Revolutionizing Pharmaceutical Intermediate Production with High-Pressure Mannich Chemistry
The pharmaceutical industry constantly seeks robust synthetic routes for critical building blocks, particularly those serving as precursors for neuroactive compounds. Patent CN1665773A introduces a transformative methodology for the preparation of N-monosubstituted beta-amino alcohols and their corresponding protic acid addition salts. This technology addresses long-standing inefficiencies in the synthesis of these vital intermediates by utilizing a direct, high-pressure Mannich-type reaction. Unlike traditional approaches that struggle with selectivity, this novel process enables the direct formation of N-monosubstituted beta-ketoamines, which serve as stable and versatile precursors. The ability to synthesize these compounds with high yield and purity represents a significant leap forward for manufacturers aiming to produce active pharmaceutical ingredients (APIs) such as duloxetine. By shifting the reaction equilibrium through controlled pressure conditions, the method effectively bypasses the formation of unwanted tertiary amine by-products that have plagued chemists for decades.
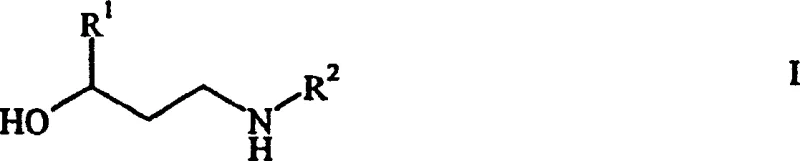
For R&D directors evaluating process viability, understanding the mechanistic shift is crucial. The core innovation lies in the stabilization of the intermediate species under specific thermodynamic conditions. The general structure of the target N-monosubstituted beta-amino alcohols, as depicted in the patent, allows for substantial structural diversity where R1 and R2 can be independently selected from alkyl, cycloalkyl, aryl, or aralkyl groups. This flexibility is paramount for generating libraries of analogues or optimizing the physicochemical properties of the final drug substance. Furthermore, the process accommodates various substituents including halogens and alkoxy groups, ensuring broad applicability across different therapeutic classes. The direct access to these structures without requiring elaborate protection-deprotection sequences streamlines the overall synthetic timeline, reducing the potential for impurity accumulation during multi-step transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-monosubstituted beta-ketoamines has been fraught with significant chemical challenges that impact both yield and operational cost. Conventional Mannich reactions conducted at atmospheric pressure using primary alkylamines invariably favor the formation of tertiary beta-ketoamines over the desired mono-substituted products. Literature from as early as 1922 indicates that attempting to isolate the desired secondary amine often results in a complex mixture requiring rigorous separation techniques. Previous attempts to mitigate this issue involved steam distillation of the tertiary amine hydrochlorides, a process that notoriously leads to the loss of more than 50 percent of the starting material due to decomposition into vinyl compounds and other by-products. Additionally, alternative methods relying on N-methylammonium oxalate as a nitrogen source introduce unnecessary complexity, as these salts are not commercially available and require additional synthesis and purification steps. These inefficiencies create a bottleneck in the supply chain, driving up the cost of goods and limiting the scalability of the final pharmaceutical product.
The Novel Approach
The methodology disclosed in CN1665773A fundamentally alters the reaction landscape by introducing elevated pressure as a critical control parameter. By conducting the reaction between methyl ketones, primary amines, and formaldehyde sources at pressures greater than 1.5 bar, specifically within the range of 1.5 to 5 bar, the process selectively drives the formation of the N-monosubstituted beta-ketoamine intermediate.  This intermediate, represented by Formula II in the patent, is obtained in high yields ranging approximately from 50 percent to 75 percent, a marked improvement over the 30 percent to 45 percent yields typical of atmospheric reflux methods. The use of readily available and inexpensive reagents, such as methylamine hydrochloride, further enhances the economic viability of this route. Because the reaction proceeds efficiently in common solvents like ethanol or isopropanol, the need for exotic or hazardous reagents is eliminated. This approach not only simplifies the work-up procedure but also ensures that the resulting intermediate is stable enough for storage and subsequent reduction without immediate degradation.
This intermediate, represented by Formula II in the patent, is obtained in high yields ranging approximately from 50 percent to 75 percent, a marked improvement over the 30 percent to 45 percent yields typical of atmospheric reflux methods. The use of readily available and inexpensive reagents, such as methylamine hydrochloride, further enhances the economic viability of this route. Because the reaction proceeds efficiently in common solvents like ethanol or isopropanol, the need for exotic or hazardous reagents is eliminated. This approach not only simplifies the work-up procedure but also ensures that the resulting intermediate is stable enough for storage and subsequent reduction without immediate degradation.
Mechanistic Insights into Pressurized Mannich Condensation
The success of this synthesis relies on the subtle manipulation of reaction kinetics and thermodynamics afforded by the pressurized environment. In a standard atmospheric Mannich reaction, the nucleophilic attack of the primary amine on the iminium ion intermediate is often followed by a second alkylation event, leading to the thermodynamically more stable tertiary amine. However, under the specific pressure conditions defined in this patent, the equilibrium is shifted to favor the mono-alkylated species. The presence of a protic acid, such as hydrochloric acid, facilitates the formation of the iminium ion while simultaneously protonating the amine product to form a stable salt. This protonation effectively deactivates the nitrogen lone pair, preventing further alkylation to the tertiary amine. The result is a clean conversion to the beta-ketoamine hydrochloride, which can be isolated as a solid powder. This mechanistic control is essential for maintaining high purity levels, as it minimizes the generation of closely related impurities that are difficult to remove in later stages of API synthesis.
