Advanced High-Pressure Mannich Base Synthesis for Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes for key intermediates, particularly those leading to neurologically active compounds like duloxetine. Patent CN1891683A discloses a groundbreaking process for the preparation of N-monosubstituted beta-amino alcohols of general formula (I) and their addition salts. This technology represents a significant leap forward by enabling the direct preparation of N-monosubstituted beta-ketoamines of formula (II), which serve as the critical precursors. Unlike traditional methods that struggle with selectivity, this invention utilizes a high-pressure Mannich-type condensation to bypass the formation of unwanted tertiary amine byproducts. The ability to directly synthesize these secondary amine intermediates with high yield provides a reliable foundation for producing high-purity pharmaceutical intermediates used in the treatment of depression and chronic pain.

Furthermore, the versatility of this synthetic approach allows for a broad range of substituents, including thiophenyl and furyl groups, which are essential scaffolds in modern medicinal chemistry. The process described in the patent not only improves chemical efficiency but also addresses long-standing economic and logistical challenges in the supply chain. By utilizing readily available starting materials such as methyl ketones and primary amine salts, manufacturers can achieve substantial cost savings while maintaining stringent quality standards. This report analyzes the technical merits of this high-pressure synthesis route and its implications for global procurement strategies in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-monosubstituted beta-ketoamines has been plagued by poor selectivity and low yields due to the inherent reactivity of primary amines in Mannich reactions. As illustrated in the background art, classical conditions typically lead to the predominant formation of tertiary beta-ketoamines of general formula (III) rather than the desired secondary amines. This side reaction occurs because the initially formed secondary amine is often more nucleophilic than the starting primary amine, leading to over-alkylation. Consequently, conventional atmospheric pressure methods often result in yields below 50%, with significant loss of valuable starting materials to these unwanted byproducts.
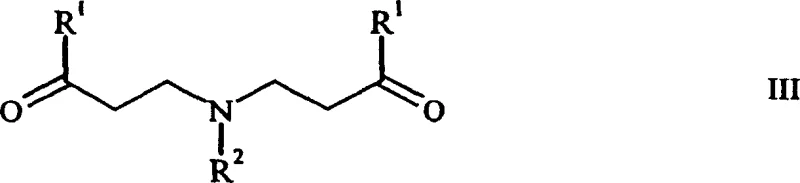
Moreover, separating the desired secondary amine from the tertiary amine byproducts is chemically challenging and economically inefficient, often requiring complex purification steps that further erode overall process yield. Previous attempts to mitigate this issue, such as using N-methylene oxalamides as nitrogen sources, introduced new complexities including the need for additional reducing equivalents and difficult-to-source reagents. These limitations have historically constrained the commercial viability of producing N-monosubstituted beta-amino alcohols on a large scale, creating bottlenecks for the manufacturing of active pharmaceutical ingredients that rely on this structural motif.
The Novel Approach
The novel approach disclosed in patent CN1891683A fundamentally alters the reaction kinetics by conducting the Mannich condensation under elevated pressure, specifically greater than 1.5 bar. This modification effectively suppresses the formation of the tertiary amine byproducts, allowing for the direct and selective synthesis of the N-monosubstituted beta-ketoamine of formula (II). By shifting the equilibrium and kinetic profile through pressure control, the process achieves yields ranging from 50% to 75%, a marked improvement over the 30% to 45% typical of atmospheric reflux methods. This breakthrough eliminates the need for complex protecting group strategies or exotic nitrogen sources, streamlining the synthetic pathway significantly.
In addition to pressure control, the method optimizes the use of solvents and acid additives to stabilize the product as a protonic acid salt, which prevents decomposition during storage and isolation. The use of commercially available methyl ketones and primary amine hydrochlorides simplifies the raw material sourcing, making the process highly attractive for industrial application. This direct route not only enhances the purity of the intermediate but also reduces the environmental footprint by minimizing waste generation associated with purification and byproduct disposal, aligning with modern green chemistry principles.
Mechanistic Insights into High-Pressure Mannich Condensation
The core mechanistic advantage of this process lies in the manipulation of reaction kinetics through autogenous pressure generated in a closed system. Under standard atmospheric conditions, the volatility of reactants like formaldehyde and methylamine can lead to concentration gradients that favor sequential alkylation. However, maintaining a system pressure between 1.5 and 10 bar ensures that the concentration of reactive species remains high and consistent throughout the reaction vessel. This environment favors the initial nucleophilic attack of the primary amine on the iminium ion intermediate while sterically or kinetically hindering the second alkylation step that leads to the tertiary amine.
