Advanced Manufacturing of High-Purity Cefotaxime Acid via Phase-Transfer Catalysis
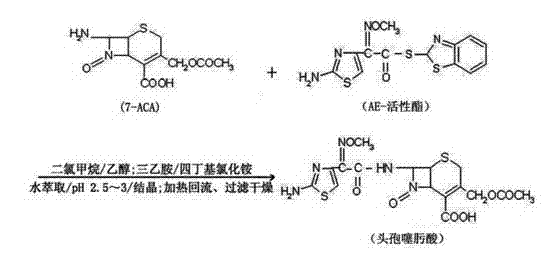
The global demand for third-generation cephalosporins continues to drive innovation in the synthesis of their critical precursors. Patent CN102702230A introduces a transformative methodology for the preparation of cefotaxime acid, a pivotal intermediate in the production of cefotaxime sodium sterile injections. This intellectual property addresses long-standing challenges in the pharmaceutical industry regarding the purity and yield of beta-lactam antibiotics. By integrating a specialized phase-transfer catalytic system with a unique reflux purification protocol, this technology offers a robust solution for manufacturers seeking to optimize their production lines. The method leverages the reaction between 7-aminocephalosporanic acid (7-ACA) and AE-active ester, but distinguishes itself through precise solvent engineering and catalytic enhancement. For R&D directors and procurement specialists, understanding this patented route is essential for securing a reliable cefotaxime acid supplier capable of meeting stringent regulatory standards while maintaining cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of cefotaxime acid has relied heavily on the AE-active ester method due to its operational simplicity compared to phosphorous or triazone active ester routes. However, traditional implementations of this chemistry often suffer from significant drawbacks that impact the final quality of the API. Conventional processes typically involve mixing 7-ACA with the active ester in chlorinated solvents followed by the addition of an organic base. While this approach yields the target molecule, it frequently results in products with suboptimal purity profiles and substantial levels of by-products. These impurities necessitate complex downstream purification steps, which not only increase manufacturing costs but also reduce the overall yield coefficient. Furthermore, the moisture content in traditionally produced cefotaxime acid can be difficult to control, posing stability risks during storage and subsequent formulation into sterile powders. These limitations create bottlenecks for supply chain heads who require consistent, high-quality batches to ensure uninterrupted drug production.
The Novel Approach
The methodology disclosed in the patent data represents a significant evolution in cephalosporin intermediate manufacturing. The core innovation lies in the strategic introduction of a phase-transfer catalyst alongside a specifically engineered solvent system comprising methylene dichloride and a solubility promoter such as isopropanol or ethanol. This dual-modification strategy fundamentally alters the reaction kinetics, allowing for efficient coupling at room temperature within a shortened timeframe of 1 to 2.5 hours. Unlike older methods that might struggle with incomplete conversion or heterogeneous reaction conditions, this novel approach ensures a homogeneous and rapid transformation. Crucially, the process incorporates a dedicated reflux purification step for the wet filter cake, utilizing organic solvents like acetone or ethyl acetate. This post-reaction treatment is instrumental in stripping away residual impurities and controlling moisture levels, directly addressing the purity deficits of legacy technologies. The result is a streamlined workflow that delivers cefotaxime acid with exceptional quality metrics suitable for high-value pharmaceutical applications.
Mechanistic Insights into Phase-Transfer Catalyzed Acylation
To fully appreciate the technical superiority of this synthesis, one must examine the mechanistic role of the phase-transfer catalyst in the acylation of the beta-lactam nucleus. In the reaction mixture, 7-ACA and the AE-active ester exist in an organic phase, while the deprotonation of the amine group on the 7-ACA often requires a base like triethylamine. Without a catalyst, the interaction between these species can be hindered by phase boundaries or steric factors, leading to slower reaction rates and potential degradation of the sensitive beta-lactam ring. The addition of quaternary ammonium salts, such as tetrabutyl ammonium bromide or chloride, facilitates the transport of ionic species across the phase boundary. This catalytic action effectively increases the local concentration of the reactive nucleophile near the electrophilic active ester, thereby accelerating the formation of the amide bond. This enhanced kinetic profile minimizes the time the substrate spends in the reaction vessel, reducing the opportunity for hydrolytic degradation or isomerization side reactions that typically plague cephalosporin synthesis.
Furthermore, the purification mechanism employed in this patent is equally critical for achieving the reported purity levels of greater than 98%. The process involves dissolving the crude wet cefotaxime acid in a heated organic solvent and subjecting it to reflux conditions before controlled cooling and crystallization. This thermal treatment serves multiple functions: it ensures the complete dissolution of the product while leaving behind insoluble mechanical impurities, and upon cooling, it promotes the formation of a highly ordered crystal lattice that excludes soluble impurities. The specific control of pH during the initial crystallization (adjusted to 2.5-3.0 using hydrochloric acid) ensures that the carboxylic acid group is protonated, maximizing precipitation efficiency. This rigorous control over both the reaction catalysis and the crystallization thermodynamics allows for the production of high-purity cefotaxime acid with moisture content consistently below 1%, a vital parameter for the stability of the final antibiotic formulation.
