Revolutionizing Ortho-Sulfonylated Arylamine Production via Metal-Free [2,3]-Rearrangement for Commercial Scale-Up
The landscape of organic synthesis for high-value sulfur-containing intermediates is undergoing a significant transformation, driven by the urgent need for greener, more efficient manufacturing protocols. Patent CN113929605B introduces a groundbreaking methodology for the preparation of ortho-sulfonylated arylamine compounds, a structural motif prevalent in numerous bioactive molecules and functional materials. Unlike traditional approaches that rely on harsh oxidizing environments or expensive transition metal catalysis, this invention leverages a clever tandem rearrangement strategy. By utilizing aryl hydroxylamine compounds as the primary substrate and reacting them with readily available sulfinyl chlorides, the process achieves high-efficiency synthesis under remarkably mild conditions. This technological leap not only simplifies the synthetic route but also addresses critical pain points regarding functional group tolerance and regioselectivity, making it an ideal candidate for the production of complex pharmaceutical intermediates and agrochemical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of sulfonyl groups onto aromatic rings has been fraught with chemical challenges and operational inefficiencies. The most ubiquitous method involves the oxidation of sulfides, a pathway that typically necessitates the use of malodorous thiols and strong oxidizing agents such as peracids or hydrogen peroxide combinations. 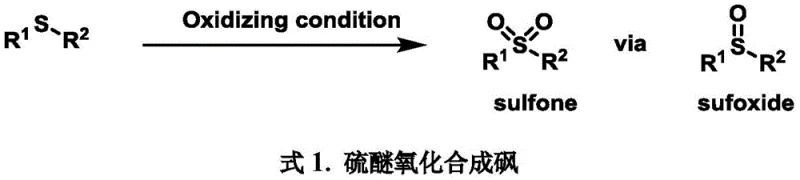 As illustrated in the conventional oxidation pathways, these reactions often suffer from poor functional group compatibility and require excessive amounts of oxidants to drive the conversion to completion. Furthermore, controlling the oxidation state is difficult; incomplete conversion leads to sulfoxide impurities that are notoriously difficult to separate from the desired sulfone product, thereby complicating downstream purification and reducing overall yield. Alternatively, direct C-H functionalization using transition metals like palladium or ruthenium offers a different set of drawbacks, including the requirement for stoichiometric Lewis acids, high temperatures, and the necessity of installing specific directing groups which adds extra synthetic steps and cost.
As illustrated in the conventional oxidation pathways, these reactions often suffer from poor functional group compatibility and require excessive amounts of oxidants to drive the conversion to completion. Furthermore, controlling the oxidation state is difficult; incomplete conversion leads to sulfoxide impurities that are notoriously difficult to separate from the desired sulfone product, thereby complicating downstream purification and reducing overall yield. Alternatively, direct C-H functionalization using transition metals like palladium or ruthenium offers a different set of drawbacks, including the requirement for stoichiometric Lewis acids, high temperatures, and the necessity of installing specific directing groups which adds extra synthetic steps and cost.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113929605B presents a paradigm shift by employing a metal-free [2,3]-sigmatropic rearrangement. This novel approach bypasses the need for external oxidants entirely, relying instead on the intrinsic reactivity of the aryl hydroxylamine substrate.  The reaction proceeds through a tandem mechanism where the sulfinyl chloride reacts with the hydroxylamine to form an intermediate that spontaneously rearranges to place the sulfonyl group exclusively at the ortho-position. This strategy eliminates the generation of heavy metal waste and avoids the safety hazards associated with strong oxidizers. The process operates at low temperatures, typically around 0°C, and completes within minutes, representing a drastic improvement in energy efficiency and throughput compared to thermal processes that require prolonged heating. This makes the novel approach particularly attractive for the cost reduction in pharmaceutical intermediate manufacturing where purity and speed are paramount.
The reaction proceeds through a tandem mechanism where the sulfinyl chloride reacts with the hydroxylamine to form an intermediate that spontaneously rearranges to place the sulfonyl group exclusively at the ortho-position. This strategy eliminates the generation of heavy metal waste and avoids the safety hazards associated with strong oxidizers. The process operates at low temperatures, typically around 0°C, and completes within minutes, representing a drastic improvement in energy efficiency and throughput compared to thermal processes that require prolonged heating. This makes the novel approach particularly attractive for the cost reduction in pharmaceutical intermediate manufacturing where purity and speed are paramount.
Mechanistic Insights into Metal-Free [2,3]-Sigmatropic Rearrangement
The core of this innovation lies in the elegant mechanistic pathway that ensures both high yield and exceptional regioselectivity. The reaction initiates with the nucleophilic attack of the nitrogen atom in the aryl hydroxylamine on the sulfur center of the sulfinyl chloride. This step generates a reactive O-sulfinyl hydroxylamine intermediate. Unlike traditional electrophilic aromatic substitutions which are governed by electronic effects that can lead to mixtures of ortho, meta, and para isomers, this intermediate undergoes a concerted [2,3]-sigmatropic rearrangement. This pericyclic process is thermally allowed and proceeds through a cyclic transition state that inherently directs the sulfonyl group to the carbon atom adjacent to the nitrogen, i.e., the ortho-position. This mechanistic constraint effectively locks the regioselectivity, preventing the formation of unwanted isomers that would otherwise require costly chromatographic separation.
