Advanced Manufacturing of Minoxidil Intermediates via Novel Pyrimidine Activation
The pharmaceutical and cosmetic industries have long recognized the critical value of 6-amino-1,2-dihydro-1-hydroxy-2-imino-4-piperidinopyrimidine, widely known by its unregistered trade name Minoxidil. As a potent anti-stress agent and vasodilator, this compound serves as the active ingredient in numerous blood pressure-lowering compositions and has gained immense popularity as a therapeutic cosmetic for stimulating hair growth. However, the historical manufacturing landscape for this vital molecule has been plagued by inefficiency and prohibitive costs. Patent CN87104694A introduces a groundbreaking technological shift, detailing a novel process that fundamentally reimagines the synthetic pathway to this key intermediate. By leveraging activated pyrimidine derivatives, this invention overcomes the severe limitations of prior art, offering a route that is not only chemically elegant but also commercially viable for large-scale operations. The core innovation lies in the strategic use of electron-withdrawing groups to facilitate nucleophilic substitution under remarkably mild conditions, a stark contrast to the brutal thermal requirements of legacy methods.
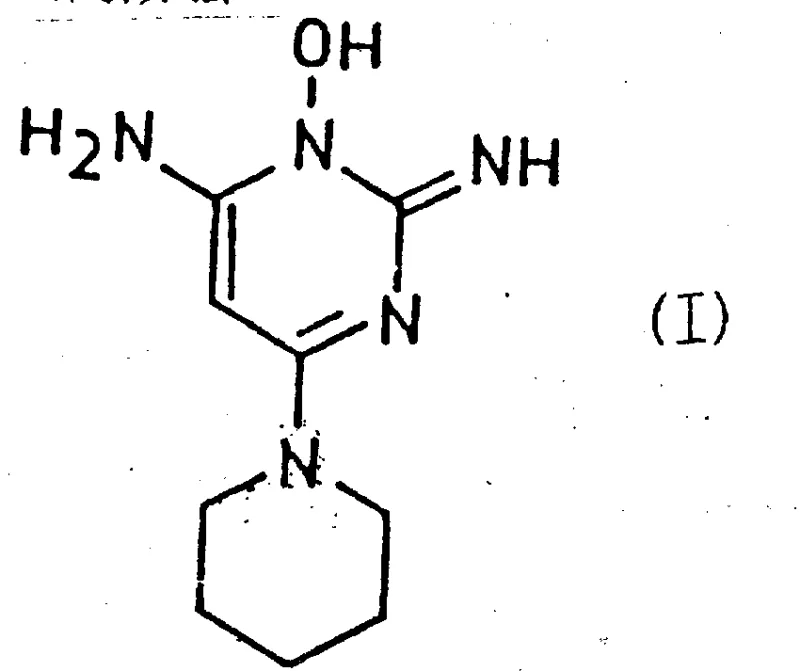
This technical breakthrough addresses the urgent demand for a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials without the baggage of complex purification protocols. The patent explicitly targets the economic and practical bottlenecks that have hindered the widespread adoption of earlier synthesis routes. For R&D directors and procurement managers alike, the implications are profound: a transition from a low-yield, high-waste process to one that boasts quantitative conversion rates and simplified downstream processing. The ability to produce this compound in high yields and high purity on an industrial scale represents a significant leap forward in fine chemical manufacturing, promising to stabilize supply chains and reduce the total cost of ownership for downstream API producers. As we delve deeper into the mechanistic nuances and commercial advantages, it becomes clear that this patent provides the blueprint for the next generation of Minoxidil production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Formula (I) compounds was fraught with chemical and economic challenges that rendered them unsuitable for modern, efficient manufacturing. The earliest documented methods, such as those described in British Patent Specification No. 1,167,735, relied on the reaction of 4-chloro-2,6-diaminopyrimidine with 2,4-dichlorophenol under extreme thermal stress. This archaic approach required heating