Furthermore, the choice of solvent plays a crucial role in stabilizing the transition states and the final product. The patent highlights the effectiveness of aliphatic alcohols such as ethanol and isopropanol, which act not only as solvents but also as proton donors that facilitate the formation of stable hydrochloride salts of the product. The presence of a protonic acid, such as hydrochloric acid or acetic acid, further drives the equilibrium towards the salt form, which precipitates out of the solution upon cooling or solvent removal. This in-situ salt formation acts as a driving force for the reaction, pulling the equilibrium towards the desired product and preventing retro-Mannich degradation.
How to Synthesize N-Monosubstituted Beta-Ketoamines Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for translating this laboratory innovation into a scalable industrial process. The procedure involves charging an autoclave with precise stoichiometric ratios of methyl ketone, primary amine hydrochloride, and paraformaldehyde in a suitable alcoholic solvent. The reaction mixture is then heated to temperatures between 100°C and 120°C for a duration of 5 to 24 hours, ensuring that the internal pressure exceeds the critical threshold of 1.5 bar. Following the reaction, the solvent is partially or fully removed under vacuum, and the product is induced to crystallize by adding a non-polar anti-solvent like ethyl acetate.
- Charge an autoclave with methyl ketone, primary amine hydrochloride, paraformaldehyde, and a solvent such as ethanol or isopropanol.
- Heat the reaction mixture to temperatures between 100°C and 120°C while maintaining a total system pressure greater than 1.5 bar for 5 to 24 hours.
- Cool the reaction, remove solvent under vacuum, and induce crystallization using a non-polar solvent like ethyl acetate to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this high-pressure synthesis route offers compelling strategic advantages centered around cost efficiency and supply security. The elimination of expensive and hard-to-source reagents, such as N-methylene oxalamides, drastically simplifies the bill of materials. Instead, the process relies on commodity chemicals like methylamine hydrochloride and paraformaldehyde, which are available globally in bulk quantities. This shift from specialty reagents to commodity feedstocks significantly reduces the risk of supply disruption and mitigates price volatility associated with niche chemical markets.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this manufacturing process is the substantial increase in reaction yield and the simplification of downstream processing. By avoiding the formation of tertiary amine byproducts, the need for resource-intensive purification steps such as column chromatography or multiple recrystallizations is minimized. Furthermore, the direct use of amine hydrochlorides eliminates the cost and safety hazards associated with handling volatile free amines, leading to lower operational expenditures and reduced waste disposal costs.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures a robust and resilient supply chain. Methyl ketones and formaldehyde sources are produced by major chemical manufacturers worldwide, reducing dependency on single-source suppliers for critical intermediates. Additionally, the stability of the product as a hydrochloride salt allows for easier storage and transportation, extending shelf life and providing greater flexibility in inventory management for both the manufacturer and the end-user.
- Scalability and Environmental Compliance: The process is inherently scalable as it utilizes standard high-pressure reactors (autoclaves) that are common infrastructure in fine chemical plants. The use of alcohol solvents and the absence of heavy metal catalysts simplify wastewater treatment and solvent recovery, ensuring compliance with increasingly stringent environmental regulations. This ease of scale-up facilitates the rapid transition from pilot batches to multi-ton commercial production, supporting the growing demand for these pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-pressure Mannich synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN1891683A, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these details is crucial for R&D teams planning process validation and procurement teams assessing vendor capabilities.
Q: Why is high pressure critical in this Mannich reaction?
A: Operating at pressures greater than 1.5 bar kinetically favors the formation of the desired N-monosubstituted beta-ketoamine over the thermodynamically stable tertiary amine byproducts, significantly improving yield.
Q: What are the advantages of using amine hydrochlorides over free amines?
A: Amine hydrochlorides are solids that are easier to handle, store, and dose accurately compared to volatile free amines, enhancing operational safety and supply chain stability.
Q: Can this process be scaled for commercial API production?
A: Yes, the process utilizes standard industrial autoclaves and common solvents like ethanol, making it highly scalable from pilot plant to multi-ton commercial manufacturing without specialized catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Monosubstituted Beta-Amino Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of pharmaceutical manufacturing. Our team of expert chemists has extensively evaluated the high-pressure Mannich condensation technology described in CN1891683A and possesses the technical capability to implement this process at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel chemistry are realized in terms of both purity and throughput. Our rigorous QC labs and stringent purity specifications guarantee that every batch of N-monosubstituted beta-amino alcohol meets the exacting standards required for API synthesis.
We invite you to collaborate with us to optimize your supply chain for these vital intermediates. By leveraging our expertise in high-pressure synthesis and process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical partner available in the market today.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
