Revolutionizing Ortho-Sulfonylated Arylamine Production: A Metal-Free Synthetic Strategy for Commercial Scale-Up
The landscape of organic synthesis is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective methodologies, particularly in the production of high-value pharmaceutical intermediates. A recent technological breakthrough documented in patent CN113929605A introduces a novel synthetic route for ortho-sulfonylated arylamine compounds, addressing long-standing challenges in regioselectivity and reaction harshness. This invention leverages a unique [2,3]-sigmatropic rearrangement strategy involving aryl hydroxylamine and sulfinyl chloride, bypassing the need for transition metal catalysts or aggressive oxidizing agents. For R&D directors and process chemists, this represents a paradigm shift towards milder, cleaner chemistry that maintains high functional group tolerance. The ability to synthesize these valuable aryl sulfone scaffolds under such benign conditions opens new avenues for drug discovery and process optimization, ensuring that complex molecular architectures can be accessed with greater precision and reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the introduction of sulfonyl groups onto aromatic rings has been fraught with significant chemical and operational hurdles that complicate large-scale manufacturing. The most ubiquitous method involves the oxidation of sulfides, which typically necessitates the use of malodorous mercaptans and strong oxidizing conditions that often lead to poor functional group compatibility and difficult purification processes.  Furthermore, electrophilic substitution using sulfonic acids or sulfonyl chlorides generally requires stoichiometric amounts of Lewis acids and high temperatures, creating safety hazards and increasing energy consumption. While transition metal-catalyzed C-H functionalization strategies have emerged, they often rely on expensive palladium or ruthenium catalysts, require specific directing groups, and suffer from limited substrate scope. These conventional pathways frequently result in incomplete conversions, generating undesirable by-products like sulfoxides that are challenging to separate from the target sulfone, thereby inflating production costs and extending lead times for critical intermediates.
Furthermore, electrophilic substitution using sulfonic acids or sulfonyl chlorides generally requires stoichiometric amounts of Lewis acids and high temperatures, creating safety hazards and increasing energy consumption. While transition metal-catalyzed C-H functionalization strategies have emerged, they often rely on expensive palladium or ruthenium catalysts, require specific directing groups, and suffer from limited substrate scope. These conventional pathways frequently result in incomplete conversions, generating undesirable by-products like sulfoxides that are challenging to separate from the target sulfone, thereby inflating production costs and extending lead times for critical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113929605A offers a streamlined, metal-free alternative that fundamentally simplifies the synthetic workflow. By utilizing aryl hydroxylamine as a starting substrate, the process enables a direct [2,3]-rearrangement that installs the sulfonyl group with exceptional ortho-regioselectivity. 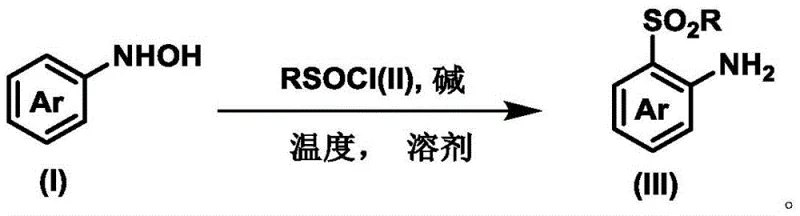 This approach eliminates the dependency on precious metal catalysts and complex oxidants, operating effectively at mild temperatures around 0°C. The reaction kinetics are remarkably fast, often reaching completion within mere minutes, which drastically reduces reactor occupancy time. For procurement and supply chain teams, this translates to a robust process that utilizes readily available reagents like trifluoromethylsulfinyl chloride, avoiding the supply chain volatility associated with specialized catalysts. The high efficiency and selectivity of this novel route ensure that high-purity products are obtained with minimal downstream processing, setting a new standard for the manufacturing of ortho-sulfonylated arylamines.
This approach eliminates the dependency on precious metal catalysts and complex oxidants, operating effectively at mild temperatures around 0°C. The reaction kinetics are remarkably fast, often reaching completion within mere minutes, which drastically reduces reactor occupancy time. For procurement and supply chain teams, this translates to a robust process that utilizes readily available reagents like trifluoromethylsulfinyl chloride, avoiding the supply chain volatility associated with specialized catalysts. The high efficiency and selectivity of this novel route ensure that high-purity products are obtained with minimal downstream processing, setting a new standard for the manufacturing of ortho-sulfonylated arylamines.
