Advanced Recycling Synthesis of AE-Active Ester for Scalable Cephalosporin Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical cephalosporin intermediates to ensure supply chain stability and cost efficiency. A pivotal advancement in this domain is documented in patent CN1321989C, which outlines a novel chemical synthesis method for AE-active ester. This specific intermediate is indispensable for the production of third-generation cephalosporin antibiotics, serving as a key acylating agent. The traditional manufacturing landscape has long been plagued by inefficient atom economy and significant environmental burdens due to the accumulation of phosphine oxide byproducts. However, the methodology described in this patent introduces a transformative recycling mechanism that converts waste streams back into valuable starting materials. By integrating a regeneration step using bis(trichloromethyl) carbonate, the process not only enhances the overall reaction yield but also aligns with modern green chemistry principles. For R&D directors and procurement specialists, understanding this closed-loop system is essential for evaluating long-term supply viability and cost structures in antibiotic manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of this recycling technology, the standard industrial synthesis of AE-active ester relied on stoichiometric amounts of triphenylphosphine and dibenzothiazole disulfide without any recovery strategy. In these legacy processes, the reaction generates substantial quantities of triphenylphosphine oxide and 2-mercaptobenzothiazole as inevitable byproducts. Historically, these compounds were treated as waste residues, often requiring incineration or complex disposal procedures that incurred high operational costs and environmental liabilities. The accumulation of triphenylphosphine oxide, in particular, represents a significant loss of phosphorus resources, driving up the raw material cost per kilogram of the final active ester. Furthermore, the disposal of sulfur-containing waste like 2-mercaptobenzothiazole poses serious pollution challenges, necessitating expensive wastewater treatment infrastructure. From a supply chain perspective, the reliance on continuous fresh inputs of expensive reagents without recovery creates vulnerability to market price fluctuations and raw material shortages.
The Novel Approach
The innovative process described in the patent fundamentally restructures the synthesis workflow by introducing a chemical regeneration cycle for the byproducts. Instead of discarding the reaction filtrate, the method employs bis(trichloromethyl) carbonate to chemically transform the waste triphenylphosphine oxide and 2-mercaptobenzothiazole back into triphenylphosphine and dibenzothiazole disulfide. This regeneration step effectively closes the material loop, allowing the recovered reagents to be reused in subsequent synthesis batches. The technical implication is a drastic reduction in the net consumption of high-value phosphine and sulfur reagents, directly translating to lower variable costs per production run. Additionally, the process operates under mild conditions, typically between 20°C and 60°C, using common solvents like methylene dichloride, which simplifies operational safety and equipment requirements. This approach not only mitigates the environmental impact by eliminating the three wastes but also stabilizes the production cost structure against volatile raw material markets.
Mechanistic Insights into Byproduct Recycling and Esterification
The core chemical transformation involves the activation of cefotaxime acid through a coupling reaction with dibenzothiazole disulfide and triphenylphosphine. In the presence of a catalyst such as pyridine or triethylamine, the triphenylphosphine attacks the disulfide bond, generating a reactive phosphonium intermediate that facilitates the esterification of the carboxylic acid group on the cefotaxime side chain. This step yields the target AE-active ester along with the byproducts triphenylphosphine oxide and 2-mercaptobenzothiazole. The novelty lies in the subsequent treatment of the mother liquor, where bis(trichloromethyl) carbonate acts as a chlorinating and oxidizing agent. It reacts with the triphenylphosphine oxide to form dichlorotriphenylphosphorane, which then reacts with the thiol group of the mercaptobenzothiazole. This sequence regenerates the disulfide bond and releases triphenylphosphine, effectively resetting the chemical state of the reagents for another cycle.
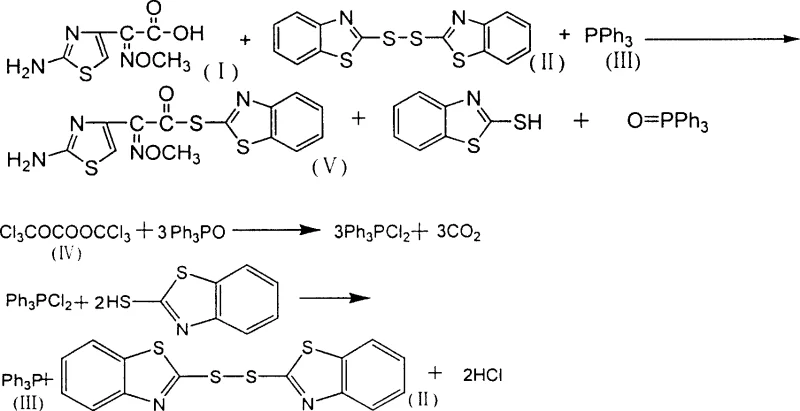
Controlling impurities in this synthesis is critical for ensuring the quality of the final cephalosporin antibiotic. The patent specifies that the reaction mixture is filtered to isolate the AE-active ester filter cake, which is then washed with methanol to remove residual solvents and soluble impurities. The use of specific solvents like methylene dichloride is preferred as it provides optimal solubility for the reactants while allowing for easy crystallization of the product upon cooling. The regeneration step also contributes to purity control by preventing the accumulation of degradation products that might occur if fresh reagents were constantly added without purification. By maintaining a consistent molar ratio of reactants, typically around 1:1.2:1.2 for acid, disulfide, and phosphine, the process minimizes side reactions. The resulting product consistently demonstrates high purity levels, often exceeding 98.0% as determined by liquid chromatography, which is essential for meeting the stringent regulatory standards required for pharmaceutical intermediates.
