Revolutionizing Cinnamamide Production: A Green Aqueous Convergent Synthesis Strategy for Commercial Scale
Introduction to Advanced Cinnamamide Synthesis Technology
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign methodologies for constructing complex molecular scaffolds. Patent CN1740145A introduces a transformative approach to synthesizing cinnamamide compounds, a critical class of structures found in numerous bioactive molecules ranging from cardiovascular agents to anti-inflammatory drugs. This technology pivots away from traditional linear synthetic pathways, opting instead for a convergent strategy executed entirely within an aqueous phase. By leveraging triphenylphosphine, inorganic bases, and inorganic salts in water, this method achieves high conversion rates under mild conditions, typically ranging from room temperature to reflux. The significance of this innovation lies not merely in the chemical transformation itself but in its profound implications for process safety and environmental compliance. For R&D directors and process chemists, this represents a viable pathway to streamline the production of high-purity cinnamamide derivatives while drastically reducing the reliance on hazardous volatile organic compounds (VOCs). The ability to synthesize these valuable intermediates without the need for intermediate isolation marks a significant leap forward in process intensification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of cinnamamide compounds has relied heavily on linear synthetic sequences that are inherently inefficient and environmentally burdensome. As illustrated in the traditional pathways, aromatic aldehydes are first condensed with malonic acid via the Knoevenagel reaction to form cinnamic acids, which subsequently require activation with thionyl chloride or oxalyl chloride to generate reactive acid chlorides. 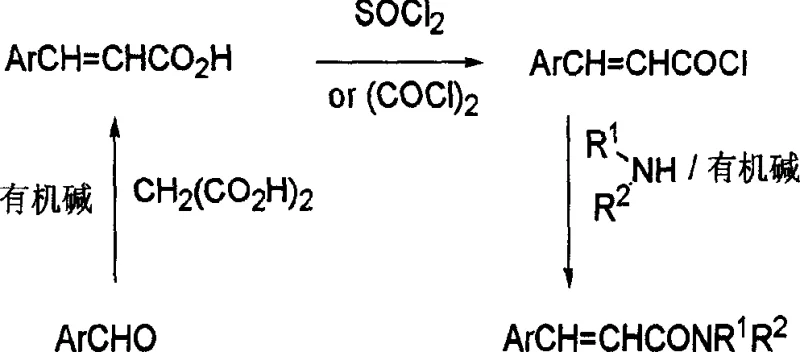 These acid chlorides are then reacted with amines in the presence of bases to yield the final amide products. Alternatively, Wittig or Horner-Emmons reactions are employed to generate cinnamate esters, which must then undergo hydrolysis and subsequent amidation. These multi-step linear processes suffer from low overall yields due to the cumulative losses at each isolation stage. Furthermore, they necessitate the use of toxic, flammable, and expensive organic solvents such as benzene, toluene, tetrahydrofuran, and dimethylformamide. From a supply chain perspective, the handling of corrosive reagents like thionyl chloride poses significant safety risks and requires specialized equipment for waste gas scrubbing, thereby inflating capital expenditure and operational complexity.
These acid chlorides are then reacted with amines in the presence of bases to yield the final amide products. Alternatively, Wittig or Horner-Emmons reactions are employed to generate cinnamate esters, which must then undergo hydrolysis and subsequent amidation. These multi-step linear processes suffer from low overall yields due to the cumulative losses at each isolation stage. Furthermore, they necessitate the use of toxic, flammable, and expensive organic solvents such as benzene, toluene, tetrahydrofuran, and dimethylformamide. From a supply chain perspective, the handling of corrosive reagents like thionyl chloride poses significant safety risks and requires specialized equipment for waste gas scrubbing, thereby inflating capital expenditure and operational complexity.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent employs a convergent synthesis strategy that merges the carbon skeleton construction and amide bond formation into a streamlined sequence. 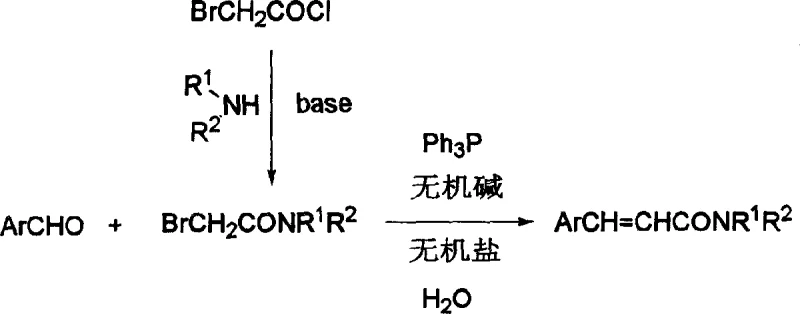 This approach utilizes alpha-bromoacetamide and aromatic aldehydes as the primary building blocks, reacting them directly in the presence of triphenylphosphine and an inorganic base within a water medium. The reaction proceeds through the in situ generation of a phosphonium ylide, which immediately undergoes olefination with the aldehyde to furnish the cinnamamide structure. Crucially, the intermediates formed during this two-step sequence do not require separation, effectively collapsing multiple unit operations into a single pot. This convergence not only saves considerable time and energy but also significantly enhances the overall mass balance of the process. By shifting the reaction medium to water, the process eliminates the procurement and disposal costs associated with hazardous organic solvents, offering a cleaner, safer, and more economically attractive route for the manufacturing of reliable cinnamamide suppliers.
