Scalable Manufacturing of Lopinavir Intermediates via Stable Acyl Imidazole Coupling Technology
Scalable Manufacturing of Lopinavir Intermediates via Stable Acyl Imidazole Coupling Technology
The global demand for effective antiretroviral therapies continues to drive innovation in the synthesis of HIV protease inhibitors. Patent CN1264823C introduces a pivotal advancement in the manufacturing of Lopinavir, a critical component in HIV treatment regimens. This technology addresses long-standing challenges in peptide coupling by replacing unstable acid chloride intermediates with robust acyl imidazole derivatives. The core innovation lies in the preparation of ((2S,3S,5S)-2-(2,6-dimethylphenoxyacetyl)-amino-3-hydroxy-5-(2S-(1-tetrahydropyrimid-2-onyl)-3-methyl-butanoyl)amino-1,6-diphenylhexane, commonly known as Lopinavir. By shifting the synthetic strategy, this method significantly enhances the purity profile and operational safety of the production line. The structural complexity of the target molecule requires precise stereochemical control, which is vividly illustrated in the molecular architecture below.
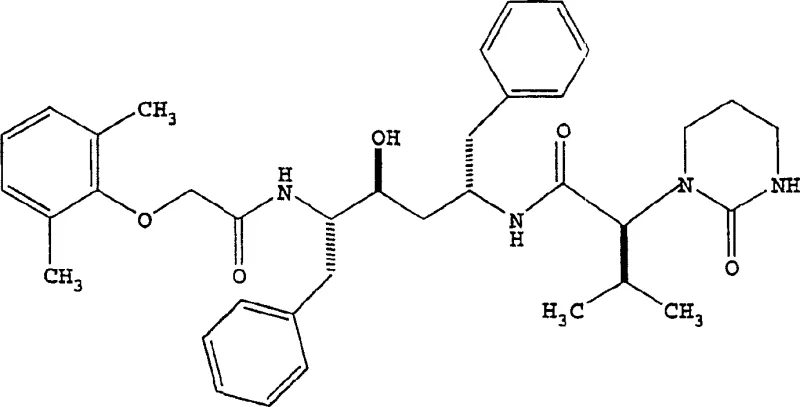
This patent provides a comprehensive roadmap for producing high-purity pharmaceutical intermediates that are essential for downstream API formulation. For R&D directors and process chemists, the shift away from thionyl chloride-based activation represents a major improvement in impurity management. The document details specific reaction conditions, solvent systems, and stoichiometric ratios that optimize yield while minimizing side reactions. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is crucial for ensuring consistent batch-to-batch quality. The following analysis dissects the technical superiority of this approach compared to legacy methods, highlighting its potential for cost reduction in pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lopinavir intermediates relied heavily on the formation of acid chlorides using reagents like thionyl chloride. This conventional pathway, while chemically feasible on a small scale, presents severe drawbacks when translated to industrial production. The primary issue is the inherent instability of the acid chloride intermediate, which complicates storage and handling logistics. Furthermore, the use of thionyl chloride generates byproducts that are notoriously difficult to remove completely. These residual impurities act as catalyst poisons in subsequent reaction steps, leading to incomplete conversions and reduced overall yields. Additionally, the harsh conditions required for acid chloride formation often promote racemization of the sensitive amino acid side chains. This loss of stereochemical integrity is unacceptable in the production of chiral drugs, as it necessitates expensive and time-consuming purification steps to restore enantiomeric excess. Consequently, the conventional method suffers from low atom economy and high waste generation, making it economically and environmentally unsustainable for modern green chemistry standards.
