity formation. The process initiates with a dehydration reaction where 11-alpha-hydroxy-ADD is treated with a dehydrating agent, preferably phosphorus pentachloride, in an organic solvent such as tetrahydrofuran at cryogenic temperatures ranging from -70°C to 10°C. This step is crucial for establishing the conjugated diene system necessary for subsequent transformations. Following dehydration, the intermediate undergoes a halogenation reaction using agents like dibromohydantoin in the presence of an acid catalyst. This electrophilic addition is carefully quenched to prevent over-halogenation, ensuring the formation of the specific halogenated intermediate required for the next stage. The precision in controlling reaction temperatures and stoichiometry during these early stages sets the foundation for the high purity observed in the final product.
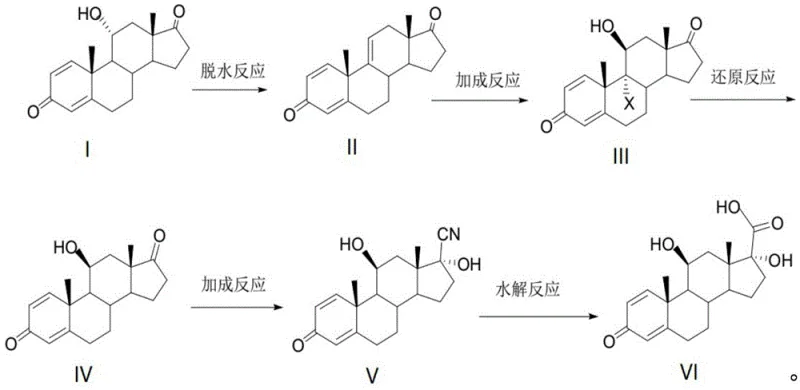
Subsequent steps involve a reductive dehalogenation using metal reducing agents like zinc powder in the presence of thioglycolic acid, which effectively restores the desired double bond configuration while removing the halogen atoms. This is followed by a nucleophilic addition reaction where a cyaniding reagent, such as acetone cyanohydrin, is introduced under basic catalysis. This step is pivotal as it installs the nitrile group that will eventually become the carboxylic acid. Finally, acidic hydrolysis converts the nitrile into the target 17-beta carboxylic acid. The mechanistic elegance of this route lies in its ability to build complexity gradually while maintaining structural integrity. By avoiding harsh oxidative conditions and utilizing mild, selective reagents, the process effectively suppresses the formation of difficult-to-remove byproducts, thereby achieving purity levels consistently above 98.5%.
How to Synthesize 17-beta Carboxylic Acid Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding temperature, solvent choice, and reagent ratios to ensure reproducibility and safety. The protocol is designed to be scalable, moving seamlessly from laboratory benchtop to pilot plant operations. Operators must pay particular attention to the exothermic nature of the dehydration and halogenation steps, utilizing appropriate cooling systems to maintain the specified low-temperature ranges. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a blueprint for process engineers aiming to replicate these results.
- Dehydration of 11-alpha-hydroxy-ADD with phosphorus pentachloride in THF at low temperature to form the delta-1,4-diene system.
- Halogenation of the resulting diene using dibromohydantoin and perchloric acid in acetone to introduce the halogen functionality.
- Reductive dehalogenation using zinc powder and thioglycolic acid in tetrahydrofuran to restore the desired stereochemistry.
- Nucleophilic addition of acetone cyanohydrin under basic conditions to install the nitrile group at the C17 position.
- Acidic hydrolysis of the nitrile intermediate in dichloromethane to yield the final 17-beta carboxylic acid with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere technical superiority. The shift to a lower-cost starting material directly impacts the bottom line, allowing for more competitive pricing models in a crowded generic pharmaceutical market. Moreover, the simplification of the process flow reduces the dependency on a wide array of specialized auxiliary chemicals, streamlining inventory management and reducing the risk of supply disruptions. The robustness of the reaction conditions means that production schedules are less likely to be impacted by batch failures or extended cycle times, enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the substitution of high-value prednisolone with 11-alpha-hydroxy-ADD. This raw material swap results in substantial cost savings at the very beginning of the value chain. Additionally, the elimination of periodate oxidation removes the need for expensive and hazardous oxidizing agents, further lowering operational expenditures. The high yields reported in the patent examples indicate that less raw material is wasted, maximizing the output per kilogram of input and driving down the unit cost of the intermediate significantly.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as zinc powder, acetone, and common mineral acids, are commodity chemicals with stable global supply chains. Unlike specialized catalysts or bespoke reagents that may face availability issues, these materials are readily accessible from multiple vendors. This diversification of the supply base mitigates the risk of single-source bottlenecks. Furthermore, the simplified workup procedures, which rely on standard extraction and crystallization techniques rather than complex chromatography, reduce the turnaround time between batches, ensuring a consistent and predictable flow of materials to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The avoidance of heavy metal oxidants and the use of recyclable solvents like dichloromethane and tetrahydrofuran simplify waste treatment protocols. The process generates fewer hazardous byproducts, reducing the burden on effluent treatment plants and lowering disposal costs. This environmental efficiency not only ensures compliance with increasingly stringent global regulations but also facilitates easier scale-up to commercial volumes, as the engineering controls required are standard for any modern fine chemical facility.
Frequently Asked Questions (FAQ)
Understanding the nuances of this new manufacturing technology is essential for stakeholders evaluating its integration into their supply networks. The following questions address common inquiries regarding the technical feasibility, quality attributes, and commercial implications of the process. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making.
Q: How does this new synthesis route compare to traditional prednisolone-based methods in terms of cost?
A: The traditional route relies on periodate oxidation of prednisolone, which involves expensive starting materials and hazardous oxidants. The patented method utilizes 11-alpha-hydroxy-ADD, a significantly lower-cost substrate, thereby drastically reducing raw material expenses and simplifying the supply chain for large-scale manufacturing.
Q: What purity levels can be achieved with this 17-beta carboxylic acid synthesis?
A: According to the patent data, the optimized process consistently achieves purity levels exceeding 98.5% as determined by HPLC. This high purity is critical for ophthalmic applications where impurity profiles must be strictly controlled to ensure patient safety and regulatory compliance.
Q: Is this process suitable for commercial scale-up?
A: Yes, the process is designed for scalability. It avoids complex chromatographic purifications in favor of crystallization and extraction steps. The use of common industrial solvents like THF, acetone, and dichloromethane, along with robust reagents like zinc powder, facilitates easy transition from laboratory to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Loteprednol Etabonate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a new synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical, large-volume outputs. Our facilities are equipped with state-of-the-art reactors capable of handling the cryogenic conditions and sensitive reagents required for this synthesis, guaranteeing consistent quality. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch of 17-beta carboxylic acid meets the exacting standards necessary for ophthalmic drug formulation.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains. By leveraging this advanced synthesis technology, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can secure a sustainable, high-quality supply of this critical intermediate, driving efficiency and innovation in the global ophthalmic market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
