Advanced Imidazole Preparation: Eliminating Heavy Metals for Scalable API Manufacturing
The pharmaceutical and agrochemical industries rely heavily on the imidazole scaffold due to its prevalence in bioactive molecules, ranging from antifungal agents to anti-HIV therapeutics. However, the historical reliance on hazardous reagents has long plagued the supply chain for these critical intermediates. Patent CN1091130A introduces a transformative preparation method that fundamentally alters the synthetic landscape for formula (I) imidazoles. By utilizing a direct condensation between alpha,alpha-dihalo aldehyde compounds, standard aldehydes, and ammoniacal liquor, this technology bypasses the need for toxic heavy metal catalysts and noxious gases. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this innovation represents a pivotal shift towards safer, more sustainable, and cost-efficient manufacturing protocols that do not compromise on chemical complexity or yield.
The Limitations of Conventional Methods vs. The Novel Approach
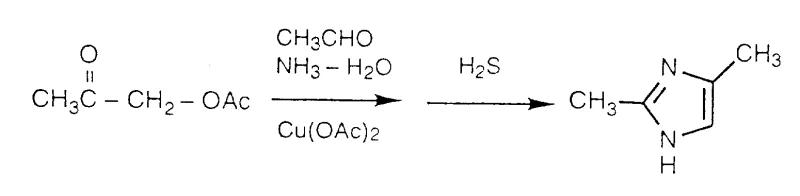
The Limitations of Conventional Methods
Historically, the synthesis of imidazoles has been dominated by methodologies described in literature such as those by Weidenhagen and Longenback, which necessitate the use of copper acetate (Cu(OAc)2) and hydrogen sulfide (H2S). As illustrated in the reaction schemes of the prior art, these processes introduce significant operational risks and environmental burdens. The handling of hydrogen sulfide gas poses acute toxicity hazards to laboratory personnel and requires specialized containment infrastructure, drastically increasing capital expenditure for production facilities. Furthermore, the use of copper salts introduces a persistent contamination risk; removing trace heavy metals to meet the stringent purity specifications required for Active Pharmaceutical Ingredients (APIs) often demands additional purification steps, such as chelation or extensive chromatography, which erode overall process efficiency and drive up the cost of goods sold.
The Novel Approach
In stark contrast, the method disclosed in CN1091130A offers a streamlined alternative that operates under remarkably mild conditions. By reacting an alpha,alpha-dihalo aldehyde with a chosen aldehyde and ammoniacal liquor, the process achieves cyclization without the need for transition metal catalysts or sulfur sources. This approach not only eliminates the safety risks associated with H2S but also simplifies the workup procedure significantly. The reaction can be conducted in common inert organic solvents like acetonitrile, methanol, or dichloromethane at room temperature or with slight heating. This simplicity translates directly into operational excellence, allowing for the production of high-purity imidazoles with reduced waste generation and lower energy consumption, addressing the core concerns of modern green chemistry initiatives in fine chemical manufacturing.
Mechanistic Insights into Alpha-Dihalo Aldehyde Condensation
The core of this technological advancement lies in the unique reactivity of the alpha,alpha-dihalo aldehyde precursor. In this mechanism, the dihalo species acts as a versatile electrophile that undergoes nucleophilic attack by ammonia, facilitating the formation of the imidazole ring through a cascade of condensation and cyclization events. The versatility of this pathway is evidenced by the broad scope of substituents tolerated at the R1 and R2 positions. R1 can range from simple hydrogen or alkyl groups to complex functionalities such as haloalkyls, acyloxyalkyls, cyanoalkyls, and various aromatic or heteroaromatic systems. Similarly, R2 offers extensive diversification, including cycloalkyls, thioethers, and trifluoromethyl-containing groups. This mechanistic flexibility allows chemists to access a vast chemical space of 2,4-disubstituted imidazoles, making it an invaluable tool for medicinal chemistry campaigns aiming to optimize structure-activity relationships (SAR) without being constrained by synthetic feasibility.