The Novel Approach
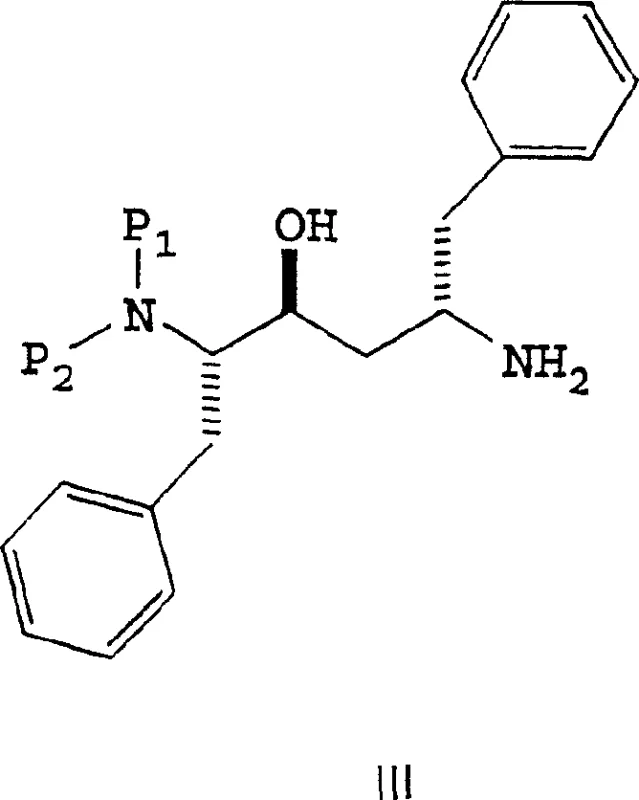
The methodology disclosed in CN1264823C circumvents these issues by utilizing an activated ester strategy, specifically employing N-acyl imidazoles (Formula V). This approach involves reacting the carboxylic acid precursor with carbonyldiimidazole (CDI) to form a stable activated species that retains its integrity under reaction conditions. Unlike acid chlorides, these acyl imidazoles do not require cryogenic storage and are less prone to hydrolysis, simplifying the supply chain for raw materials. The coupling reaction with the amine core (Formula III) proceeds efficiently in common organic solvents like ethyl acetate, eliminating the need for exotic or hazardous reagents. A striking feature of this novel route is the beneficial effect of water; the patent reveals that adding 1% to 3% water accelerates the reaction kinetics, a counter-intuitive finding that allows for faster cycle times. This one-pot capability, where the activated ester is generated and consumed without isolation, drastically reduces processing time and solvent consumption. The structural representation of this key activated intermediate highlights the stability imparted by the imidazole ring.
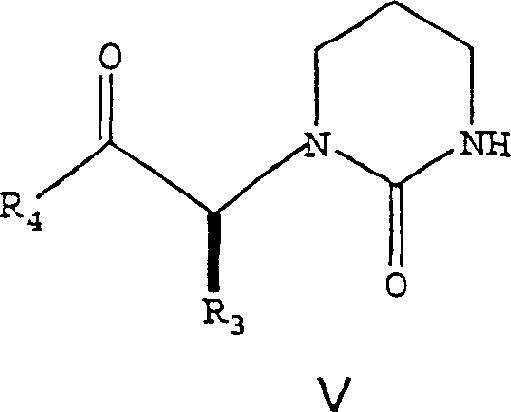
Mechanistic Insights into Acyl Imidazole-Mediated Peptide Coupling
The core chemical transformation in this process is the nucleophilic attack of the amine nitrogen in Formula III upon the carbonyl carbon of the activated ester in Formula V. This mechanism is fundamentally cleaner than acid chloride coupling because the leaving group, imidazole, is a solid that is easily removed during aqueous workup. The reaction preserves the critical (2S, 3S, 5S) stereochemistry of the hexane backbone, which is vital for the biological activity of the final HIV inhibitor. The patent specifies a molar ratio of approximately 1.0 mol of amine to 1.15-1.3 mol of activated ester, ensuring complete consumption of the valuable amine core without excessive waste of the coupling agent. The reaction temperature is maintained between 75°C and 80°C, which corresponds to the reflux temperature of ethyl acetate. This thermal energy is sufficient to overcome the activation barrier for amide bond formation without inducing thermal degradation or epimerization. The presence of water likely facilitates proton transfer steps in the transition state, enhancing the nucleophilicity of the amine. This mechanistic understanding allows process engineers to fine-tune reaction parameters for maximum efficiency.
Impurity control is another critical aspect of this mechanism. In traditional routes, racemization often occurs via the formation of oxazolone intermediates or through base-catalyzed enolization. The mild basicity of the imidazole byproduct and the neutral conditions of the coupling reaction minimize these risks. The resulting intermediate, Formula II, features benzyl protecting groups (P1 and P2) that shield the amine functionalities during the coupling event. These protecting groups are subsequently removed via catalytic hydrogenation, a standard and scalable unit operation. The robustness of this protection-deprotection strategy ensures that the final product meets stringent purity specifications required by regulatory bodies. By avoiding the formation of sulfonate esters or other hard-to-remove byproducts associated with thionyl chloride, the impurity profile of the crude product is significantly cleaner. This reduces the burden on downstream purification units such as crystallization or chromatography, directly impacting the cost of goods sold.
How to Synthesize Lopinavir Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for producing the target intermediate with high fidelity. The process begins with the activation of the carboxylic acid using CDI in ethyl acetate, followed by the addition of the amine core under reflux conditions. The inclusion of a controlled amount of water is a critical process parameter that must be strictly monitored to achieve the accelerated reaction rates described. Following the coupling, the reaction mixture undergoes a series of aqueous washes to remove imidazole and unreacted starting materials. The organic layer is then concentrated and subjected to debenzylation using palladium on carbon and ammonium formate, yielding the free amine salt. This sequence is designed to be telescoped where possible, minimizing the number of isolation steps and maximizing throughput. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis protocol below.