How to Synthesize Cefotaxime Acid Efficiently
The implementation of this patented process requires strict adherence to the specified stoichiometric ratios and environmental controls to replicate the high yields and purity described in the technical literature. The synthesis begins with the precise weighing of 7-ACA and AE-active ester at a mass ratio of approximately 1:1.3, ensuring an excess of the acylating agent to drive the reaction to completion. These starting materials are dissolved in a mixed solvent system where the volume ratio of methylene dichloride to the solubility promoter is tightly controlled, typically around 6.5:1 to 7.0:1. Following dissolution, the sequential addition of triethylamine and the phase-transfer catalyst initiates the exothermic acylation. The reaction is maintained at ambient temperature, eliminating the need for energy-intensive cryogenic cooling often required in other beta-lactam syntheses. Upon completion, the workup involves aqueous extraction and pH adjustment to isolate the crude product, which is then subjected to the critical reflux purification step to achieve pharmaceutical grade quality. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Dissolve 7-ACA and AE-active ester in a mixed solvent of methylene dichloride and a solubility promoter, then add triethylamine and a phase-transfer catalyst.
- Stir the mixture at room temperature for 1 to 2.5 hours to complete the acylation reaction under catalytic conditions.
- Extract the reaction liquid with water, adjust pH to 2.5-3.0 for crystallization, and perform reflux purification on the wet cake to obtain the final dry product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of complex low-temperature requirements and the use of common, commercially available solvents significantly simplify the manufacturing infrastructure needed for production. This simplification translates directly into reduced capital expenditure for specialized cooling equipment and lower operational energy costs. Moreover, the robustness of the phase-transfer catalytic system ensures consistent batch-to-batch reproducibility, a critical factor for maintaining supply continuity in the volatile pharmaceutical market. By minimizing the formation of difficult-to-remove by-products, the process reduces the burden on waste management systems and lowers the cost associated with solvent recovery and disposal. These factors combine to create a highly efficient production model that supports cost reduction in pharmaceutical intermediates manufacturing without compromising on the stringent quality standards required for antibiotic production.
- Cost Reduction in Manufacturing: The integration of phase-transfer catalysis allows the reaction to proceed efficiently at room temperature, removing the need for expensive cryogenic cooling systems and significantly lowering energy consumption. Additionally, the high transformation efficiency reduces the amount of raw material wasted on side reactions, optimizing the utilization of costly starting materials like 7-ACA. The simplified purification workflow further decreases processing time and labor costs, contributing to substantial overall cost savings in the production lifecycle.
- Enhanced Supply Chain Reliability: The reliance on standard solvents such as methylene dichloride, acetone, and ethanol ensures that raw material sourcing remains stable and unaffected by niche supply shortages. The room temperature operation also reduces the risk of process deviations caused by equipment failure in cooling systems, thereby enhancing the reliability of production schedules. This operational stability is crucial for meeting the tight delivery windows demanded by downstream API manufacturers and ensures a steady flow of high-purity cephalosporin intermediates.
- Scalability and Environmental Compliance: The straightforward nature of the reaction and workup procedures makes this method highly amenable to commercial scale-up of complex antibiotic precursors. The process generates fewer hazardous by-products compared to traditional methods, simplifying effluent treatment and ensuring better compliance with increasingly strict environmental regulations. The ability to produce material with low moisture content also reduces the energy load during the final drying stages, further supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of cefotaxime acid synthesized via this novel method. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and quality benchmarks associated with this technology. Understanding these details is vital for partners evaluating the feasibility of integrating this intermediate into their supply chains. The answers reflect the consensus on best practices for handling and processing this sensitive beta-lactam compound to ensure maximum efficacy and stability.
Q: How does the phase-transfer catalyst improve cefotaxime acid purity?
A: The addition of phase-transfer catalysts like tetrabutyl ammonium bromide significantly accelerates the reaction rate and improves transformation efficiency, reducing the formation of side products that typically lower purity in conventional methods.
Q: What is the advantage of the reflux purification step?
A: The novel reflux purification process applied to the wet filter cake effectively removes residual solvents and impurities, ensuring the final product achieves a content greater than 98% and moisture content below 1%.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the process operates at room temperature with simple solvent systems and standard filtration techniques, making it highly scalable and cost-effective for commercial manufacturing compared to complex low-temperature protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotaxime Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the global fight against bacterial infections. Our technical team has extensively analyzed the pathway described in CN102702230A and possesses the expertise to implement this phase-transfer catalytic method at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets rigorous specifications. Our facilities are equipped with state-of-the-art reactors and stringent purity specifications are enforced through our rigorous QC labs, guaranteeing that every batch of cefotaxime acid delivered adheres to the highest international standards for content and moisture levels.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their antibiotic supply chains. By leveraging our technical capabilities, you can achieve significant efficiencies in your production processes. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