How to Synthesize AE-Active Ester Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for industrial implementation, emphasizing the importance of precise stoichiometry and temperature control. The process begins with the dissolution of cefotaxime acid, dibenzothiazole disulfide, and a catalyst in an organic solvent, followed by the controlled addition of triphenylphosphine. Maintaining the reaction temperature between 20°C and 25°C during the initial esterification phase is crucial for maximizing yield and minimizing decomposition. After the reaction is complete, the mixture is cooled in an ice bath to precipitate the product, which is then filtered and dried. The filtrate is not discarded but is instead subjected to the regeneration treatment with bis(trichloromethyl) carbonate at similar temperatures to recover the valuable reagents. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- React cefotaxime acid with dibenzothiazole disulfide and triphenylphosphine in an organic solvent like methylene dichloride with a catalyst such as pyridine.
- Filter the reaction mixture to isolate the AE-active ester filter cake, washing and drying it to achieve high purity specifications.
- Treat the filtrate containing byproducts with bis(trichloromethyl) carbonate to regenerate dibenzothiazole disulfide and triphenylphosphine for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this recycling synthesis method offers substantial strategic advantages beyond simple technical feasibility. The primary benefit is the significant reduction in raw material costs achieved through the internal regeneration of expensive reagents like triphenylphosphine and dibenzothiazole disulfide. By converting waste byproducts back into usable inputs, the process drastically lowers the net consumption of these chemicals, insulating the manufacturing operation from external price volatility. This cost efficiency is compounded by the elimination of waste disposal fees associated with hazardous phosphine oxides and sulfur compounds, which can be a major hidden cost in traditional chemical manufacturing. Furthermore, the simplified workflow reduces the dependency on complex waste treatment infrastructure, allowing for more flexible and scalable production facilities.
- Cost Reduction in Manufacturing: The closed-loop recycling mechanism fundamentally alters the cost equation by recovering high-value reagents that would otherwise be lost. In traditional processes, the cost of triphenylphosphine and disulfide reagents constitutes a major portion of the variable cost; recovering these materials significantly lowers the cost of goods sold. Additionally, the reduction in waste generation eliminates the need for expensive hazardous waste disposal services, further enhancing the overall economic viability of the production line. This efficiency allows for more competitive pricing strategies in the global market for cephalosporin intermediates without compromising on quality or margin.
- Enhanced Supply Chain Reliability: Relying on a process that regenerates its own key reagents reduces the vulnerability to supply chain disruptions for raw materials. In scenarios where global demand for triphenylphosphine spikes or logistics are constrained, the ability to recycle internal stocks provides a critical buffer against production stoppages. This self-sufficiency ensures a more consistent and reliable supply of AE-active ester to downstream antibiotic manufacturers. Moreover, the use of common solvents and mild reaction conditions simplifies the procurement of auxiliary materials, reducing the complexity of the supply chain and minimizing the risk of delays due to specialized chemical shortages.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations like filtration, washing, and crystallization that are easily adapted from pilot to commercial scale. The significant reduction in three wastes (waste water, gas, and solid residue) aligns with increasingly stringent environmental regulations, reducing the risk of regulatory fines or shutdowns. This environmental compliance is a key asset for supply chain heads managing risk, as it ensures long-term operational continuity. The ability to scale from 100 kgs to 100 MT annual commercial production with minimal environmental footprint makes this technology a sustainable choice for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this AE-active ester synthesis method. These answers are derived directly from the patent data and practical considerations for industrial scale-up. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their specific production needs. The focus is on clarity regarding yield, purity, and the practical implications of the recycling loop.
Q: How does this synthesis method improve environmental compliance compared to traditional routes?
A: Traditional methods generate significant waste byproducts like triphenylphosphine oxide and 2-mercaptobenzothiazole which are often incinerated. This patented process chemically converts these byproducts back into usable raw materials, drastically reducing solid waste and eliminating the need for hazardous disposal.
Q: What are the typical purity levels achieved with this recycling process?
A: Experimental data from the patent indicates that the AE-active ester produced via this method consistently achieves purity levels exceeding 98.0% as measured by liquid chromatography, meeting stringent requirements for antibiotic manufacturing.
Q: Can the recovered raw materials be used immediately in subsequent batches?
A: Yes, the process is designed to recover dibenzothiazole disulfide and triphenylphosphine with high efficiency. These recovered materials can be directly reintroduced into the synthesis cycle, ensuring a closed-loop system that minimizes raw material procurement costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable AE-Active Ester Supplier
The synthesis route detailed in patent CN1321989C represents a significant leap forward in the efficient production of cephalosporin intermediates, offering a blend of high yield and environmental sustainability. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex recycling chemistries are managed with precision and safety. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest industry standards. We understand that the transition to a recycling-based process requires a partner with deep technical expertise to manage the nuances of reagent recovery and impurity control effectively.
We invite you to collaborate with us to optimize your supply chain for AE-active ester and related pharmaceutical intermediates. Our team is ready to provide a Customized Cost-Saving Analysis to demonstrate how this recycling technology can impact your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. By leveraging our manufacturing capabilities, you can secure a stable, cost-effective, and compliant supply of critical antibiotic intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