From an impurity control perspective, this mechanism offers distinct advantages for R&D teams focused on quality. Because the reaction does not involve radical species generated by photoredox catalysts or high-energy oxidants, the potential for non-specific radical coupling or over-oxidation side reactions is minimized. The absence of transition metals means there is no risk of metal-catalyzed decomposition of sensitive functional groups, such as halides or esters, which are common in drug scaffolds. Furthermore, the mild basic conditions employed (using bases like triethylamine) are sufficient to scavenge the HCl byproduct without promoting hydrolysis of other sensitive moieties. This robustness allows for the synthesis of structurally diverse aryl sulfones, including those with complex heterocyclic cores, ensuring that the final API intermediate meets stringent purity specifications required for clinical applications.
How to Synthesize Ortho-Sulfonylated Arylamine Efficiently
The practical execution of this synthesis is designed for simplicity and scalability, making it accessible for both laboratory discovery and pilot plant operations. The standard protocol involves dissolving the aryl hydroxylamine precursor in an anhydrous solvent such as dichloromethane under an inert nitrogen atmosphere to prevent moisture interference. The reaction mixture is cooled to 0°C to manage the exotherm upon addition of the reagents. A base, optimally triethylamine, is added to neutralize the acid generated during the reaction, followed by the dropwise addition of the sulfinyl chloride. The detailed standardized synthesis steps are provided in the guide below.
- Under a nitrogen atmosphere, dissolve the aryl hydroxylamine compound (Compound I) in anhydrous dichloromethane (DCM) and cool the solution to 0°C.
- Slowly add triethylamine (1.5 equivalents) to the mixture, followed by the dropwise addition of sulfinyl chloride (Compound II, 1.2 equivalents).
- Stir the reaction mixture at 0°C for 10 minutes, monitor by TLC, then concentrate and purify via column chromatography to obtain the target ortho-sulfonylated arylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route translates into tangible strategic benefits beyond mere chemical elegance. The elimination of precious metal catalysts such as palladium or ruthenium removes a significant cost driver from the bill of materials. More importantly, it obviates the need for specialized and expensive heavy metal scavenging resins or activated carbon treatment steps that are mandatory when using transition metal catalysis. This streamlining of the downstream processing significantly reduces the consumption of auxiliary materials and shortens the overall production cycle time, leading to substantial cost savings in API intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic model of this process is highly favorable due to the use of commodity chemicals. Sulfinyl chlorides and aryl hydroxylamines are generally more affordable and stable than the specialized ligands and catalysts required for C-H activation. By removing the dependency on volatile precious metal markets, manufacturers can achieve more stable pricing and better margin protection. Additionally, the high atom economy of the rearrangement reaction minimizes waste generation, reducing the costs associated with waste disposal and environmental compliance.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity of the reagent list. The starting materials are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The mild reaction conditions (0°C to room temperature) mean that the process does not require specialized high-pressure or high-temperature reactors, allowing for production in a wider range of standard multipurpose facilities. This flexibility ensures consistent supply continuity even during periods of high demand or equipment maintenance.
- Scalability and Environmental Compliance: Scaling this reaction from grams to tons is straightforward because the reaction kinetics are fast and the heat load is manageable. The short reaction time of approximately 10 minutes allows for high throughput in batch reactors or potential adaptation to continuous flow chemistry for even greater efficiency. From an environmental standpoint, the absence of heavy metals and strong oxidants aligns perfectly with green chemistry principles, simplifying the regulatory approval process for new drug filings and reducing the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ortho-sulfonylation technology. These insights are derived directly from the experimental data and scope analysis provided in the patent documentation, offering clarity on its applicability to your specific project needs.
Q: Does this synthesis method involve transition metal catalysts?
A: No, the method described in patent CN113929605B is completely metal-free. It utilizes a tandem rearrangement reaction between aryl hydroxylamines and sulfinyl chlorides, eliminating the need for expensive palladium or ruthenium catalysts and the subsequent heavy metal removal steps.
Q: What is the regioselectivity of this sulfonylation reaction?
A: The reaction exhibits excellent regioselectivity, specifically targeting the ortho-position of the aromatic ring relative to the amino group. This is achieved through a [2,3]-sigmatropic rearrangement mechanism, avoiding the mixtures of isomers often seen in traditional electrophilic substitutions.
Q: What types of substrates are compatible with this protocol?
A: The protocol demonstrates broad substrate scope, successfully accommodating substituted phenyl rings, naphthyl systems, and various heteroaryl compounds. Functional groups such as halogens, alkoxy groups, and esters are well-tolerated under the mild reaction conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Sulfonylated Arylamine Supplier
The technological potential of metal-free [2,3]-rearrangement for synthesizing ortho-sulfonylated arylamines represents a significant opportunity for accelerating drug development pipelines. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required by top-tier pharmaceutical companies.
We invite you to leverage our technical expertise to optimize your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this advanced synthesis method can enhance your project's efficiency and profitability.
