Advanced Metal-Free Synthesis of Cinnamamide Compounds for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic methodologies that balance high efficiency with stringent regulatory compliance, particularly regarding impurity profiles and environmental impact. Patent CN111018735B introduces a transformative approach to the preparation of cinnamamide compounds, a critical class of intermediates widely utilized in the synthesis of anticancer, antituberculosis, and antimicrobial agents. This innovation fundamentally shifts the paradigm from traditional transition metal-catalyzed systems to a metal-free protocol utilizing thiuram disulfide as a key reagent. By eliminating the reliance on expensive and potentially toxic catalysts such as copper or rhodium, this method addresses significant pain points in modern process chemistry, including heavy metal residue control and complex waste stream management. The technical breakthrough lies in the ability to drive the amidation coupling directly under mild thermal conditions without the need for additional activating agents or additives. For R&D directors and process chemists, this represents a substantial opportunity to streamline development timelines while ensuring the final active pharmaceutical ingredients meet the rigorous purity standards required by global health authorities. The versatility of this system allows for the accommodation of diverse functional groups, ensuring that complex molecular architectures can be accessed without compromising on yield or selectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cinnamamide derivatives has heavily relied on transition metal catalysis, often employing copper or rhodium complexes to facilitate the coupling between carboxylic acids and amines. While effective in laboratory settings, these conventional methods present severe drawbacks when translated to commercial manufacturing scales, primarily due to the inherent risks associated with heavy metal contamination. The presence of residual metals in pharmaceutical intermediates necessitates extensive downstream purification processes, such as specialized scavenging treatments or repeated recrystallization steps, which drastically increase production costs and extend lead times. Furthermore, the disposal of metal-laden waste streams poses significant environmental compliance challenges, requiring specialized treatment facilities that add to the overall operational expenditure. The sensitivity of these metal catalysts to air and moisture often demands inert atmosphere conditions, adding complexity to the reactor setup and increasing the energy consumption required for maintaining strict anhydrous environments. Additionally, the cost volatility of precious metals like rhodium can introduce unpredictable financial risks into the supply chain, making long-term budget planning difficult for procurement managers. These cumulative factors render traditional metal-catalyzed routes less attractive for high-volume production where cost efficiency and regulatory safety are paramount concerns for stakeholders.
The Novel Approach
In stark contrast to the complexities of metal-catalyzed systems, the novel method disclosed in patent CN111018735B utilizes a metal-free strategy that leverages thiuram disulfide to drive the reaction forward efficiently. This approach simplifies the reaction setup by allowing the coupling to proceed in common organic solvents under aerobic conditions, thereby removing the need for expensive inert gas protection or rigorous moisture exclusion. The use of thiuram disulfide not only acts as a promoter for the amidation but also ensures that the reaction byproducts are easier to manage compared to heavy metal salts. This simplification of the reaction matrix significantly reduces the burden on the purification team, as the absence of metal ions eliminates the need for specific metal-scavenging resins or complex chromatographic separations aimed at reducing metal content to ppm levels. The operational simplicity extends to the workup procedure, where standard concentration and crystallization techniques are often sufficient to isolate the target cinnamamide compounds in high purity. For supply chain leaders, this translates to a more resilient manufacturing process that is less susceptible to raw material shortages of precious metals and more adaptable to varying production scales. The robustness of this metal-free protocol ensures consistent batch-to-batch quality, which is critical for maintaining supply continuity in the competitive pharmaceutical intermediate market.