the mixture to 150°C in the presence of concentrated potassium hydroxide, a condition that inevitably promoted undesirable side reactions and degradation of the sensitive pyrimidine ring. The subsequent oxidation step to introduce the N-oxide functionality was notoriously inefficient, proceeding in low yields that crippled the overall economics of the process. Ultimately, the final substitution of the 2,4-dichlorophenoxy group with piperidine yielded a dismal 45% conversion, contributing to an abysmal overall yield of approximately 2.5%. Such inefficiency is unacceptable in a contemporary supply chain, where material throughput and waste minimization are paramount KPIs for any cost reduction in API manufacturing initiative.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the process disclosed in CN87104694A utilizes a highly activated pyrimidine derivative of general formula (II) as the starting material. This innovative strategy replaces the inert chlorophenoxy leaving group with electron-withdrawing acyl or acyloxy substituents, which dramatically increase the electrophilicity of the C-4 position on the pyrimidine ring. This activation allows for a facile nucleophilic attack by piperidine, occurring rapidly at temperatures ranging from 0°C to 100°C, and often merely at room temperature. The versatility of this approach is further enhanced by the tolerance for various solvents, including protic solvents like ethanol, dipolar aprotic solvents like acetonitrile, or even the use of excess piperidine as the reaction medium itself. By shifting the paradigm from thermal forcing to electronic activation, this novel approach eliminates the need for harsh conditions that degrade product quality and damage reactor infrastructure.
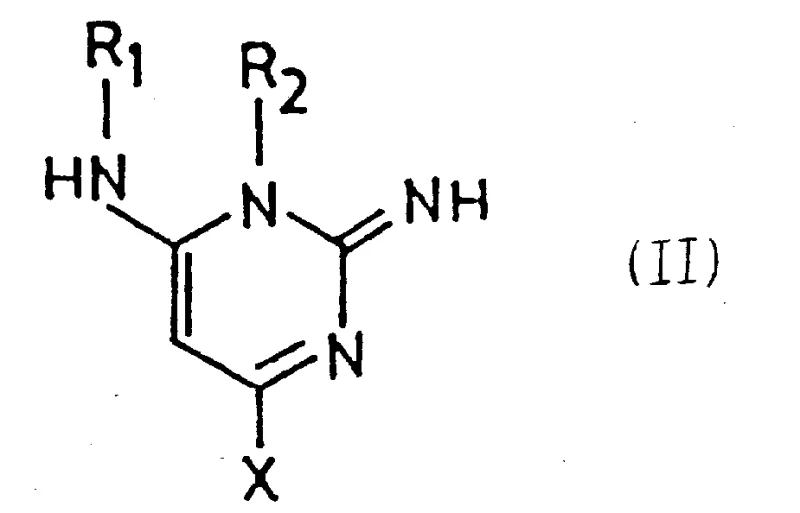
The structural flexibility of Formula (II) allows manufacturers to tailor the process to their specific operational capabilities, whether they prioritize speed or ease of isolation. The electron-withdrawing nature of the R1 and R2 groups—typically acetyl or acetoxy moieties—ensures that the nucleophilic substitution proceeds with virtually quantitative yield, a massive improvement over the fractional conversions of the past. Furthermore, the starting materials for Formula (II) are themselves novel compounds that can be prepared in high yields from readily available 2,6-diaminopyrimidine derivatives, ensuring a robust and continuous supply of feedstock. This fundamental redesign of the synthetic entry point transforms the production of Minoxidil intermediates from a bottleneck into a streamlined, high-efficiency operation that aligns perfectly with the goals of a reliable pharmaceutical intermediates supplier.
