Advanced Minoxidil Manufacturing: High-Yield Oxidation and Condensation Strategies
The global pharmaceutical landscape is witnessing a sustained surge in demand for effective alopecia treatments, placing Minoxidil at the forefront of dermatological therapeutics. Patent CN107235919B introduces a robust and scalable synthesis methodology that addresses historical bottlenecks in producing this vital active pharmaceutical ingredient (API). The disclosed process delineates a streamlined three-stage sequence: the selective oxidation of 2,4-diamino-6-chloropyrimidine, followed by a nucleophilic condensation with piperidine, and concluding with a precision crystallization protocol. This technical breakthrough is not merely an academic exercise but represents a viable industrial solution capable of delivering high-purity Minoxidil with exceptional consistency. By leveraging m-chloroperoxybenzoic acid (mCPBA) as a specialized oxidant, the route ensures the formation of the critical N-oxide intermediate with minimal side reactions, setting a new benchmark for efficiency in heterocyclic chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of Minoxidil has been plagued by complex multi-step sequences that often result in suboptimal yields and challenging purification hurdles. Traditional pathways frequently rely on harsh reaction conditions that can degrade the sensitive pyrimidine ring, leading to a broad spectrum of impurities that are difficult to separate. Furthermore, legacy processes often utilize stoichiometric amounts of expensive reagents without efficient recovery systems, driving up the cost of goods sold (COGS) significantly. The lack of thermal control in older exothermic oxidation steps poses safety risks and limits the ability to scale up production safely in large-scale reactors. Consequently, procurement teams have faced volatility in supply continuity and pricing, as manufacturers struggle to maintain batch-to-batch consistency under these demanding and inefficient operational parameters.
The Novel Approach
In stark contrast, the methodology outlined in CN107235919B employs a strategic oxidation-condensation cascade that simplifies the operational workflow while maximizing output. The novel approach utilizes a controlled reflux system in chloroform to manage the exothermic nature of the oxidation, ensuring complete conversion of the starting material into the 2,6-diamino-4-chloropyrimidine-1-oxide intermediate. This is followed by a base-mediated condensation in acetone, which proceeds smoothly at moderate temperatures (55-60°C), thereby preserving the structural integrity of the molecule. The integration of a low-temperature crystallization step using isopropanol allows for the rejection of trace impurities, resulting in a white crystalline powder that meets rigorous quality standards. This streamlined architecture eliminates unnecessary unit operations, directly translating to reduced processing time and enhanced throughput for commercial manufacturing facilities.
Mechanistic Insights into mCPBA-Mediated N-Oxidation and Substitution
The cornerstone of this synthesis is the selective N-oxidation of the pyrimidine ring, a transformation that activates the heterocycle for subsequent nucleophilic attack. In the first stage, m-chloroperoxybenzoic acid acts as an oxygen transfer agent, targeting the ring nitrogen atom to form the N-oxide functionality. This electronic modification significantly increases the electrophilicity of the adjacent carbon atoms, particularly the C4 position bearing the chlorine substituent. The reaction is meticulously managed by maintaining an initial temperature of 15-18°C during reagent addition to prevent runaway exotherms, followed by a prolonged reflux period to drive the equilibrium toward completion. The use of chloroform as the solvent is pivotal, as it provides an ideal polarity balance to solubilize both the polar oxidant and the organic substrate, facilitating intimate molecular contact.
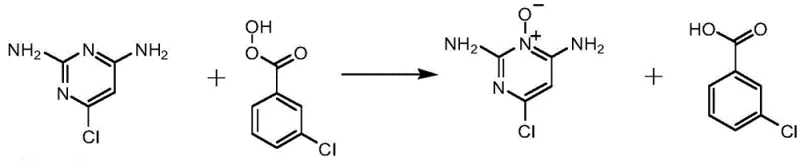
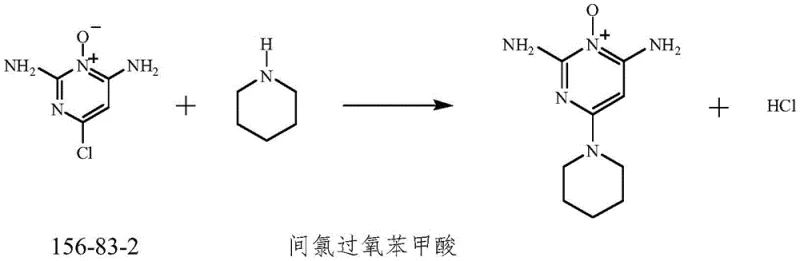
Following the formation of the N-oxide intermediate, the process advances to a nucleophilic aromatic substitution where piperidine displaces the chloride ion. The presence of the N-oxide group and the electron-donating amino substituents creates a highly activated system that readily accepts the piperidine nucleophile under mild alkaline conditions provided by potassium carbonate. This step is conducted in acetone, a solvent chosen for its ability to dissolve the inorganic base and the organic intermediates while allowing for easy removal post-reaction. The mechanistic pathway avoids the formation of stable by-products, ensuring that the crude product obtained is predominantly the desired Minoxidil species. This high selectivity minimizes the burden on downstream purification, a critical factor in achieving the reported 75% overall yield and high purity profile.
