Advanced Catalytic Hydrogenation Route for High-Purity 5-Aminobenzimidazole Manufacturing
Introduction to Advanced Pigment Intermediate Synthesis
The global demand for high-performance organic pigments, particularly benzimidazolone-based varieties, necessitates robust and environmentally compliant supply chains for their key precursors. Patent CN102002002A introduces a refined preparation method for 5-aminobenzimidazole, a critical building block in the synthesis of advanced azo pigments used extensively in plastics, printing inks, and coating formulations. This technology represents a significant departure from traditional reduction methodologies, addressing long-standing issues regarding waste generation and product purity that have plagued the fine chemical industry. By shifting towards a catalytic hydrogenation framework, the process ensures complete reaction conversion with minimal side products, directly aligning with the stringent quality requirements of modern pigment manufacturers who demand consistent color strength and fastness properties.
For R&D directors and technical procurement specialists, understanding the nuances of this synthetic pathway is vital for securing a reliable pigment intermediate supplier capable of meeting future regulatory standards. The patent outlines a four-stage sequence that optimizes yield while drastically reducing the environmental footprint associated with legacy manufacturing techniques. This report analyzes the technical merits of this approach, highlighting how the integration of selective chemical reductions and high-pressure catalysis creates a superior value proposition for downstream users in the specialty chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 5-aminobenzimidazole has relied heavily on iron powder reduction technologies or hydrazine hydrate methods, both of which present substantial operational and environmental drawbacks. The iron powder reduction process, while technically mature and utilizing inexpensive reagents, generates massive quantities of iron mud sludge as a byproduct. This solid waste not only complicates the filtration and isolation of the target amine but also imposes severe burdens on wastewater treatment facilities, leading to increased operational expenditures and environmental compliance risks. Furthermore, the presence of residual iron species can detrimentally affect the quality of the final pigment, potentially catalyzing degradation or altering hue stability in end-use applications.
Alternatively, hydrazine hydrate reduction offers milder conditions and avoids solid waste, yet it is generally restricted to small-scale production due to safety concerns and cost inefficiencies associated with handling large volumes of hydrazine. These conventional pathways often struggle to balance economic viability with the high-purity specifications required for premium pigment grades. Consequently, manufacturers face a dichotomy where they must choose between a dirty, low-cost process or a clean, high-cost process that lacks scalability, creating a bottleneck in the supply chain for high-quality dye intermediates.
The Novel Approach
The methodology disclosed in CN102002002A circumvents these limitations by employing a catalytic hydrogenation strategy for the final reduction step, coupled with a selective sulfide reduction earlier in the sequence. This novel approach utilizes Raney Nickel as a heterogeneous catalyst under controlled hydrogen pressure, facilitating a clean conversion of the nitro group to the amine without generating inorganic sludge. The process is designed to be exhaustive, ensuring that the reaction proceeds to completion with negligible formation of hydroxylamine intermediates or other reduction byproducts that typically complicate purification. By eliminating the iron mud issue entirely, the workflow simplifies downstream processing, allowing for more efficient crystallization and drying operations that result in a product with superior physical characteristics.
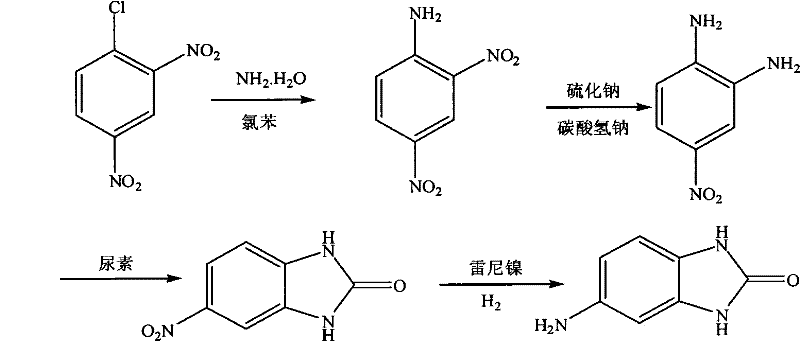
Furthermore, the integration of this catalytic step into a multi-stage synthesis involving urea cyclization demonstrates a holistic optimization of the entire value chain. The transition from 4-nitro-1,2-phenylenediamine to 5-nitrobenzimidazolone via thermal condensation is meticulously controlled to maximize ring closure efficiency before the final hydrogenation. This strategic sequencing ensures that the substrate entering the hydrogenation reactor is of high integrity, thereby protecting the expensive catalyst from poisoning and extending its operational lifecycle. For procurement managers, this translates to a more stable and predictable manufacturing process that mitigates the risk of batch failures and supply interruptions common in older, less controlled synthetic routes.
Mechanistic Insights into Catalytic Hydrogenation and Urea Cyclization
The core of this technological advancement lies in the precise execution of the urea cyclization and the subsequent catalytic hydrogenation. In the cyclization step, 4-nitro-1,2-phenylenediamine reacts with urea through a nucleophilic attack followed by dehydration. The patent specifies a gradient temperature profile, initiating the reaction at 140-150°C to facilitate initial mixing and activation, then ramping to 165-170°C for ring closure, and finally holding at 200-205°C to drive the equilibrium towards the formation of the benzimidazolone ring. This thermal staging is critical for preventing the sublimation of urea and ensuring that the cyclic urea structure forms with high regioselectivity, minimizing the presence of open-chain urea derivatives that could act as impurities in the final pigment application.
