Advanced Manufacturing of Loteprednol Etabonate Intermediate via Optimized Steroid Functionalization
Advanced Manufacturing of Loteprednol Etabonate Intermediate via Optimized Steroid Functionalization
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value ophthalmic corticosteroids, and patent CN111072743A presents a transformative approach to producing the critical 17β-carboxylic acid intermediate for Loteprednol Etabonate. This technology fundamentally shifts the raw material paradigm by utilizing 11α-hydroxy-ADD (Compound I) instead of the traditionally expensive prednisolone, thereby addressing significant cost bottlenecks in the supply chain. The disclosed method employs a sophisticated six-step sequence involving dehydration, Grignard addition, oxidative hydrolysis, chromic acid oxidation, regioselective bromination, and final reduction to achieve the target molecular architecture with high fidelity. By leveraging widely available steroid precursors and optimizing reaction conditions such as temperature control during Grignard additions and specific solvent systems for bromination, this process offers a scalable solution for commercial API manufacturing. For R&D directors and procurement strategists, this patent represents a viable alternative that balances chemical complexity with economic feasibility, ensuring a stable supply of high-purity intermediates essential for anti-inflammatory eye drop formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Loteprednol Etabonate intermediates has relied heavily on prednisolone as the foundational starting material, a strategy fraught with economic and logistical challenges for large-scale manufacturers. Prednisolone is a high-cost raw material due to its own complex synthesis and limited global availability, which directly inflates the cost of goods sold (COGS) for the final active pharmaceutical ingredient. Furthermore, existing literature methods often require harsh oxidation conditions at the C21 position to generate the necessary carboxylic acid functionality, leading to potential over-oxidation issues and difficult impurity profiles that complicate downstream purification. The reliance on such expensive starting materials creates a fragile supply chain vulnerable to market fluctuations, making it difficult for generic drug manufacturers to compete on price while maintaining stringent quality standards. Additionally, traditional routes may involve multiple protection and deprotection steps that lower overall atom economy and increase waste generation, conflicting with modern green chemistry initiatives and environmental compliance regulations.
The Novel Approach
The innovative methodology described in patent CN111072743A circumvents these historical limitations by initiating the synthesis with 11α-hydroxy-ADD, a significantly more economical and accessible steroid precursor. This novel route strategically constructs the C17 side chain early in the synthesis through a Grignard reaction with dimethylisopropoxysilanemethylmagnesium chloride, followed by a mild oxidative hydrolysis that efficiently converts the silyl group into a primary alcohol without affecting other sensitive functionalities on the steroid nucleus. The subsequent oxidation to the carboxylic acid is achieved under controlled conditions using chromic acid, ensuring high conversion rates while minimizing degradation of the steroid backbone. Crucially, the process introduces a unique bromination-reduction sequence to install the critical 11β-hydroxyl group and remove the C9 bromine, effectively restoring the biological activity required for the final drug substance. This stepwise logic not only improves overall yield but also simplifies the purification process, making it an ideal candidate for reliable loteprednol etabonate intermediate supplier operations aiming for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Grignard Addition and Oxidative Functionalization
The core chemical innovation of this process lies in the precise execution of the Grignard reaction and the subsequent oxidative transformation, which together build the critical C17 carboxylic acid side chain. In the second step, the delta-9(11) olefin (Compound II) reacts with dimethylisopropoxysilanemethylmagnesium chloride at low temperatures ranging from -10°C to 20°C, ensuring stereochemical control and preventing side reactions such as 1,4-addition to the enone system. The use of a silyl-protected Grignard reagent is particularly ingenious, as it acts as a masked hydroxymethyl group that can be revealed later under mild conditions. Following the Grignard addition, the hydrolysis step utilizes hydrogen peroxide in the presence of alkaline reagents like potassium bicarbonate and complexing agents such as potassium fluoride. This specific combination facilitates the migration of the carbon-silicon bond to a carbon-oxygen bond, a transformation mechanistically similar to the Tamao-Fleming oxidation, yielding the primary alcohol (Compound IV) with excellent regioselectivity. This mechanism avoids the use of hazardous fluorinating agents typically required in classical Tamao-Fleming protocols, thereby enhancing operational safety and scalability for commercial production environments.