Mechanistic Insights into Metal-Free [2,3]-Sigmatropic Rearrangement
The core innovation of this synthesis lies in its elegant mechanistic pathway, which circumvents the need for external activation via metals or light. The reaction initiates with the interaction between the nucleophilic nitrogen of the aryl hydroxylamine and the electrophilic sulfur of the sulfinyl chloride, forming a transient intermediate. This intermediate then undergoes a concerted [2,3]-sigmatropic rearrangement, a pericyclic process that migrates the sulfonyl group directly to the ortho-position of the aromatic ring. This intramolecular migration is driven by thermodynamic stability and orbital symmetry, ensuring that the sulfonyl group is installed precisely where needed without the formation of meta- or para-isomers. Understanding this mechanism is crucial for R&D teams, as it highlights the intrinsic directing capability of the hydroxylamine moiety, removing the need for installing and subsequently removing temporary directing groups common in C-H activation chemistry.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed alternatives. Because the rearrangement is highly specific and occurs under mild conditions, side reactions such as over-oxidation or non-selective radical coupling are effectively suppressed. The absence of transition metals also means there is no risk of metal leaching into the final product, a critical quality attribute for pharmaceutical intermediates intended for clinical use. The process tolerates a wide array of functional groups, including halogens, ethers, and esters, because the mild reaction environment does not promote their degradation. This high level of chemoselectivity ensures that the impurity profile of the final API intermediate remains clean and predictable, simplifying the regulatory filing process and reducing the burden on analytical quality control laboratories.
How to Synthesize Ortho-Sulfonylated Arylamine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process begins with the preparation of the aryl hydroxylamine substrate, which can be derived from the corresponding nitro compound via reduction, followed by immediate reaction with the sulfinyl chloride in a dry, inert solvent system. The choice of base and solvent plays a pivotal role; experimental data indicates that triethylamine in dichloromethane provides optimal results, facilitating the rapid formation of the target sulfone. Detailed standardized operating procedures regarding stoichiometry, addition rates, and workup protocols are essential for reproducibility.
- Dissolve the aryl hydroxylamine substrate in an ultra-dry solvent such as dichloromethane (DCM) under a nitrogen atmosphere and cool the mixture to 0°C.
- Slowly add a base such as triethylamine followed by the dropwise addition of the sulfinyl chloride reagent while maintaining stirring at low temperature.
- Stir the reaction mixture for approximately 10 minutes, monitor by TLC, then concentrate and purify the crude product via column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this patented methodology offers compelling economic and logistical benefits that address traditional pain points in fine chemical manufacturing. By eliminating the requirement for expensive transition metal catalysts such as palladium or ruthenium, the raw material costs are significantly reduced, and the complex, costly steps associated with metal scavenging and removal are entirely avoided. This simplification of the downstream processing workflow not only lowers operational expenditures but also accelerates the overall production cycle, allowing for faster turnaround times on customer orders. Furthermore, the reliance on commodity chemicals like aryl hydroxylamines and sulfinyl chlorides ensures a stable supply chain, mitigating the risks associated with sourcing specialized or proprietary reagents that may face availability constraints.
- Cost Reduction in Manufacturing: The exclusion of precious metal catalysts represents a direct and substantial saving in raw material costs, while the mild reaction conditions reduce energy consumption for heating and cooling. Additionally, the high regioselectivity minimizes the formation of isomeric by-products, leading to higher crude yields and reducing the solvent and silica usage required for purification. This cumulative effect results in a leaner, more cost-efficient manufacturing process that enhances profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials ensures that production is not bottlenecked by the scarcity of exotic reagents. The robustness of the reaction conditions means that the process is less sensitive to minor variations in input quality, providing a consistent output that supports reliable delivery schedules. This stability is crucial for maintaining long-term contracts with pharmaceutical partners who demand uninterrupted supply of critical intermediates for their drug development pipelines.
- Scalability and Environmental Compliance: The mild nature of the reaction, operating near room temperature and completing rapidly, makes it inherently safer and easier to scale from kilogram to multi-ton quantities. The absence of heavy metals and strong oxidants aligns with increasingly stringent environmental regulations, reducing the cost and complexity of waste treatment and disposal. This green chemistry profile not only lowers compliance risks but also enhances the corporate sustainability metrics of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ortho-sulfonylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this method. Understanding these details helps potential partners evaluate the feasibility of integrating this route into their existing supply chains.
Q: Does this synthesis method require transition metal catalysts?
A: No, the patented method described in CN113929605A operates under transition metal-free conditions, utilizing a [2,3]-sigmatropic rearrangement mechanism instead of traditional palladium or ruthenium catalysis.
Q: What are the typical reaction conditions for this sulfonylation?
A: The reaction proceeds under mild conditions, typically at temperatures ranging from -10°C to 10°C (optimally 0°C), completing within a short timeframe of approximately 10 minutes without the need for high heat or strong oxidants.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process demonstrates excellent scalability due to its mild conditions, short reaction time, and the use of commercially available reagents, making it highly suitable for industrial production of complex aryl sulfone intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Sulfonylated Arylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic route for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ortho-sulfonylated arylamine delivered meets the exacting standards required for global drug registration. We are committed to leveraging advanced technologies like this rearrangement strategy to provide our clients with superior chemical solutions that drive their projects forward.
We invite you to engage with our technical procurement team to discuss how this efficient synthesis can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits tailored to your volume needs. We encourage you to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your supply chain with reliable, high-quality intermediates. Let us collaborate to engineer a more efficient and sustainable future for your chemical manufacturing needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