Mechanistic Insights into Activated Pyrimidine Substitution
The success of this novel synthesis hinges on a sophisticated understanding of electronic effects within the heterocyclic system. The key mechanistic driver is the nucleophilic substitution of the compound of formula (II) with piperidine, a reaction that is exceptionally facile due to the specific electronic environment created by the R1 and R2 substituents. When R1 or R2 represents an acyl or acyloxy group, these moieties exert a strong electron-withdrawing effect that significantly decreases the electron density at the 4-position of the pyrimidine ring. This depletion of electron density renders the carbon atom at position 4 highly susceptible to nucleophilic attack by the lone pair of electrons on the piperidine nitrogen. Unlike the stubborn chlorophenoxy group of the prior art, which required 150°C to displace, the activated leaving groups in Formula (II) depart readily under mild thermal conditions. This electronic activation not only accelerates the reaction kinetics, allowing completion in minutes to hours, but also ensures high regioselectivity, preventing the formation of isomeric by-products that complicate purification.
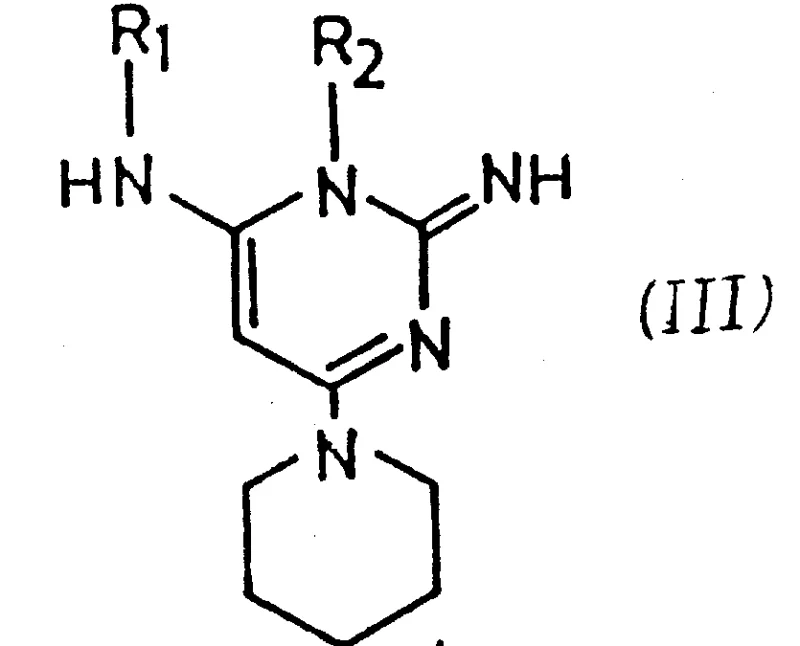
Following the substitution step, the process leverages the unique structural characteristics of the N-oxide portion of the molecule to achieve rapid deprotection. The intermediate 4-piperidino derivative of general formula (III) contains acyl or acyloxy groups that serve a dual function: activation and protection. These groups shield the sensitive amino functionalities from oxidation or other side reactions during the substitution phase. Surprisingly, these protecting groups are easily eliminated by hydrolysis, even at room temperature, when treated with a base such as aqueous sodium hydroxide. This ease of elimination is attributed to the neighboring N-oxide group, which facilitates the hydrolytic cleavage of the ester or amide bonds. Any acyl groups initially present at other positions migrate to the 1-position under acidic or basic conditions and are subsequently hydrolyzed at a rate equivalent to the ester hydrolysis. This self-correcting mechanistic feature ensures that the final isolated compound is a highly pure crystal, free of any detectable by-products, thereby satisfying the stringent purity specifications required for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 6-amino-1,2-dihydro-1-hydroxy-2-imino-4-piperidinopyrimidine Efficiently
The practical execution of this synthesis is designed for operational simplicity and robustness, making it accessible for facilities ranging from pilot plants to multi-ton production suites. The general procedure involves reacting the activated pyrimidine derivative with piperidine, followed by a straightforward hydrolysis step that can be performed with or without isolating the intermediate. Detailed examples in the patent demonstrate that mixing 1-acetoxy-6-amino-4-chloro-1,2-dihydro-2-iminopyrimidine with piperidine in ethanol, followed by reflux and subsequent treatment with aqueous sodium hydroxide, yields the target compound in upwards of 82% purity. The flexibility of the protocol allows for variations in solvent and temperature, enabling process engineers to optimize for their specific equipment constraints while maintaining high efficiency. For a comprehensive breakdown of the standardized operating procedures and safety parameters, please refer to the technical guide below.
