Advanced Continuous Flow Synthesis of N,N'-Dialkylalkanediamines for Global Pharmaceutical Supply Chains
The global demand for high-purity diamine derivatives continues to surge across the pharmaceutical and agrochemical sectors, driven by their critical role as building blocks for complex active ingredients. Patent CN1222505C introduces a transformative methodology for the preparation of N,N'-dialkylalkanediamines, addressing long-standing inefficiencies in nucleophilic substitution reactions between dihaloalkanes and lower alkylamines. This innovation shifts the paradigm from traditional batch processing to a precision-controlled continuous flow regime, fundamentally altering the impurity profile of the final product. By rigorously managing the concentration of the transient haloalkaneamine intermediate within the reaction matrix, the process effectively suppresses the thermodynamic drive towards cyclic byproduct formation. For R&D directors and procurement specialists alike, this represents a pivotal advancement in securing reliable pharmaceutical intermediates supplier partnerships that prioritize both molecular integrity and process economics. The following analysis dissects the technical nuances of this patent to demonstrate its viability for large-scale commercial adoption.
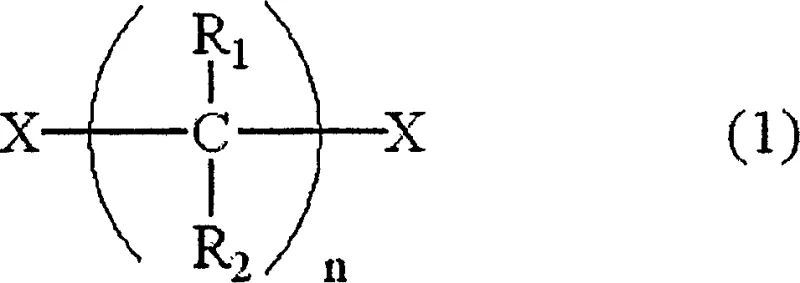
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N'-dialkylalkanediamines via the reaction of dihaloalkanes with alkylamines has been plagued by significant selectivity challenges inherent to batch reactor dynamics. In traditional setups, the simultaneous presence of high concentrations of both reactants often leads to an uncontrollable accumulation of the mono-substituted haloalkaneamine intermediate. This accumulation creates a kinetic environment where the intermediate is statistically more likely to undergo intramolecular cyclization or react with another molecule of the intermediate rather than the intended second substitution with the amine. Consequently, conventional processes typically yield substantial quantities of cyclic dialkyldiamines and trialkylalkanetriamines, often ranging from 1% to 5% and 9% to 13% respectively. These persistent impurities necessitate rigorous and costly downstream purification protocols, including complex distillation columns and extensive solvent washing, which erode profit margins and extend production lead times significantly.
The Novel Approach
The methodology disclosed in CN1222505C circumvents these kinetic pitfalls through a sophisticated manipulation of reactant stoichiometry and addition profiles within a flow chemistry context. Instead of allowing the haloalkaneamine intermediate to accumulate, the process mandates that its residual amount in the reaction system be maintained at an extremely low level, specifically 0.002 mol or less per 1 mol of alkylamine. This is achieved by employing a vast excess of the lower alkylamine, acting simultaneously as both reactant and solvent, while introducing the dihaloalkane continuously or in multiple discrete batches. By ensuring that the concentration of the electrophilic dihaloalkane is always the limiting factor relative to the nucleophilic amine at any given point in the reactor, the reaction pathway is forcibly directed towards the desired linear diamine. This strategic control not only enhances the yield of the target N,N'-dialkylalkanediamine but also drastically simplifies the workup procedure, offering a clear pathway for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Controlled Intermediate Concentration
To fully appreciate the efficacy of this process, one must delve into the competitive reaction mechanisms governing the substitution events. The primary reaction involves the nucleophilic attack of the lower alkylamine on the dihaloalkane to form the haloalkaneamine intermediate, followed by a second attack to form the final diamine. However, parallel side reactions compete fiercely when the intermediate concentration rises. The haloalkaneamine possesses both a nucleophilic amine group and an electrophilic halide group, making it susceptible to self-reaction. When two molecules of the intermediate collide, they can form a trialkylalkanetriamine, or a single molecule can cyclize to form a stable heterocyclic ring. The patent elucidates that the rate of these side reactions is directly proportional to the square of the intermediate concentration. Therefore, by leveraging continuous flow technology to keep the intermediate concentration near zero throughout the reaction volume, the rate of byproduct formation is mathematically suppressed to negligible levels. This mechanistic understanding allows chemists to tune the residence time and feed rates to optimize the ratio of linear to cyclic products dynamically.
Furthermore, the choice of reactor geometry plays a critical role in maintaining this delicate chemical balance. The patent advocates for the use of tubular flow reactors or multistage container type flow reactors, which provide superior heat and mass transfer characteristics compared to stirred tank reactors. In a tubular system, the dihaloalkane can be introduced at multiple injection points along the length of the tube, ensuring that the local concentration never spikes. This spatial distribution of reagents prevents the formation of hot spots where side reactions might accelerate. Additionally, the ability to operate at elevated pressures, typically between 2 MPa and 8 MPa, allows the reaction to proceed at temperatures of 80°C to 140°C without solvent boiling, further enhancing reaction kinetics. This precise control over the physical parameters of the reaction environment ensures that the chemical mechanism proceeds exclusively through the desired linear substitution pathway, resulting in a product stream with exceptional purity specifications suitable for sensitive pharmaceutical applications.