This approach utilizes alpha-bromoacetamide and aromatic aldehydes as the primary building blocks, reacting them directly in the presence of triphenylphosphine and an inorganic base within a water medium. The reaction proceeds through the in situ generation of a phosphonium ylide, which immediately undergoes olefination with the aldehyde to furnish the cinnamamide structure. Crucially, the intermediates formed during this two-step sequence do not require separation, effectively collapsing multiple unit operations into a single pot. This convergence not only saves considerable time and energy but also significantly enhances the overall mass balance of the process. By shifting the reaction medium to water, the process eliminates the procurement and disposal costs associated with hazardous organic solvents, offering a cleaner, safer, and more economically attractive route for the manufacturing of reliable cinnamamide suppliers.
Mechanistic Insights into Aqueous Phase Wittig-Type Olefination
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the aqueous environment and the specific reagent combination. The reaction initiates with the nucleophilic attack of triphenylphosphine on the alpha-bromoacetamide, generating a quaternary phosphonium salt intermediate. In traditional organic solvents, the subsequent deprotonation to form the ylide might require strong, moisture-sensitive bases; however, in this system, mild inorganic bases such as lithium hydroxide or sodium carbonate are sufficient. The presence of inorganic salts, particularly lithium chloride, plays a pivotal catalytic role, likely enhancing the reaction rate through ionic strength effects or specific ion interactions that stabilize the transition state. Once the ylide is generated, it reacts rapidly with the aromatic aldehyde to form the carbon-carbon double bond, yielding the cinnamamide product with high stereoselectivity, predominantly favoring the E-isomer. This mechanism avoids the formation of free cinnamic acid intermediates, thereby bypassing the need for harsh activating agents. For technical teams, understanding this mechanism is vital for optimizing reaction parameters such as pH and ionic strength to maximize yield and minimize byproduct formation.
Furthermore, the impurity profile of this aqueous process is markedly cleaner compared to organic solvent-based methods. In conventional syntheses, side reactions such as ester hydrolysis or over-chlorination can lead to complex impurity spectra that are difficult to purge. In the aqueous system described, the solubility differences between the organic product and inorganic byproducts (such as triphenylphosphine oxide and salts) facilitate straightforward workup procedures. The product can be efficiently extracted using ethyl acetate, leaving the bulk of inorganic residues in the aqueous phase. This inherent partitioning behavior simplifies downstream processing and reduces the load on purification columns. Additionally, the tolerance of this method towards various functional groups on the aromatic ring—including electron-withdrawing nitro groups and electron-donating methoxy groups—demonstrates its robustness. The ability to maintain high reactivity even with substrates containing active hydrogen atoms, such as hydroxyl groups, underscores the mildness of the reaction conditions, making it an ideal candidate for synthesizing complex, functionalized pharmaceutical intermediates without extensive protecting group strategies.
How to Synthesize Cinnamamide Compounds Efficiently
Implementing this synthesis route requires precise control over stoichiometry and reaction conditions to ensure optimal performance. The standard protocol involves suspending the alpha-bromoacetamide and aromatic aldehyde in an aqueous solution containing the inorganic base and salt, followed by the addition of triphenylphosphine. The mixture is then heated to reflux, where the reaction typically completes within a remarkably short timeframe, often ranging from 5 minutes to 2 hours depending on the electronic nature of the aldehyde substrate. Following the reaction, the mixture is cooled and extracted, and the organic layer is washed and dried to afford the crude product, which can be further purified by standard chromatographic techniques.