- React a pyrimidine derivative of general formula (II), containing electron-withdrawing acyl or acyloxy groups, with piperidine under mild conditions (0-100°C).
- Isolate the intermediate 4-piperidino derivative of general formula (III) or proceed directly without isolation depending on solvent choice.
- Hydrolyze the acyl or acyloxy protecting groups using aqueous alkali metal hydroxide to yield the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers compelling strategic advantages that extend far beyond simple chemistry. The primary benefit lies in the drastic simplification of the manufacturing workflow, which directly translates to substantial cost savings and enhanced supply chain reliability. By eliminating the need for high-temperature reactors and exotic reagents like 'magic-methyl' or trimethylfluoroborate, the process significantly lowers the barrier to entry for production and reduces the capital expenditure required for specialized equipment. The mild reaction conditions also imply a longer lifespan for reactor vessels and reduced energy consumption, contributing to a lower carbon footprint and improved sustainability metrics. These factors collectively position this technology as a cornerstone for cost reduction in pharmaceutical intermediates manufacturing, allowing companies to remain competitive in a price-sensitive global market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and harsh oxidizing agents removes significant line items from the bill of materials. Furthermore, the high yields achieved (70-80% compared to the historical 2.5%) mean that less raw material is wasted per kilogram of finished product, drastically improving the material balance and reducing waste disposal costs. The ability to use common solvents like ethanol and water for workup further minimizes solvent recovery expenses, creating a leaner and more cost-effective production model that maximizes margin potential.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically the activated pyrimidine derivatives, are synthesized from readily available precursors like 2,6-diaminopyrimidine, ensuring a stable and continuous supply of feedstock. Unlike legacy methods that relied on hard-to-source reagents prone to supply disruptions, this route utilizes commodity chemicals that are widely produced and stocked globally. This accessibility reduces lead time for high-purity pharmaceutical intermediates and mitigates the risk of production stoppages due to raw material shortages, providing a buffer against market volatility.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram-scale experiments to multi-kilogram batches without loss of efficiency. The mild conditions and aqueous workup simplify the handling of effluents, making it easier to meet stringent environmental regulations regarding wastewater treatment. The reduction in hazardous by-products and the avoidance of heavy metals streamline the regulatory approval process for new manufacturing sites, facilitating faster time-to-market for generic formulations and ensuring long-term operational compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and theoretical framework presented in CN87104694A, providing clarity for stakeholders evaluating the feasibility of adoption. Understanding these nuances is critical for making informed decisions about process integration and supplier selection.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: The novel process eliminates the need for harsh reaction conditions (150°C) and difficult starting materials like 2,4-dichlorophenol. It achieves significantly higher yields (70-80% vs 2.5% overall) and utilizes readily available activated pyrimidine derivatives, reducing both cost and environmental impact.
Q: How does the process ensure high purity of the final Minoxidil intermediate?
A: The acyl or acyloxy groups serve a dual purpose: they activate the ring for substitution and simultaneously protect sensitive amino groups from side reactions. The final hydrolysis step is highly selective due to the N-oxide structure, resulting in a crystalline product free of detectable by-products.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the reaction proceeds rapidly at room temperature or under mild reflux, using common solvents like ethanol or excess piperidine. The simplicity of the workup (filtration and washing) and the high conversion rates make it ideal for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-amino-1,2-dihydro-1-hydroxy-2-imino-4-piperidinopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible production capabilities to create real value. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity promised by this novel chemistry are realized in every batch. Our state-of-the-art facilities are equipped to handle the specific solvent systems and mild thermal profiles required by this process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every gram of material meets the exacting standards of the global pharmaceutical industry. We are committed to being a partner that not only supplies chemicals but also optimizes the entire value chain for our clients.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-quality intermediates with the reliability and consistency your business demands.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