Mechanistic Insights into Thiuram Disulfide Mediated Amidation
The core of this technological advancement lies in the unique reactivity of thiuram disulfide, which facilitates the activation of the carboxylic acid moiety without the need for traditional coupling reagents like carbodiimides. Mechanistically, the thiuram disulfide likely undergoes homolytic cleavage or nucleophilic attack under thermal conditions to generate reactive sulfur species that activate the carbonyl group of the cinnamic acid. This activation lowers the energy barrier for the nucleophilic attack by the amine component, allowing the amide bond to form smoothly at temperatures ranging from 90°C to 100°C. The absence of external catalysts means that the reaction pathway is governed primarily by the intrinsic electronic properties of the substrates and the reagent, leading to a cleaner reaction profile with fewer side products. Understanding this mechanism is crucial for process optimization, as it allows chemists to fine-tune reaction parameters such as solvent polarity and stoichiometry to maximize conversion rates. The compatibility of this mechanism with a wide range of substituents, including electron-withdrawing halogens and electron-donating alkoxy groups, suggests a high degree of tolerance to steric and electronic variations. This mechanistic robustness ensures that the process remains reliable even when scaling up to multi-kilogram batches, providing confidence to technical teams regarding the reproducibility of the synthesis.
Impurity control is a critical aspect of this synthesis, particularly given the potential for over-reaction or decomposition of the thiuram reagent at elevated temperatures. The patent data indicates that by carefully controlling the reaction time between 36 to 48 hours and maintaining the temperature within the specified 90-100°C window, the formation of undesired byproducts is minimized. The purification strategy typically involves thin-layer chromatography or standard column chromatography using ethyl acetate and petroleum ether systems, which effectively separate the target cinnamamide from any unreacted starting materials or sulfur-containing byproducts. The high yields reported, ranging from 40% to 92% across various substrates, demonstrate that the reaction selectivity is high, reducing the loss of valuable raw materials. For quality assurance teams, the predictable impurity profile simplifies the validation process, as the absence of metal-related impurities removes a major category of potential contaminants from the risk assessment. This level of control over the chemical outcome is essential for meeting the stringent specifications required for pharmaceutical intermediates, ensuring that the final product is suitable for subsequent downstream transformations without requiring extensive reprocessing.
How to Synthesize Cinnamamide Compounds Efficiently
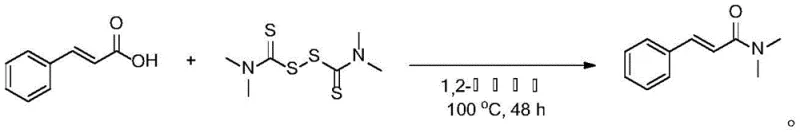
Implementing this synthesis route in a production environment requires a clear understanding of the operational parameters to ensure safety and efficiency. The process begins with the precise weighing of cinnamic acid derivatives and thiuram disulfide, typically in a molar ratio favoring the disulfide slightly to drive the reaction to completion. These reagents are dissolved in a suitable organic solvent such as 1,2-dichloroethane, which provides the necessary solubility and thermal stability for the reaction conditions. The mixture is then heated in an oil bath or reactor jacket to maintain a consistent temperature between 90°C and 100°C for a duration of 36 to 48 hours, allowing the coupling to proceed fully. Following the reaction period, the mixture is cooled to room temperature, and the solvent is removed under reduced pressure to yield a crude concentrate. The detailed standardized synthesis steps, including specific safety precautions and equipment requirements, are outlined in the guide below for technical reference.
- Mix cinnamic acid derivatives with thiuram disulfide in an organic solvent such as dichloroethane at a molar ratio of approximately 1: 1.1.
- Heat the reaction mixture to 90-100°C and stir for 36-48 hours to facilitate the coupling reaction without metal catalysts.
