Optimized Industrial Synthesis of Parecoxib Sodium: Enhancing Purity and Scalability
Introduction to Advanced Parecoxib Sodium Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational safety, particularly for potent analgesics like parecoxib sodium. Patent CN111100084B introduces a transformative preparation method that fundamentally reengineers the synthesis of this COX-2 inhibitor prodrug. Unlike legacy processes that depend on hazardous organolithium reagents or corrosive superacids, this innovation leverages a mild, organic acid-assisted sulfonation strategy. By substituting aggressive dehydrating agents with biocompatible acids such as tartaric or citric acid, the process drastically mitigates equipment corrosion and hazardous waste generation. This technical breakthrough not only enhances the impurity profile of the final active pharmaceutical ingredient but also streamlines the supply chain by utilizing readily available, cost-effective reagents. For global procurement teams, this represents a shift towards a more sustainable and economically viable manufacturing model for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of parecoxib sodium has been plagued by significant safety and economic bottlenecks. Prominent prior art, such as the routes disclosed in WO2003029230, relies heavily on the use of n-butyllithium for the cyclization step to form the isoxazole core. As illustrated in the reaction scheme below, this reagent is not only prohibitively expensive but also poses severe safety risks due to its pyrophoric nature, requiring stringent inert atmosphere controls and cryogenic temperatures. Furthermore, alternative routes utilizing trifluoroacetic acid (TFA) for dehydration introduce substantial environmental liabilities. The generation of fluoride-containing wastewater necessitates complex and costly effluent treatment protocols, while the strong acidity of TFA accelerates the degradation of standard stainless-steel reactors, leading to frequent maintenance downtime and potential metal contamination of the product.
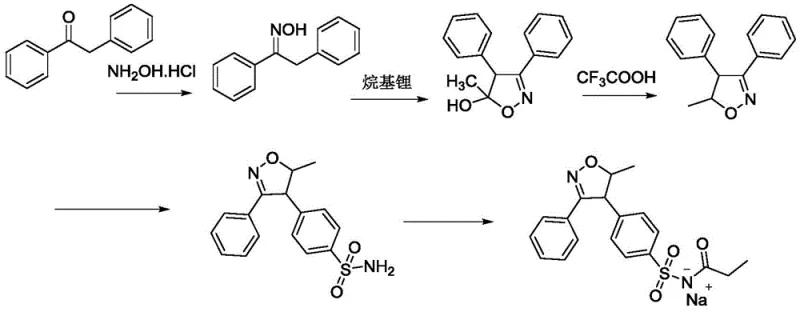
The Novel Approach
The methodology outlined in CN111100084B offers a decisive break from these inefficiencies by introducing a catalytic system based on sodium iodide and organic acids. Instead of relying on the harsh conditions of the past, this novel approach facilitates the sulfonation and subsequent ammoniation under mild thermal conditions, typically between 10-20°C. The integration of a dichloromethane-acetonitrile mixed solvent system during the propionylation step further optimizes reaction kinetics, allowing the process to proceed efficiently at room temperature. This strategic modification eliminates the need for expensive acid-binding agents like 2,6-lutidine and removes the requirement for protective group chemistry involving 2,5-hexanedione. The result is a streamlined three-step sequence that delivers superior yields and purity while significantly lowering the barrier to entry for safe, large-scale production.
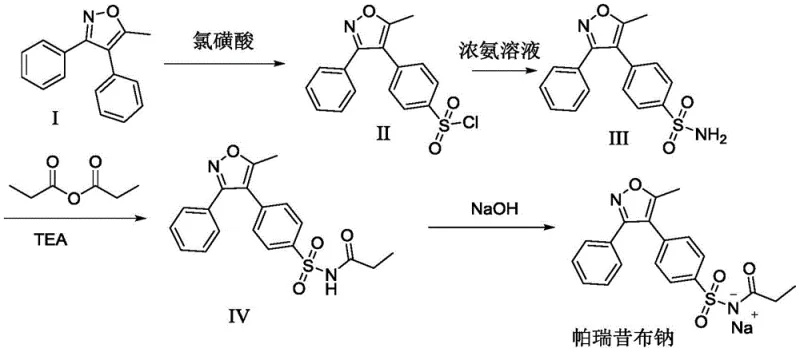
Mechanistic Insights into Organic Acid-Assisted Sulfonation
The core innovation of this patent lies in the modulation of the reaction environment during the critical sulfonation phase. By incorporating tartaric acid, citric acid, or formic acid alongside chlorosulfonic acid, the process effectively adjusts the proton concentration within the reaction matrix. This buffering effect prevents the localized overheating and runaway reactions often associated with direct chlorosulfonation, thereby preserving the integrity of the sensitive isoxazole ring. The addition of sodium iodide acts as a crucial kinetic accelerator, likely functioning through a Finkelstein-type mechanism or by enhancing the nucleophilicity of the intermediate species, which drives the equilibrium forward more rapidly. This synergistic interaction allows the reaction to reach completion in significantly shorter timeframes compared to traditional methods, minimizing the exposure of the intermediate to potentially degradative acidic conditions and reducing the formation of chlorinated byproducts.