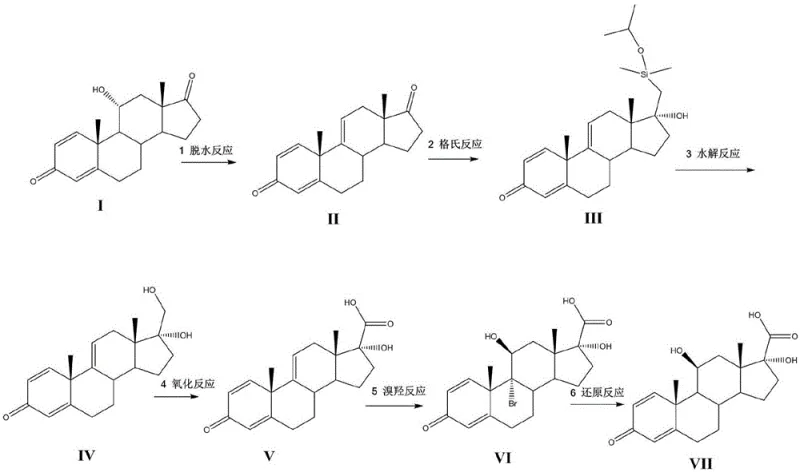
Following the construction of the side chain, the process employs a Jones-type oxidation using chromic acid to convert the primary alcohol into the corresponding carboxylic acid (Compound V), a transformation that must be carefully managed to prevent epimerization or degradation of the steroid nucleus. The subsequent bromination step involves reacting Compound V with N-bromosuccinimide (NBS) in the presence of an acidic catalyst, which introduces a bromine atom at the C9 position and simultaneously hydrates the C9(11) double bond to form the 11β-hydroxyl group found in Compound VI. This electrophilic addition is highly regioselective due to the directing effects of the C3 ketone and the C17 side chain. Finally, the removal of the C9 bromine is achieved through a reduction reaction using trivalent chromium salts and zinc powder in ethanol. This reductive debromination restores the delta-1 double bond characteristic of corticosteroids while retaining the newly installed 11β-hydroxyl and C17-carboxylic acid groups, resulting in the high-purity 17β-carboxylic acid (Compound VII). Understanding these mechanistic nuances is vital for quality control teams to monitor critical process parameters and ensure the consistent production of high-purity pharmaceutical intermediates.
How to Synthesize 17β-Carboxylic Acid Efficiently
The synthesis of this critical ophthalmic drug intermediate requires strict adherence to the optimized reaction conditions outlined in the patent to ensure maximum yield and purity. The process begins with the dehydration of the starting material using phosphoric acid in dichloromethane, followed by the critical low-temperature Grignard addition which sets the stereochemistry for the side chain. Operators must maintain precise temperature control during the hydrolysis and oxidation steps to prevent exothermic runaways and ensure complete conversion of the silyl group and alcohol respectively. The detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and workup procedures for each of the six transformations, are provided in the technical guide below to facilitate immediate technology transfer and pilot plant trials.
- Dehydration of 11α-hydroxy-ADD using phosphoric acid to form the delta-9(11) olefin.
- Grignard reaction with dimethylisopropoxysilanemethylmagnesium chloride followed by oxidative hydrolysis to install the C17 hydroxymethyl group.
