Advanced Manufacturing of Lobaplatin Trihydrate: A Technical Breakthrough in Platinum Antineoplastic Synthesis
The landscape of antineoplastic drug manufacturing is constantly evolving, driven by the need for higher purity, greater stability, and more scalable synthetic routes. A pivotal advancement in this domain is documented in Chinese Patent CN102020679A, which details a novel method for preparing Lobaplatin Trihydrate utilizing an oxalate precursor. This third-generation platinum-based anticancer agent, chemically known as 1,2-diaminomethyl-cyclobutane-lactic acid platinum, represents a significant therapeutic option for treating breast cancer, small cell lung cancer, and chronic myelocytic leukemia. Unlike its predecessors, cisplatin and carboplatin, Lobaplatin offers a distinct toxicity profile and lacks cross-resistance, making its efficient production critical for global oncology supply chains. The patent introduces a strategic shift from using unstable liquid intermediates to stable solid oxalate salts, fundamentally altering the feasibility of large-scale commercial production.
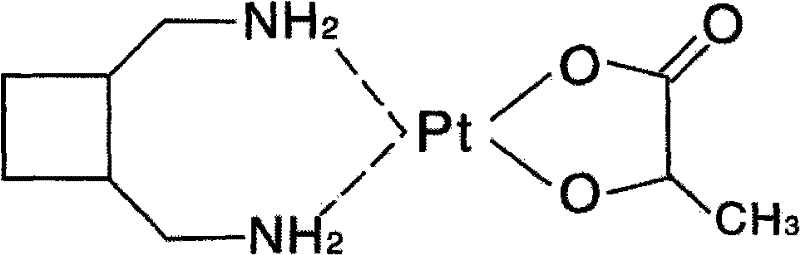
This technical breakthrough addresses long-standing challenges in the synthesis of high-purity pharmaceutical intermediates. By leveraging diaminomethyl cyclobutane oxalate as a starting material, the process mitigates the risks associated with the volatility and instability of the free amine base. Furthermore, the substitution of traditional chloroplatinate precursors with iodoplatinate species enhances reaction kinetics, ensuring a more thorough conversion and minimizing residual metal impurities. For R&D directors and procurement managers alike, understanding this mechanistic shift is essential for evaluating the robustness of the supply chain and the potential for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lobaplatin relied heavily on routes originating from vinyl cyanide or 1,2-dicyanotetramethylene. These conventional pathways involve the generation of trans-1,2-diaminomethylcyclobutane as a key intermediate. However, this intermediate exists as a liquid at room temperature and possesses inherent chemical instability, making it exceptionally difficult to store, transport, and handle safely in an industrial setting. The liquid nature of the amine complicates precise dosing and quality control, often leading to batch-to-batch variability. Moreover, traditional methods frequently employed potassium tetrachloroplatinate (K2PtCl4) as the platinum source. The chloride ligands in K2PtCl4 are relatively poor leaving groups compared to iodides, resulting in slower reaction rates and incomplete coordination. This kinetic barrier often necessitates harsher reaction conditions or extended reaction times, which can degrade sensitive organic ligands and lower the overall yield of the final platinum complex.
The Novel Approach
The methodology outlined in patent CN102020679A circumvents these bottlenecks by introducing diaminomethyl cyclobutane oxalate as the primary amine source. As a crystalline solid, this oxalate salt offers superior stability and ease of handling, eliminating the logistical nightmares associated with liquid amines. The process begins by converting potassium chloroplatinite into potassium iodoplatinite (K2PtI4) in situ. This iodoplatinate species is significantly more reactive towards nucleophilic attack by the amine ligands. The subsequent reaction proceeds under milder conditions with greater completeness, driving the formation of the diiodo-platinum intermediate with high efficiency. Following this, the iodide ligands are cleanly removed via precipitation with silver nitrate, allowing for a seamless transition to the final lactate coordination. This streamlined approach not only boosts yield but also simplifies the purification workflow, making it ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ligand Exchange and Chelation
The core of this synthetic innovation lies in the strategic manipulation of ligand exchange equilibria. The initial step involves the halogen exchange where chloride ligands on the platinum center are replaced by iodide ions from potassium iodide. Iodide is a softer base and a better leaving group than chloride, which labilizes the platinum center and facilitates the subsequent coordination of the bulky 1,2-diaminomethylcyclobutane ligand. This results in the formation of a diiodo-platinum(II) complex. The stability of this intermediate is crucial; it allows for rigorous purification steps, such as recrystallization from N,N-dimethylformamide and acetone, which effectively strip away unreacted starting materials and side products before the final chelation step.
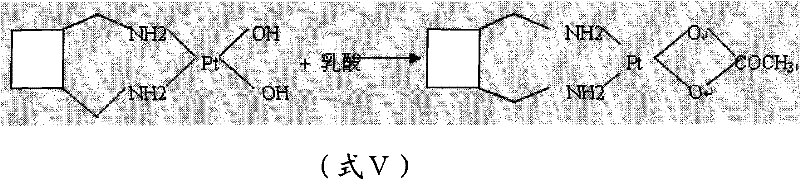
Following purification, the diiodo complex undergoes a metathesis reaction with silver nitrate. The driving force here is the formation of insoluble silver iodide (AgI), which precipitates out of the solution, effectively pulling the equilibrium towards the aquated platinum species. This aquated intermediate is then passed through an anion exchange resin to remove residual iodide and silver ions, ensuring a clean environment for the final chelation. The addition of L-lactic acid then completes the coordination sphere, forming the thermodynamically stable five-membered chelate ring characteristic of Lobaplatin. The final recrystallization from a water/acetone mixture controls the hydration state, yielding the stable trihydrate form rather than the hygroscopic anhydride.