Furthermore, the stability of the intermediate salt form offers distinct advantages for impurity control and supply chain management. Unlike the free base forms of beta-ketoamines, which are prone to decomposition upon distillation or prolonged storage, the acid addition salts exhibit remarkable stability. This characteristic allows manufacturers to produce the intermediate in large batches and store it for extended periods without significant loss of quality. For quality assurance teams, this means that the impurity profile remains consistent over time, reducing the risk of batch-to-batch variability. The ability to isolate the intermediate as a crystalline solid also facilitates rigorous quality control testing before proceeding to the reduction step. By ensuring the purity of the beta-ketoamine precursor, the final reduction to the beta-amino alcohol proceeds with higher fidelity, resulting in a final product that meets stringent pharmaceutical specifications with minimal downstream purification requirements.
How to Synthesize N-Monosubstituted Beta-Amino Alcohol Efficiently
The practical implementation of this synthesis involves a straightforward two-step sequence that is highly amenable to industrial scale-up. The first step entails the condensation of a methyl ketone with a primary amine salt and paraformaldehyde in an alcoholic solvent within a pressurized reactor. Following the reaction, the solvent is partially removed to induce precipitation of the beta-ketoamine salt, which is then filtered and dried. The second step involves the reduction of the carbonyl group of this isolated intermediate to yield the final beta-amino alcohol. Detailed standardized synthesis steps see the guide below.
- React commercially available methyl ketones with primary amines (or their salts) and formaldehyde sources in a solvent under pressure greater than 1.5 bar.
- Isolate the resulting N-monosubstituted beta-ketoamine intermediate, preferably as a stable protic acid addition salt.
- Reduce the carbonyl group of the beta-ketoamine intermediate using standard reducing agents to obtain the final N-monosubstituted beta-amino alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this pressurized synthesis route offers compelling economic and logistical benefits. The primary advantage stems from the drastic simplification of the raw material portfolio and the elimination of inefficient processing steps. By utilizing commodity chemicals like methylamine hydrochloride and paraformaldehyde, manufacturers can secure a stable and cost-effective supply of inputs that are not subject to the volatility associated with specialized reagents. The avoidance of steam distillation, which traditionally resulted in massive material losses, translates directly into improved atom economy and reduced waste disposal costs. This efficiency gain is critical for maintaining competitive pricing in the global market for pharmaceutical intermediates, where margin compression is a constant challenge.
- Cost Reduction in Manufacturing: The transition to this high-yield process eliminates the need for expensive and hard-to-source reagents like N-methylammonium oxalate, which previously required custom synthesis. Furthermore, the ability to achieve yields exceeding 70 percent compared to the sub-45 percent yields of legacy methods means that significantly less raw material is required to produce the same amount of product. This reduction in material consumption lowers the variable cost per kilogram substantially. Additionally, the simplified work-up procedure, which relies on simple filtration rather than complex distillation or chromatography, reduces energy consumption and labor hours. These cumulative efficiencies contribute to a leaner manufacturing operation that is better positioned to absorb fluctuations in raw material pricing while maintaining profitability.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route significantly mitigates supply chain risks associated with production delays and batch failures. Because the intermediate beta-ketoamine salts are stable and can be stockpiled, production planning becomes more flexible, allowing manufacturers to decouple the synthesis of the intermediate from the final reduction step. This buffer capacity ensures continuity of supply even if there are temporary disruptions in the downstream processing units. Moreover, the use of standard autoclave equipment means that the process can be easily transferred between different manufacturing sites or contracted manufacturing organizations (CMOs) without requiring specialized infrastructure. This flexibility enhances the resilience of the supply network, ensuring that critical intermediates for drugs like duloxetine remain available to meet global demand.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the use of well-understood unit operations and benign solvents. The reaction conditions, involving moderate temperatures and pressures, are well within the safety limits of standard industrial reactors, reducing the capital expenditure required for plant upgrades. From an environmental standpoint, the higher selectivity of the reaction results in fewer by-products and less chemical waste, aligning with increasingly stringent regulatory requirements for green chemistry. The elimination of steam distillation also reduces the volume of aqueous waste streams that require treatment. These factors collectively lower the environmental footprint of the manufacturing process, facilitating smoother regulatory approvals and enhancing the sustainability profile of the final pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making.
Q: Why is the pressurized Mannich reaction superior to conventional atmospheric methods?
A: Conventional atmospheric Mannich reactions with primary amines predominantly form unwanted tertiary beta-ketoamines. The pressurized method described in CN1665773A suppresses this side reaction, directly yielding the desired N-monosubstituted beta-ketoamines with significantly higher purity and yield (up to 75% vs 45%).
Q: What are the key advantages of using amine salts in this synthesis?
A: Using addition salts of primary amines, such as methylamine hydrochloride, enhances the stability of the intermediate beta-ketoamines. Unlike free bases which may decompose upon distillation, the salt forms can be stored for extended periods without degradation, simplifying inventory management and logistics.
Q: Can this process be scaled for industrial production of neuroactive compounds?
A: Yes, the process utilizes standard industrial equipment like autoclaves and common solvents like ethanol or isopropanol. The elimination of complex purification steps like steam distillation makes it highly suitable for the commercial scale-up of complex pharmaceutical intermediates like duloxetine precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Monosubstituted Beta-Amino Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN1665773A can be successfully translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of N-monosubstituted beta-amino alcohol meets the highest standards of quality and consistency required by global regulatory agencies. Our commitment to technical excellence allows us to navigate the complexities of pressurized chemistry safely and effectively.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthetic route for their specific pipeline needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your project volume and timeline. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can accelerate your development programs while reducing overall production costs. Let us be your trusted partner in bringing high-quality neuroactive intermediates to the market.
