Advanced Manufacturing Of Lobaplatin Trihydrate: Technical Breakthroughs And Commercial Scalability
Advanced Manufacturing Of Lobaplatin Trihydrate: Technical Breakthroughs And Commercial Scalability
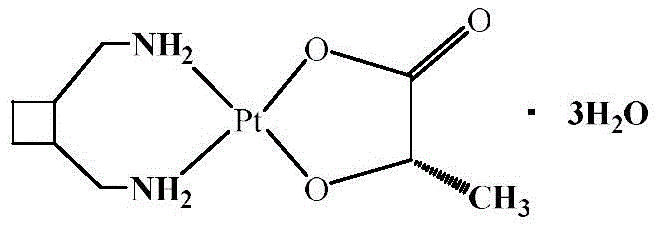
The pharmaceutical industry constantly seeks robust synthetic routes for critical anticancer agents, and the preparation method disclosed in patent CN112225757A represents a significant leap forward in the manufacturing of Lobaplatin Trihydrate. This third-generation platinum-based antitumor drug requires precise coordination chemistry to ensure efficacy and safety, yet traditional methods have often been plagued by complexity and instability. The patented process utilizes potassium chloroplatinite and potassium iodide to generate potassium iodoplatinate in situ, which then reacts with trans-1,2-diaminomethyl-cyclobutane under alkaline conditions. This strategic shift in starting materials allows for the formation of a diiodide intermediate that is subsequently hydrolyzed and coupled with sodium lactate to yield the final trihydrate product. The structural integrity of the final molecule, featuring a platinum center coordinated with a cyclobutane diamine ligand and a lactate group, is critical for its biological activity and is depicted below.
Beyond the chemical transformation, the true value of this patent lies in its operational efficiency and product stability. Unlike the anhydrous form, which suffers from deliquescence and stickiness, the trihydrate form produced by this method exhibits excellent stability and consistent water content. The process achieves high purity levels exceeding 99% and yields around 70%, while drastically reducing the synthesis time from over 20 hours to approximately 10 hours. For R&D directors and procurement managers alike, this translates to a reliable source of high-purity pharmaceutical intermediates that can be scaled with confidence. The ability to recycle acetone with a utilization rate of over 95% further underscores the economic and environmental viability of this approach, positioning it as a superior alternative for commercial scale-up of complex anticancer APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Lobaplatin Trihydrate has relied on two primary methodologies, both of which present significant bottlenecks for large-scale manufacturing. The first method, often involving ion exchange resins, requires the hydrolysate of the diiodide to pass through an anion exchange column. This step is notoriously prone to generating monohydroxy nitrate impurities due to incomplete ion exchange, which complicates downstream purification and necessitates additional decolorization and filtration steps. Furthermore, the second conventional method, which adjusts the pH of the reaction solution, introduces silver chloride as a byproduct during hydrolysis. Silver chloride is an extremely difficult impurity to remove completely, often requiring sequential filtration through filter paper and microporous membranes, thereby imposing stringent requirements on filtering equipment and driving up production costs. Both methods typically require at least 20 hours for the recrystallization and drying stages, creating a bottleneck in throughput.
The Novel Approach
In stark contrast, the novel approach detailed in CN112225757A circumvents these pitfalls by optimizing the ligand substitution sequence and purification strategy. By replacing potassium chloroplatinite with potassium iodoplatinate early in the sequence, the reaction proceeds with greater selectivity, reducing the introduction of extraneous impurities. The refinement of the diiodide intermediate using specific solvents like DMSO prior to hydrolysis ensures that the subsequent reaction with silver nitrate is cleaner, effectively removing silver ions as insoluble silver iodide without the need for complex resin columns. This streamlined workflow eliminates the need for activated carbon decolorization and reduces the number of filtration steps to a single pass through filter paper before concentration. Consequently, the total processing time from recrystallization to drying is cut by half, enabling a faster turnaround for high-purity pharmaceutical intermediates and significantly enhancing the overall economic feasibility of the production line.
Mechanistic Insights into Platinum Coordination And Ligand Substitution
The core of this synthesis relies on the precise manipulation of platinum(II) coordination geometry, specifically leveraging the trans-effect and ligand lability to construct the target molecule. The initial formation of potassium iodoplatinate serves as a crucial activation step, where the softer iodide ligands facilitate the subsequent displacement by the nitrogen donors of the trans-1,2-diaminomethyl-cyclobutane. This substitution occurs under mild alkaline conditions, ensuring that the amine is in its free base form and sufficiently nucleophilic to attack the platinum center. The resulting cis-diiodo complex is thermodynamically stable enough to be isolated and refined, yet labile enough to undergo hydrolysis when treated with silver nitrate. The silver ion acts as a halide scavenger, precipitating iodide as silver iodide and generating a highly reactive aqua-platinum species in situ. This reactive intermediate is then immediately trapped by the lactate anion, locking the platinum into the desired square-planar configuration with the bidentate lactate ligand completing the coordination sphere.
Impurity control is meticulously managed through the physical properties of the intermediates and byproducts. The refinement of the diiodo intermediate in polar aprotic solvents like DMSO followed by precipitation in water exploits solubility differences to exclude organic contaminants before the critical hydrolysis step. This pre-purification is vital because any residual iodide or organic impurities carried into the hydrolysis stage could lead to side reactions or difficult-to-remove metal complexes. Furthermore, the avoidance of ion exchange resins prevents the leaching of organic matrix components or the formation of nitrate adducts that often plague resin-based purifications. The final recrystallization from an acetone-water mixture not only fixes the hydration state to the stable trihydrate form but also serves as a final polishing step, ensuring that the elemental analysis matches theoretical values for carbon, hydrogen, nitrogen, and platinum with high precision.