How to Synthesize N,N'-Dialkylalkanediamines Efficiently
Implementing this synthesis route requires a shift from standard batch protocols to a continuous processing mindset, focusing on the real-time monitoring of intermediate levels. The core principle involves establishing a steady state where the consumption of the haloalkaneamine matches its formation rate, preventing any backlog of reactive species. Operators must utilize inline analytical tools, such as gas chromatography, to verify that the molar ratio of the intermediate to the alkylamine remains below the critical threshold of 0.002 throughout the run. The detailed standardized synthesis steps see the guide below for specific operational parameters regarding feed rates and temperature gradients.
- Prepare a reaction system using a tubular or multistage container flow reactor charged with a large excess of lower alkylamine solvent.
- Continuously feed dihaloalkane into the reactor at multiple points or at a controlled rate to maintain the haloalkaneamine intermediate concentration below 0.002 mol per mol of alkylamine.
- Maintain reaction temperature between 80-140°C and pressure at 2-8 MPa, followed by distillation to recover excess amine and isolate the pure diamine product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the transition to this patented continuous flow process offers compelling economic and logistical advantages that extend far beyond simple yield improvements. The primary value proposition lies in the drastic simplification of the purification train. Since the formation of difficult-to-separate cyclic and triamine byproducts is inhibited at the molecular level, the crude product emerging from the reactor is of significantly higher quality. This reduction in impurity load translates directly into reduced energy consumption for distillation and lower solvent usage for washing, driving down the overall variable cost of production. Moreover, the continuous nature of the process allows for a smaller physical footprint compared to large batch vessels, enabling decentralized manufacturing capabilities that enhance supply chain resilience against regional disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts often required in alternative amination routes, combined with the reduced need for complex purification infrastructure, results in substantial operational expenditure savings. By avoiding the formation of tenacious byproducts, the process minimizes the loss of valuable raw materials to waste streams, thereby improving the overall atom economy of the synthesis. This efficiency gain allows manufacturers to offer more competitive pricing structures without compromising on margin, making it an attractive option for high-volume commodity chemical procurement strategies focused on long-term cost stability.
- Enhanced Supply Chain Reliability: Continuous flow systems are inherently more scalable and easier to ramp up than batch processes, as capacity can be increased by running the reactor for longer durations or numbering up identical reactor units rather than building massive new vessels. This modularity ensures that supply can be adjusted rapidly to meet fluctuating market demands for high-purity N,N'-dialkylalkanediamines without the long lead times associated with traditional plant expansion. Furthermore, the robustness of the process against variations in raw material quality ensures consistent output, reducing the risk of batch failures that could interrupt the supply of critical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The closed-loop nature of flow chemistry significantly reduces the emission of volatile organic compounds (VOCs) and minimizes the generation of hazardous waste, aligning with increasingly stringent global environmental regulations. The ability to operate safely at higher pressures and temperatures within a contained system reduces the risk of thermal runaway incidents, enhancing workplace safety. These factors collectively contribute to a more sustainable manufacturing profile, which is becoming a key differentiator for suppliers seeking to partner with environmentally conscious multinational corporations committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. They are derived from the specific constraints and beneficial effects outlined in the patent documentation, providing clarity on how this method outperforms legacy techniques in terms of selectivity and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into existing production lines.
Q: How does controlling the intermediate concentration improve yield in diamine synthesis?
A: By maintaining the haloalkaneamine intermediate concentration below 0.002 mol relative to the alkylamine, the kinetic probability of the intermediate reacting with itself to form cyclic byproducts or triamines is drastically reduced, favoring the formation of the linear N,N'-dialkylalkanediamine.
Q: What reactor types are suitable for this patented process?
A: The patent specifically highlights the superiority of tubular flow reactors and multistage container type flow reactors over traditional batch reactors, as they allow for precise spatial control of reagent addition and intermediate concentration management.
Q: Can this process eliminate the need for complex purification steps?
A: Yes, by suppressing the formation of cyclic dialkyldiamines and trialkylalkanetriamines at the source, the crude product purity is significantly higher, thereby simplifying downstream distillation and reducing the load on purification equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-Dialkylalkanediamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of patent CN1222505C must be translated into tangible commercial reality through expert engineering and rigorous quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of intermediate concentration control is maintained even at industrial scales. Our facilities are equipped with state-of-the-art continuous flow reactors and stringent purity specifications are enforced through our rigorous QC labs, guaranteeing that every shipment of N,N'-dialkylalkanediamine meets the exacting standards required for pharmaceutical and agrochemical synthesis. We bridge the gap between academic innovation and industrial application, delivering consistent quality that supports your downstream drug development timelines.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how the adoption of this flow chemistry approach can reduce your total landed cost. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to validate the superior impurity profile and economic benefits of our manufacturing process before committing to large-scale orders. Let us engineer a supply solution that drives efficiency and reliability for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