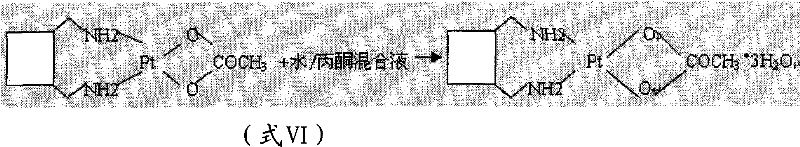
How to Synthesize Lobaplatin Trihydrate Efficiently
The synthesis of Lobaplatin Trihydrate via this oxalate route is a multi-step process that demands precise control over stoichiometry, temperature, and light exposure to prevent photodegradation of the platinum complexes. The procedure generally involves the preparation of the potassium iodoplatinite solution, the neutralization of the oxalate salt to release the free amine in situ, and the subsequent coupling to form the diiodide. After purification of the diiodide, the silver nitrate displacement and ion exchange steps are critical for removing halide impurities. Finally, the reaction with lactic acid and controlled crystallization yields the target API. For a comprehensive, step-by-step technical guide including specific solvent ratios, temperatures, and drying protocols, please refer to the standardized synthesis instructions below.
- Prepare potassium iodoplatinite by reacting potassium chloroplatinite with potassium iodide, then react with neutralized diaminomethyl cyclobutane oxalate to form the diiodide intermediate.
- Purify the diiodide crude product using N,N-dimethylformamide and acetone precipitation to remove impurities.
- React the purified diiodide with silver nitrate to precipitate silver iodide, perform ion exchange, react with lactic acid, and finally recrystallize in water/acetone to obtain the trihydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to the oxalate-based synthesis method described in CN102020679A offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the stabilization of the supply chain for raw materials. By utilizing a solid oxalate salt instead of a volatile liquid amine, manufacturers can significantly reduce storage costs, minimize safety hazards related to flammability and toxicity, and extend the shelf life of key intermediates. This stability translates directly into reducing lead time for high-purity pharmaceutical intermediates, as there is less need for immediate processing upon receipt of materials. Furthermore, the improved reaction kinetics associated with the iodoplatinate route mean that reactor occupancy times can be optimized, potentially increasing throughput without requiring additional capital investment in equipment.
- Cost Reduction in Manufacturing: The enhanced reactivity of the iodoplatinate intermediate leads to higher conversion rates and reduced formation of side products. This intrinsic efficiency means that less raw material is wasted, and the burden on downstream purification processes—such as chromatography or extensive recrystallization—is alleviated. Additionally, the ability to produce a stable trihydrate form directly avoids the complications of handling the hygroscopic anhydride, which often requires specialized packaging and controlled atmosphere storage, thereby lowering packaging and logistics costs substantially.
- Enhanced Supply Chain Reliability: The reliance on stable, solid-state starting materials like diaminomethyl cyclobutane oxalate mitigates the risk of supply disruptions caused by the degradation of liquid intermediates during transport. Solid salts are less susceptible to temperature fluctuations and oxidation, ensuring that the quality of incoming materials remains consistent regardless of shipping duration. This consistency is vital for maintaining GMP compliance and ensures that production schedules are not derailed by out-of-specification raw materials, providing a more predictable and reliable supply of the final active ingredient.
- Scalability and Environmental Compliance: The process utilizes standard unit operations such as precipitation, filtration, and recrystallization, which are easily scalable from pilot plant to multi-ton production. The removal of iodide as silver iodide allows for the potential recovery of silver, adding a value-recovery stream that offsets reagent costs. Moreover, the avoidance of harsh reducing agents like lithium aluminum hydride (used in older routes) improves the overall safety profile of the plant and reduces the generation of hazardous waste, aligning with modern green chemistry principles and environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, purity, and scalability of the Lobaplatin Trihydrate synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear picture of the method's advantages over prior art.
Q: Why is the oxalate salt of diaminomethyl cyclobutane preferred over the free base in Lobaplatin synthesis?
A: The free base (trans-1,2-diaminomethylcyclobutane) is a liquid that is unstable and difficult to store or transport. The oxalate salt is a stable solid, significantly improving handling safety and reaction reproducibility during industrial scale-up.
Q: How does the use of potassium iodoplatinite (K2PtI4) improve the reaction yield compared to potassium chloroplatinite (K2PtCl4)?
A: Potassium iodoplatinite exhibits higher reactivity than potassium chloroplatinite due to the better leaving group ability of the iodide ligand. This facilitates a more complete coordination reaction with the diamine ligand, resulting in higher overall yields of the platinum complex.
Q: What is the role of silver nitrate in the purification process of Lobaplatin?
A: Silver nitrate is used to precipitate iodide ions as insoluble silver iodide (AgI). This removal of iodide drives the equilibrium forward and allows for the subsequent replacement with lactate ligands via ion exchange resin, ensuring high purity of the final active pharmaceutical ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lobaplatin Trihydrate Supplier
The technical advancements detailed in patent CN102020679A underscore the complexity and precision required to manufacture high-quality platinum-based antineoplastics. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Lobaplatin Trihydrate meets the highest international pharmacopeial standards. We understand that the transition from lab-scale discovery to commercial reality requires a partner who can navigate both chemical challenges and regulatory landscapes with equal expertise.
We invite you to collaborate with us to optimize your supply chain for this critical oncology ingredient. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and formulation needs. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects. Let us help you secure a stable, cost-effective, and high-quality supply of Lobaplatin Trihydrate for your global markets.