- Suspend alpha-bromoacetamide, aromatic aldehyde, and triphenylphosphine in an aqueous solution containing inorganic base and inorganic salt.
- Heat the reaction mixture from room temperature to reflux for 5 minutes to 2 hours to facilitate the formation of the phosphonium ylide and subsequent olefination.
- Extract the product with ethyl acetate, wash with saturated brine, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous synthesis technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of toxic organic solvents like benzene and DMF directly translates to substantial cost reduction in pharmaceutical intermediate manufacturing. Solvent procurement, storage, and especially waste disposal represent significant line items in the operational budget of any chemical plant; replacing these with water drastically lowers these overheads. Moreover, the avoidance of thionyl chloride and oxalyl chloride removes the need for specialized corrosion-resistant reactors and complex off-gas treatment systems, further reducing capital intensity. The convergent nature of the synthesis means fewer unit operations and less equipment occupancy time, allowing for higher throughput and better asset utilization. This efficiency gain is critical for meeting tight production schedules and ensuring supply continuity for downstream API manufacturers.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the necessity for expensive and hazardous organic solvents and activating agents. Traditional routes require the purchase of thionyl chloride and large volumes of anhydrous solvents, both of which carry high price tags and regulatory compliance costs. By utilizing water as the primary medium and inexpensive inorganic salts as accelerants, the raw material cost profile is significantly improved. Additionally, the one-pot nature of the reaction reduces labor costs and energy consumption associated with multiple isolation and drying steps. The simplified workup procedure minimizes solvent usage during extraction, contributing to a leaner, more cost-effective manufacturing process that enhances overall profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically alpha-bromoacetamides and common aromatic aldehydes, are widely available commodity chemicals with stable supply chains. Unlike specialized reagents that may be subject to market volatility or long lead times, these precursors can be sourced from multiple vendors globally. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures. This reliability is paramount for supply chain heads who must guarantee consistent delivery of high-purity cinnamamide derivatives to their clients. The reduced dependency on hazardous reagents also simplifies logistics and transportation, as water-based formulations are generally safer and easier to handle than flammable organic solutions.
- Scalability and Environmental Compliance: Scaling up chemical processes often amplifies safety and environmental challenges, but this aqueous method mitigates many of those risks. Water is non-flammable and has a high heat capacity, making temperature control easier and safer on a large scale. The reduction in hazardous waste generation aligns perfectly with increasingly stringent global environmental regulations, such as REACH and TSCA. Facilities adopting this technology can expect smoother permitting processes and lower environmental liability. The high atom economy of the convergent synthesis ensures that less waste is generated per kilogram of product, supporting sustainability goals. This environmental stewardship not only protects the company from regulatory fines but also enhances its reputation as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: What are the primary advantages of the aqueous synthesis method for cinnamamides over traditional organic solvent methods?
A: The aqueous method eliminates the need for toxic and expensive organic solvents like benzene, toluene, and DMF. It simplifies the process by avoiding the isolation of intermediates, thereby reducing energy consumption and waste discharge while maintaining high yields.
Q: How does the addition of inorganic salts like LiCl impact the reaction efficiency?
A: Inorganic salts, particularly lithium chloride, significantly accelerate the reaction rate. They likely function through a salting-out effect or by stabilizing the transition state of the ylide formation, allowing reactions to complete in minutes rather than hours.
Q: Is this synthesis method suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the method is highly suitable for scale-up. The use of water as a solvent reduces fire hazards and solvent recovery costs, while the convergent nature of the synthesis improves overall atom economy and throughput for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinnamamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global market. Our technical team has extensively evaluated the aqueous convergent synthesis route for cinnamamide compounds and confirmed its potential for delivering high-quality intermediates with superior efficiency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required by top-tier pharmaceutical companies. We are committed to leveraging this green chemistry innovation to provide our partners with a sustainable and reliable source of complex amide intermediates.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through this cutting-edge technology. Our experts are ready to conduct a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a wealth of chemical expertise and a commitment to excellence that drives value across your entire organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