- Concentrate the reaction solution and purify the resulting cinnamamide product using chromatography with ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-free synthesis method offers substantial strategic advantages for procurement and supply chain management teams looking to optimize costs and reliability. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of expensive metals and the subsequent removal processes required to meet regulatory limits. This reduction in processing complexity directly translates to lower operational expenditures, as fewer unit operations are required to achieve the desired purity specifications. Furthermore, the use of readily available and economically priced raw materials like cinnamic acids and thiuram disulfides ensures a stable supply chain that is less vulnerable to the geopolitical and market fluctuations often seen with precious metals. The mild reaction conditions also contribute to energy efficiency, as the process does not require extreme temperatures or pressures that would demand specialized high-specification reactor vessels. These factors combined create a manufacturing profile that is not only cost-effective but also environmentally sustainable, aligning with the increasing corporate focus on green chemistry and reduced carbon footprints. For supply chain heads, this means a more predictable and resilient sourcing strategy that can support long-term production contracts with confidence.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the complete removal of transition metal catalysts, which are often among the most expensive components in a synthetic route. By substituting these with thiuram disulfide, manufacturers avoid the high procurement costs associated with metals like rhodium or palladium, as well as the capital investment required for metal scavenging technologies. Additionally, the simplified workup procedure reduces the consumption of solvents and chromatography media, further lowering the variable costs per kilogram of product. The high atom economy and yield of the reaction ensure that raw material utilization is optimized, minimizing waste generation and the associated disposal costs. This comprehensive approach to cost management allows companies to offer more competitive pricing to their clients while maintaining healthy profit margins. The cumulative effect of these savings is a significantly more economical manufacturing process that enhances the overall financial viability of producing cinnamamide intermediates.
- Enhanced Supply Chain Reliability: Supply chain reliability is greatly improved by the use of commodity chemicals that are widely available from multiple global suppliers, reducing the risk of single-source dependency. Unlike specialized catalysts that may have long lead times or limited production capacity, cinnamic acids and thiuram disulfides are standard industrial chemicals with robust supply networks. This availability ensures that production schedules can be maintained without interruption, even in the face of market disruptions or logistical challenges. The simplicity of the process also means that it can be easily transferred between different manufacturing sites or contract manufacturing organizations without significant requalification efforts. For procurement managers, this flexibility provides a strategic advantage, allowing for dynamic sourcing strategies that can adapt to changing market conditions. The result is a supply chain that is not only cost-efficient but also highly resilient and capable of meeting demanding delivery timelines consistently.
- Scalability and Environmental Compliance: Scalability is a key strength of this method, as the one-pot nature of the reaction minimizes the need for complex intermediate isolation steps that often bottleneck scale-up efforts. The mild thermal conditions and lack of sensitive catalysts make the process safe and manageable in large-scale reactors, facilitating a smooth transition from pilot plant to commercial production. From an environmental compliance standpoint, the metal-free nature of the synthesis significantly reduces the toxicity of the waste stream, simplifying effluent treatment and disposal procedures. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, reducing the regulatory burden on the manufacturing facility. The reduced environmental impact also enhances the company's reputation as a responsible manufacturer, which is increasingly important for securing contracts with major pharmaceutical partners. Overall, the process offers a scalable and compliant solution that meets the dual demands of production volume and environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in patent CN111018735B, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of adopting this technology for specific product lines or manufacturing campaigns. The information covers aspects ranging from reaction conditions to product quality, providing a comprehensive overview for technical assessment.
Q: Does this synthesis method leave heavy metal residues in the final product?
A: No, the patented method CN111018735B operates under completely metal-free conditions using thiuram disulfide, eliminating the risk of copper or rhodium contamination common in traditional catalytic methods.
Q: What is the substrate scope for this cinnamamide preparation technique?
A: The process demonstrates broad compatibility with various functional groups including alkyl, alkoxy, halogens, and nitro groups, allowing for the synthesis of diverse cinnamamide derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions, readily available raw materials, and simple one-pot operations, making it highly scalable for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinnamamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly evaluated the metal-free cinnamamide synthesis route and is fully equipped to implement this process at commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory to plant is seamless and efficient. Our facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest industry standards. By leveraging this innovative metal-free technology, we can offer our partners a supply of cinnamamide intermediates that are not only cost-effective but also free from the regulatory risks associated with heavy metal residues. Our commitment to technical excellence and operational reliability makes us the ideal partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this metal-free protocol for your supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate our capability to deliver high-quality cinnamamide compounds consistently. Let us collaborate to optimize your manufacturing process and secure a reliable supply of critical pharmaceutical intermediates for your future success.