How to Synthesize Lobaplatin Trihydrate Efficiently
The synthesis of Lobaplatin Trihydrate via this optimized route involves a sequence of seven distinct operational steps, beginning with the in situ generation of the iodoplatinate precursor and concluding with the recrystallization of the final hydrate. The process is designed to be executed in aqueous media wherever possible, minimizing the use of hazardous organic solvents until the final isolation stage. Operators must maintain strict control over temperature and lighting conditions, as platinum complexes can be photosensitive, necessitating reactions to be carried out in the dark. The following guide outlines the standardized protocol derived from the patent examples, ensuring reproducibility and high yield.
- Synthesize potassium iodoplatinate by reacting potassium chloroplatinite with potassium iodide in water.
- React potassium iodoplatinate with neutralized trans-1,2-diaminomethyl-cyclobutane to form the cis-diiodo platinum intermediate.
- Refine the diiodo intermediate using DMSO and water precipitation to remove impurities.
- Hydrolyze the refined intermediate with silver nitrate to remove iodide as silver iodide precipitate.
- React the filtrate with sodium lactate under controlled pH to form Lobaplatin anhydride.
- Recrystallize the anhydride in an acetone-water mixed solvent to obtain Lobaplatin Trihydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits that extend beyond mere chemical yield. The elimination of ion exchange resins and the reduction in filtration complexity directly translate to lower capital expenditure on specialized equipment and reduced maintenance costs. Traditional methods requiring microporous filtration for silver chloride removal demand high-specification hardware that is prone to clogging and failure; by switching to a precipitation-filtration model with silver iodide, the process becomes far more robust and forgiving in a plant setting. Additionally, the significant reduction in processing time—from over 20 hours to roughly 10 hours for the critical finishing stages—allows for higher batch turnover rates, effectively increasing plant capacity without the need for physical expansion. This efficiency gain is a key driver for cost reduction in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the simplification of the purification train. By removing the ion exchange step, manufacturers save on the recurring cost of resin replacement and regeneration chemicals, which are significant operational expenses. Furthermore, the ability to recycle acetone with a utilization rate exceeding 95% drastically cuts solvent procurement costs and waste disposal fees. The qualitative reduction in processing steps means less labor intensity and lower energy consumption for heating and stirring over prolonged periods. These factors combine to create a leaner production model that offers substantial cost savings compared to legacy methods, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthesis and the availability of specialized reagents. This method utilizes readily available starting materials such as potassium chloroplatinite and common salts, reducing the risk of raw material shortages. The robustness of the reaction conditions, which do not require inert gas protection, simplifies the engineering controls needed in the reactor, reducing the likelihood of batch failures due to atmospheric contamination. The improved stability of the trihydrate product also means longer shelf life and reduced risk of degradation during storage and transport. This reliability ensures that downstream formulation partners receive consistent quality material, stabilizing the entire supply chain for this critical anticancer medication.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste generation, but this route incorporates green chemistry principles through solvent recovery. The dedicated acetone recovery step ensures that volatile organic compounds are captured and reused rather than vented, aligning with increasingly strict environmental regulations. The absence of difficult-to-handle sludge from ion exchange resins simplifies waste management and reduces the environmental footprint of the facility. The process is inherently scalable because it relies on standard unit operations like precipitation, filtration, and crystallization, which are well-understood and easily modeled from pilot to commercial scale. This makes the commercial scale-up of complex anticancer agents much more predictable and environmentally sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Lobaplatin Trihydrate, based on the specific advancements detailed in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this new manufacturing standard. The answers reflect the consensus on how this method resolves historical pain points in platinum drug synthesis.
Q: How does the new synthesis method improve upon traditional ion exchange methods?
A: The new method eliminates the need for anion exchange resin, which traditionally caused issues with monohydroxy nitrate impurities and incomplete ion exchange. By directly hydrolyzing the diiodo intermediate with silver nitrate and filtering the precipitate, the process reduces impurity intake and simplifies purification.
Q: What are the stability advantages of Lobaplatin Trihydrate over the anhydrous form?
A: Lobaplatin anhydrate is known to be hygroscopic, sticky, and difficult to prepare with consistent water content. The trihydrate form offers superior stability, non-deliquescent properties, and better repeatability in purity and content, making it ideal for long-term storage and pharmaceutical formulation.
Q: Is the acetone used in recrystallization recoverable in this process?
A: Yes, the process includes a dedicated step for acetone recovery. The filtrate from recrystallization is distilled, and the fraction is dehydrated using drying agents, allowing for a utilization rate of over 95%, which significantly reduces solvent costs and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lobaplatin Trihydrate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate coordination chemistry required for Lobaplatin Trihydrate is managed with precision. We understand that high-purity pharmaceutical intermediates demand rigorous quality control, which is why our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying every batch against the highest international standards. Our commitment to technical excellence ensures that the benefits of the CN112225757A process are fully realized in every kilogram we deliver.
We invite global pharmaceutical partners to collaborate with us to optimize their supply chains and reduce manufacturing costs. By leveraging our advanced synthesis capabilities, you can secure a stable supply of this critical anticancer agent while benefiting from our continuous process improvements. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing prowess can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