- Oxidation of the primary alcohol to carboxylic acid, followed by regioselective bromination and subsequent reductive debromination to restore the 11β-hydroxy functionality.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic advantages primarily driven by the substitution of the starting material. By shifting from prednisolone to 11α-hydroxy-ADD, manufacturers can access a raw material that is not only significantly cheaper but also produced in larger volumes globally, thereby mitigating the risk of supply shortages that often plague the steroid market. This change in raw material sourcing directly translates to substantial cost savings in the overall manufacturing budget, allowing for more competitive pricing strategies in the generic ophthalmic sector without compromising on product quality. Furthermore, the elimination of complex protection-deprotection sequences found in older methods streamlines the production timeline, reducing the consumption of solvents and reagents which contributes to both cost reduction in pharmaceutical intermediates manufacturing and a smaller environmental footprint. The robustness of the reaction conditions, which utilize common industrial solvents like THF, acetone, and ethanol, ensures that the process can be easily scaled up in existing multipurpose reactors without requiring specialized equipment investments.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the utilization of 11α-hydroxy-ADD, a commodity steroid intermediate that is vastly more affordable than the prednisolone required by conventional synthesis routes. By removing the dependency on high-cost starting materials, the overall variable cost of production is drastically lowered, improving profit margins for the final API. Additionally, the high yields reported in the patent examples for each step, particularly the dehydration and oxidation stages, minimize material loss and reduce the burden on waste treatment facilities. The avoidance of precious metal catalysts in favor of standard reagents like chromium salts and zinc powder further decreases the cost of goods, as there is no need for expensive catalyst recovery systems or rigorous heavy metal scavenging steps prior to final crystallization.
- Enhanced Supply Chain Reliability: Sourcing 11α-hydroxy-ADD is inherently more stable than sourcing prednisolone, as the former is a common intermediate in the synthesis of various other corticosteroids, ensuring a diversified and resilient supply base. This diversification protects the manufacturing schedule from disruptions caused by single-source supplier issues or geopolitical trade barriers affecting specific high-value steroids. The simplified synthetic route also reduces the lead time for high-purity pharmaceutical intermediates by shortening the total number of processing days required to reach the final intermediate state. With fewer unit operations and simpler workup procedures, the throughput of the manufacturing facility can be increased, allowing for larger batch sizes and more frequent production campaigns to meet fluctuating market demand.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that are standard in the fine chemical industry and easily handled in large-scale stainless steel reactors. The reaction conditions, such as the moderate temperatures for hydrolysis and reduction, are energy-efficient and do not require extreme cryogenic cooling or high-pressure equipment, lowering utility costs. From an environmental perspective, the improved atom economy and higher yields result in less chemical waste per kilogram of product, facilitating easier compliance with increasingly strict environmental regulations regarding effluent discharge. The ability to produce the intermediate with high purity directly from the reactor reduces the need for extensive chromatographic purification, which is often a bottleneck in scaling steroid synthesis and a major source of solvent waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic pathway for Loteprednol Etabonate intermediates. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this technology into your existing manufacturing portfolio.
Q: What is the primary cost advantage of this synthetic route?
A: The process utilizes 11α-hydroxy-ADD as the starting material, which is significantly cheaper and more readily available than the conventional prednisolone starting material used in legacy methods.
Q: How is the C17 side chain constructed in this method?
A: The C17 side chain is built via a Grignard reaction using a silyl-protected methyl magnesium chloride reagent, followed by a Tamao-Fleming type oxidative hydrolysis to reveal the primary alcohol, which is then oxidized to the carboxylic acid.
Q: Does this method avoid the use of heavy metal catalysts in the final steps?
A: The final reduction step utilizes chromium salts and zinc powder, which are standard reducing agents, but the overall route avoids precious metal catalysts often found in cross-coupling reactions, simplifying purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Loteprednol Etabonate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of ophthalmic pharmaceuticals, and we are fully equipped to leverage this patented technology for your commercial needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant with cGMP standards. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 17β-carboxylic acid intermediate meets the exacting requirements for downstream API synthesis, providing you with a consistent and high-quality supply chain partner. Our commitment to technical excellence allows us to optimize the dehydration and Grignard steps further, ensuring maximum yield and minimal impurity formation for your specific application.
We invite you to contact our technical procurement team to discuss how this cost-effective synthesis can enhance your product portfolio and reduce your overall manufacturing expenses. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits of switching to this 11α-hydroxy-ADD based route for your operations. We are ready to provide specific COA data and route feasibility assessments tailored to your volume requirements, helping you secure a reliable supply of high-purity pharmaceutical intermediates for the global market.