Furthermore, the purification strategy employed in this method is meticulously designed to control the impurity profile. The use of specific solvent ratios, such as the 10:1 volume ratio of absolute ethanol to methanol during recrystallization, exploits subtle solubility differences to exclude structurally similar impurities. The subsequent propionylation step utilizes 4-pyrrolidinylpyridine as a highly efficient nucleophilic catalyst, which ensures complete acylation of the sulfonamide nitrogen without requiring excessive temperatures that could lead to racemization or decomposition. This precise control over reaction parameters ensures that the final parecoxib sodium product meets rigorous pharmacopeial standards, with single impurities consistently maintained below 0.1%, a critical metric for regulatory approval and patient safety.
How to Synthesize Parecoxib Sodium Efficiently
The synthesis of parecoxib sodium via this patented route involves a logical progression of sulfonation, acylation, and salification steps that are amenable to standard chemical processing equipment. The process begins with the careful addition of chlorosulfonic acid to a solution of the isoxazole precursor in the presence of an organic acid promoter, followed by quenching and ammoniation to yield the key sulfonamide intermediate. This intermediate is then subjected to propionylation using propionic anhydride in a mixed solvent system, a step that has been optimized to maximize conversion while minimizing side reactions. Finally, the free acid is converted to the sodium salt through a straightforward base treatment in methanol. For detailed operational parameters, stoichiometry, and safety protocols required to execute this synthesis in a GMP environment, please refer to the standardized guide below.
- Sulfonation and Ammoniation: React 5-methyl-3,4-diphenylisoxazole with chlorosulfonic acid in the presence of tartaric, citric, or formic acid and sodium iodide, followed by ammoniation to form the sulfonamide intermediate.
- Propionylation: Acylate the sulfonamide intermediate using propionic anhydride and triethylamine in a dichloromethane-acetonitrile mixed solvent with 4-pyrrolidinylpyridine as a catalyst.
- Salification: Treat the propionylated product with sodium hydroxide in methanol to form the final parecoxib sodium salt through crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound advantages for supply chain stability and cost management. By eliminating the dependency on n-butyllithium and specialized fluorinated reagents, manufacturers can decouple their production schedules from the volatile pricing and supply constraints associated with these niche chemicals. The shift to commodity-grade organic acids and common solvents like dichloromethane and acetonitrile ensures a more resilient supply chain, reducing the risk of production stoppages due to raw material shortages. Additionally, the milder reaction conditions translate directly into lower energy consumption, as the process does not require the extensive cooling or heating cycles demanded by legacy methods, contributing to a reduced carbon footprint and lower utility costs per kilogram of product.
- Cost Reduction in Manufacturing: The elimination of expensive reagents such as n-butyllithium and 2,6-lutidine results in substantial direct material cost savings. Furthermore, the removal of protective group steps reduces the total number of unit operations, which lowers labor costs and increases overall plant throughput. The avoidance of trifluoroacetic acid also removes the significant expense associated with treating fluoride-contaminated wastewater, providing a dual benefit of reduced raw material spend and lower waste disposal fees.
- Enhanced Supply Chain Reliability: Utilizing widely available organic acids and standard inorganic salts mitigates the risk of supply disruptions common with hazardous specialty reagents. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up, ensuring consistent delivery timelines for downstream formulation partners. This reliability is critical for maintaining continuous inventory levels of essential pain management medications in the global market.
- Scalability and Environmental Compliance: The process is inherently safer and more environmentally friendly, aligning with increasingly strict global regulations on hazardous waste and emissions. The reduction in HCl gas generation and the absence of fluoride waste simplify the permitting process for new manufacturing lines. This compliance advantage facilitates faster technology transfer and capacity expansion, enabling suppliers to respond rapidly to surges in market demand without facing regulatory bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the operational benefits and chemical rationale behind the process improvements. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is the new sulfonation method superior to traditional n-butyllithium routes?
A: Traditional routes rely on n-butyllithium, which is highly pyrophoric, expensive, and requires cryogenic conditions. The new method utilizes stable organic acids (tartaric/citric) and sodium iodide, significantly improving operational safety and reducing raw material costs while maintaining high yields.
Q: How does this process address environmental concerns regarding fluoride waste?
A: Conventional dehydration steps often use trifluoroacetic acid (TFA), generating hazardous fluoride-containing wastewater that requires complex treatment. This patented method replaces TFA with mild organic acids, eliminating fluoride pollution and simplifying effluent management.
Q: What purity levels can be achieved with this optimized crystallization protocol?
A: By utilizing specific solvent systems like absolute ethanol-methanol mixtures and controlling crystallization temperatures between 0-5°C, the process consistently achieves HPLC purity exceeding 99.8%, with single impurities controlled below 0.1%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Parecoxib Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to superior synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a manufacturing setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of parecoxib sodium meets the highest international standards. Our commitment to quality assurance ensures that the impurity profiles remain tightly controlled, safeguarding the efficacy and safety of the final therapeutic product.
We invite pharmaceutical companies and contract manufacturers to engage with our technical procurement team to explore how this optimized synthesis can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this greener, more efficient method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production requirements, ensuring a seamless integration of this advanced technology into your portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