From an impurity control perspective, the absence of metal catalysts is a decisive advantage. Traditional copper-mediated reactions often leave behind organometallic byproducts that are difficult to separate and can catalyze degradation pathways in the final drug product. By employing a metal-free condensation strategy, the impurity profile of the resulting imidazole is significantly cleaner. The primary byproducts are typically inorganic salts and unreacted starting materials, which are far easier to remove via standard aqueous workups or recrystallization techniques. This inherent purity reduces the burden on Quality Control (QC) laboratories and ensures that the material meets the rigorous standards required for clinical trial supplies and commercial API production, thereby de-risking the regulatory filing process.
How to Synthesize Substituted Imidazoles Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation. The general procedure involves cooling a mixture of the alpha,alpha-dihalo aldehyde and the desired aldehyde in a solvent, followed by the controlled addition of concentrated ammoniacal liquor. The reaction is then allowed to proceed, often at room temperature, for a defined period before standard extraction and purification. This straightforward protocol minimizes the need for specialized equipment or extreme reaction parameters. For detailed operational parameters, stoichiometry, and specific purification techniques tailored to your target molecule, please refer to the standardized synthesis guide below.
- Prepare a mixture of an alpha,alpha-dihalo aldehyde compound and a selected aldehyde compound in an inert organic solvent such as acetonitrile or methanol.
- Cool the mixture in an ice bath and slowly add concentrated ammoniacal liquor (ammonia water) while maintaining low temperature initially.
- Stir the reaction mixture at room temperature or slightly elevated temperatures for several hours, followed by extraction and purification via recrystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel imidazole synthesis method offers tangible strategic benefits that extend beyond mere chemical curiosity. The shift away from hazardous reagents and complex catalytic systems directly impacts the bottom line by reducing raw material costs and minimizing waste disposal fees. Furthermore, the robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining supply continuity in a global pharmaceutical market. By partnering with a supplier who utilizes this advanced technology, organizations can secure a more resilient supply chain that is less susceptible to regulatory crackdowns on hazardous chemical usage.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as hydrogen sulfide and copper acetate results in significant raw material savings. Additionally, the simplified workup process reduces the consumption of solvents and purification media, leading to a lower overall cost per kilogram. The avoidance of heavy metal removal steps further decreases processing time and resource allocation, allowing for more competitive pricing structures in the final API manufacturing cost analysis without compromising on quality standards.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically alpha,alpha-dihalo aldehydes and common aldehydes, are readily available from established chemical suppliers, reducing the risk of raw material shortages. The mild reaction conditions (room temperature) also mean that the process is less energy-intensive and less prone to thermal runaways or equipment failures, ensuring higher uptime and more predictable delivery schedules. This reliability is crucial for long-term project planning and inventory management in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of gas-handling issues associated with H2S. The reduced environmental footprint aligns with increasingly strict global environmental regulations, minimizing the risk of production shutdowns due to compliance issues. The generation of less hazardous waste simplifies disposal logistics and lowers the environmental tax burden, making this a sustainable choice for large-scale commercial production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this imidazole synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential partners and technical evaluators.
Q: Why is the elimination of Copper Acetate and Hydrogen Sulfide critical for API manufacturing?
A: Traditional imidazole synthesis often relies on copper acetate catalysts and toxic hydrogen sulfide gas. Removing these eliminates severe safety hazards for laboratory workers and avoids the complex, costly downstream processing required to remove trace heavy metal residues to meet strict pharmaceutical purity standards.
Q: What is the scope of substituents compatible with this novel condensation method?
A: The method demonstrates exceptional versatility, accommodating a wide range of R1 and R2 groups including alkyl, aryl, heteroaryl, cyanoalkyl, and various functionalized chains. This allows for the synthesis of diverse 2,4-disubstituted imidazoles suitable for different therapeutic classes.
Q: Does this process require extreme temperatures or pressures?
A: No, a key advantage of this patented technology is that the condensation reaction proceeds efficiently at room temperature or with mild heating. This significantly reduces energy consumption compared to traditional high-temperature cyclization methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazoles Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is essential for the future of pharmaceutical manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel imidazole preparation method are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of imidazole intermediate meets the highest international standards, providing our clients with the confidence needed to advance their drug development programs.
We invite you to discuss how this technology can be integrated into your specific supply chain to achieve substantial cost savings and operational improvements. Contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments tailored to your project requirements, helping you secure a competitive edge in the global market.