How to Synthesize Minoxidil Efficiently
Implementing this synthesis route requires precise adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and safety. The process begins with the careful preparation of the oxidation mixture, where temperature control is paramount to managing the reaction kinetics. Operators must monitor the transition from the initial cooling phase to the reflux stage to guarantee full conversion before proceeding to the quenching step with sodium sulfite. Following the isolation of the intermediate, the condensation reaction demands strict pH control during the workup phase to prevent product degradation. The final crystallization from isopropanol at near-freezing temperatures is the definitive step for achieving pharmaceutical-grade purity, requiring slow cooling rates to promote the growth of uniform crystals. Detailed standardized operating procedures for each of these stages are essential for technology transfer and successful commercial adoption.
- Oxidize 2,4-diamino-6-chloropyrimidine with m-chloroperoxybenzoic acid (mCPBA) in chloroform under reflux to obtain the N-oxide intermediate.
- Condense the intermediate with piperidine in acetone using potassium carbonate as a base at 55-60°C.
- Purify the crude product by crystallization in isopropanol at 0-4°C to obtain high-purity Minoxidil.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this optimized synthesis route offers tangible strategic benefits that extend beyond simple chemical yield. The process relies on commodity chemicals such as chloroform, acetone, and isopropanol, which are readily available in the global market, reducing the risk of raw material shortages. The high efficiency of the reaction sequence means that less starting material is required to produce the same amount of finished API, effectively lowering the material cost per kilogram. Furthermore, the simplicity of the workup procedures, involving standard liquid-liquid extractions and filtrations, reduces the demand for specialized equipment and highly skilled labor, contributing to significant operational expenditure (OpEx) savings. The robustness of the method ensures consistent batch quality, minimizing the financial losses associated with out-of-specification batches and reprocessing.
- Cost Reduction in Manufacturing: The elimination of complex purification columns and the use of recoverable solvents drastically reduce the variable costs associated with production. By achieving a 92% yield on the intermediate and a 75% yield on the final product, the process minimizes waste generation and maximizes the utility of every kilogram of raw material purchased. The avoidance of expensive transition metal catalysts further lowers the input costs and removes the need for costly heavy metal scavenging steps, streamlining the overall cost structure.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous reagents like potassium carbonate and piperidine ensures that the supply chain is resilient to regulatory shifts affecting controlled substances. The short reaction times and straightforward scale-up potential mean that manufacturers can respond rapidly to fluctuations in market demand without lengthy lead times. This agility allows for a more responsive inventory management strategy, ensuring that downstream formulation partners receive their API shipments on schedule.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard 2T reaction kettles and common unit operations that are easily replicated in GMP facilities. The solvent recovery protocols for chloroform and ethyl acetate align with green chemistry principles by reducing volatile organic compound (VOC) emissions. Additionally, the aqueous waste streams generated during the quenching and washing phases are manageable using standard wastewater treatment protocols, facilitating easier environmental compliance and permitting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Minoxidil synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing a clear picture of the operational realities. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for quality assurance teams establishing control strategies. The answers reflect the specific conditions and outcomes observed during the validation of this novel route.
Q: What is the overall yield of this Minoxidil synthesis process?
A: The process achieves an isolated yield of approximately 92% for the key N-oxide intermediate and a final refined product yield of around 75%, which is considered highly efficient for this class of heterocyclic compounds.
Q: Which solvents are utilized in the production workflow?
A: The synthesis utilizes chloroform for the oxidation stage, acetone for the condensation reaction, and isopropanol for the final recrystallization, all of which are standard, recoverable industrial solvents.
Q: How is product purity ensured in this method?
A: High purity is achieved through a controlled low-temperature crystallization step (0-4°C) in isopropanol, effectively removing impurities and by-products to meet stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Minoxidil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of Minoxidil for the global dermatological market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of market fluctuations. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch meets the highest international pharmacopeial standards. Our facility is equipped to handle the specific solvent systems and thermal profiles required by this optimized synthesis route, guaranteeing a product that is both cost-effective and chemically superior.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing process can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this high-yield methodology. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to enhance the efficiency and reliability of your Minoxidil supply chain today.