Following cyclization, the reduction of the nitro group in 5-nitrobenzimidazolone is achieved using Raney Nickel in a methanol solvent system under hydrogen pressures ranging from 1.5 to 2.0 MPa. Mechanistically, the nitro group adsorbs onto the active sites of the nickel catalyst surface, where it undergoes sequential hydrogenation through nitroso and hydroxylamine intermediates before arriving at the amine state. The use of methanol as a solvent is particularly advantageous as it provides excellent solubility for the intermediate while remaining inert under the reaction conditions. The high pressure ensures a sufficient concentration of dissolved hydrogen at the catalyst interface, driving the kinetics forward and preventing the accumulation of partially reduced species that could lead to azo-coupling side reactions.
Impurity control is inherently built into this mechanism through the selectivity of the Raney Nickel catalyst. Unlike non-selective reducing agents, the catalytic surface preferentially reduces the nitro functionality while leaving the sensitive benzimidazolone ring structure intact. This chemoselectivity is paramount for maintaining the structural integrity of the heterocyclic core, which is essential for the hydrogen-bonding capabilities that give benzimidazolone pigments their renowned heat and solvent resistance. By strictly controlling the reaction temperature around 100°C and monitoring hydrogen uptake, the process avoids over-reduction or ring-opening scenarios, resulting in a crude product that requires minimal purification to meet commercial specifications.
How to Synthesize 5-Aminobenzimidazole Efficiently
The synthesis of this high-value intermediate requires strict adherence to the patented parameters to ensure reproducibility and safety at scale. The process begins with the amination of 2,4-dinitrochlorobenzene, followed by selective reduction, cyclization, and final hydrogenation. Each step demands precise control over stoichiometry, temperature, and pressure to maintain the high yield and purity profiles described in the technical literature. Operators must be trained to handle high-pressure autoclaves and manage exothermic reactions safely, particularly during the addition of reducing agents and the hydrogenation phase.
- Synthesize 2,4-dinitroaniline by reacting 2,4-dinitrochlorobenzene with aqueous ammonia at 145-150°C under pressure.
- Perform selective reduction using sodium sulfide and sodium bicarbonate to obtain 4-nitro-1,2-phenylenediamine.
- Condense the diamine with urea at high temperatures (up to 205°C) to form 5-nitrobenzimidazolone.
- Execute final reduction using Raney Nickel catalyst under hydrogen pressure (1.5-2.0 MPa) to yield 5-aminobenzimidazole.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement officers, the adoption of this catalytic hydrogenation route offers compelling economic and logistical benefits that extend beyond simple unit cost calculations. The primary advantage lies in the drastic simplification of waste management protocols. By eliminating the generation of iron mud, manufacturers avoid the substantial costs associated with sludge dewatering, transport, and hazardous waste disposal. This reduction in environmental liability not only lowers direct operational expenses but also insulates the supply chain from regulatory shocks related to waste discharge limits, ensuring continuous operation even as environmental regulations tighten globally.
- Cost Reduction in Manufacturing: The elimination of iron powder and the associated acid digestion steps removes a significant variable cost component from the bill of materials. Furthermore, the recovery and potential regeneration of the Raney Nickel catalyst offer a pathway to further optimize catalyst consumption costs over time. The simplified work-up procedure, which involves filtration of the catalyst followed by solvent recovery, reduces utility consumption related to water washing and drying, leading to substantial overall cost savings in the production of fine chemical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the catalytic process contributes to higher batch success rates and more consistent cycle times. Unlike iron powder reductions which can suffer from variability due to the quality of the iron source, the use of standardized catalysts and gaseous hydrogen provides a more controllable reaction environment. This consistency allows for tighter production scheduling and more reliable delivery commitments to downstream pigment formulators, reducing the need for safety stock and improving overall inventory turnover for buyers seeking a reliable pigment intermediate supplier.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scalability, utilizing standard high-pressure reactor equipment that is readily available in modern chemical plants. The absence of heavy metal sludge simplifies the permitting process for capacity expansion, allowing suppliers to ramp up production volumes rapidly in response to market demand. Additionally, the cleaner effluent profile facilitates easier wastewater treatment, ensuring that the manufacturing site remains compliant with increasingly strict global environmental standards without requiring massive capital investment in end-of-pipe treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5-aminobenzimidazole synthesized via this advanced route. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and technical evaluators.
Q: Why is catalytic hydrogenation preferred over iron powder reduction for this intermediate?
A: Traditional iron powder reduction generates significant amounts of iron mud sludge, creating severe environmental disposal challenges and complicating product purification. The catalytic hydrogenation method described in CN102002002A eliminates solid waste byproducts, ensuring a cleaner process and higher product quality suitable for sensitive pigment applications.
Q: What are the critical reaction conditions for the urea cyclization step?
A: The cyclization of 4-nitro-1,2-phenylenediamine with urea requires a staged temperature profile, starting at 140-150°C and ramping up to 200-205°C. This precise thermal control is essential to drive the dehydration condensation to completion while minimizing side reactions that could affect the color strength of the final pigment.
Q: Is this process scalable for industrial production of pigment intermediates?
A: Yes, the patent explicitly designs the methodology for industrial suitability. The use of standard autoclaves for the amination and hydrogenation steps, combined with the elimination of difficult-to-filter iron sludge, makes the process highly scalable for commercial manufacturing of dye intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Aminobenzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the performance of final pigment products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic hydrogenation techniques described in CN102002002A can be effectively translated into large-scale manufacturing realities. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-aminobenzimidazole meets the exacting standards required for high-performance benzimidazolone pigments, providing our clients with the confidence needed for long-term product development.
We invite procurement directors and R&D leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis capabilities, we can help you reduce lead time for high-purity dye intermediates while securing a sustainable supply chain. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to green chemistry and operational excellence can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