- Prepare the activated ester (Formula V) by reacting the carboxylic acid (Formula IV) with carbonyldiimidazole in ethyl acetate at 15-50°C.
- Combine the activated ester solution with the amine core (Formula III) in a refluxing solvent system containing 1-3% water to accelerate the reaction.
- Perform aqueous workup and distillation to isolate the protected intermediate (Formula II), followed by debenzylation to yield the final active pharmaceutical ingredient precursor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost stability and operational reliability. The elimination of thionyl chloride removes a hazardous material from the supply chain, reducing regulatory compliance costs and safety training requirements. Furthermore, the stability of the acyl imidazole intermediate allows for more flexible production scheduling, as the activated species does not degrade rapidly like acid chlorides. This flexibility translates to better inventory management and reduced risk of batch failures due to reagent decomposition. The use of commodity solvents like ethyl acetate and methanol ensures that raw material costs remain low and predictable, shielding the project from volatility in specialty chemical markets. The process is also inherently safer, operating at atmospheric pressure and moderate temperatures, which lowers insurance premiums and capital expenditure on specialized high-pressure equipment.
- Cost Reduction in Manufacturing: The one-pot nature of the coupling reaction eliminates the need to isolate and purify the unstable acid chloride intermediate. This consolidation of steps reduces labor hours, solvent usage, and energy consumption associated with multiple distillations and drying cycles. Additionally, the avoidance of catalyst-poisoning impurities means that expensive catalysts in downstream steps have longer lifespans and higher turnover numbers. The overall yield improvement resulting from reduced racemization means more product is obtained per kilogram of starting material, directly lowering the variable cost per unit. These efficiencies compound to deliver substantial cost savings over the lifecycle of the product without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reagents required for this process, such as carbonyldiimidazole and ethyl acetate, are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions allows for manufacturing in diverse geographic locations, enabling a distributed supply chain strategy that mitigates regional disruptions. The scalability demonstrated in the patent, utilizing reactors up to 10,000 liters, proves that the chemistry holds up under commercial loads. This ensures that supply can be ramped up quickly to meet surges in demand for antiretroviral therapies, providing a secure source of high-purity pharmaceutical intermediates for our partners.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to the thionyl chloride route, as it avoids the production of sulfur dioxide and hydrochloric acid gases. The aqueous waste streams are easier to treat, lowering effluent disposal costs and simplifying environmental permitting. The ability to run the reaction at reflux in a common solvent simplifies solvent recovery and recycling loops, further enhancing the green chemistry profile of the manufacture. This alignment with sustainability goals is increasingly important for pharmaceutical companies aiming to reduce their carbon footprint. The commercial scale-up of complex pharmaceutical intermediates is thus achieved with a smaller environmental impact, future-proofing the supply chain against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. They are derived from the specific experimental data and comparative examples provided in the patent documentation. Understanding these details helps in assessing the feasibility of technology transfer and process validation. Our technical team has analyzed the critical process parameters to ensure that potential risks are identified and mitigated early in the development phase.
Q: Why is the acyl imidazole method superior to acid chloride activation for Lopinavir synthesis?
A: Traditional acid chloride methods using thionyl chloride generate impurities that poison catalysts and cause racemization. The acyl imidazole route (Formula V) avoids unstable acid chlorides, ensuring higher stereochemical purity and eliminating the need for complex catalyst removal steps.
Q: What role does water play in this specific coupling reaction?
A: Contrary to typical peptide couplings where water is detrimental, this patent specifies that the presence of 1% to 3% water (weight/volume) actually accelerates the reaction rate between Formula III and Formula V, allowing for milder conditions and faster throughput.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly demonstrates scalability using stainless steel and glass-lined reactors ranging from 1,000L to 10,000L. The use of common solvents like ethyl acetate and stable intermediates makes it highly viable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lopinavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the production of life-saving medications. Our CDMO capabilities are perfectly aligned with the advanced chemistry described in CN1264823C, allowing us to deliver high-quality intermediates with speed and precision. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met regardless of market volume. Our facilities are equipped with state-of-the-art reaction vessels and rigorous QC labs capable of verifying stringent purity specifications for every batch. We understand that consistency is key in the pharmaceutical industry, and our quality management systems are designed to guarantee that every gram of Lopinavir intermediate meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain for HIV protease inhibitors. Our team can provide a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this acyl imidazole-based process. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. By leveraging our expertise in peptide coupling and scale-up engineering, we can help you reduce lead time for high-purity pharmaceutical intermediates and accelerate your time to market. Let us be your partner in turning complex chemical innovations into commercial reality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
