Advanced Multi-Stage Hydrolysis for Corrosion-Free Production of Halogenated Aromatic Amines
Advanced Multi-Stage Hydrolysis for Corrosion-Free Production of Halogenated Aromatic Amines
The chemical industry constantly seeks robust methodologies for synthesizing halogenated aromatic compounds, particularly those serving as critical building blocks for pharmaceuticals and agrochemicals. Patent CN1369479A introduces a transformative approach to producing aromatics, specifically addressing the persistent challenge of reactor corrosion caused by hydrogen halide byproducts. This technology focuses on the hydrolysis and decarboxylation of aromatic cyano compounds, such as 4-amino-2,3,5,6-tetrafluorobenzonitrile, to yield valuable amines like 2,3,5,6-tetrafluoroaniline. Unlike traditional single-step processes that often suffer from equipment degradation and yield loss due to impurity-driven side reactions, this invention delineates a multi-stage reaction protocol. By strategically manipulating acid concentration and temperature across distinct reaction phases, the method effectively suppresses the generation of corrosive hydrogen fluoride, thereby enabling the use of standard glass-lined or stainless steel reactors even with lower-purity feedstocks. This breakthrough represents a significant leap forward for manufacturers aiming to optimize cost reduction in pharmaceutical intermediates manufacturing while maintaining stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
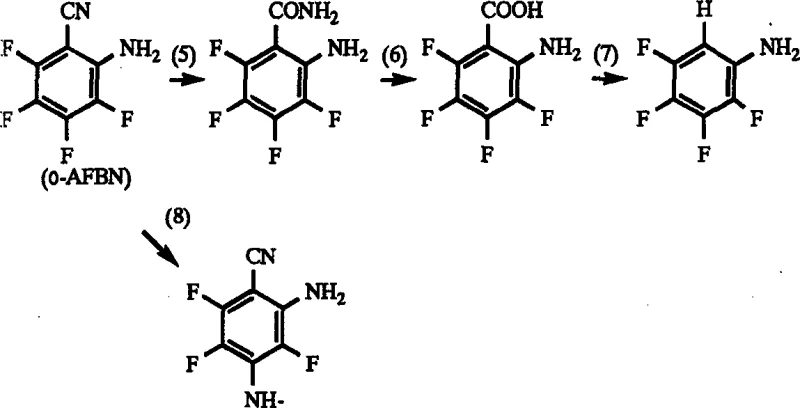
Historically, the synthesis of polyhalogenated anilines has been plagued by severe engineering constraints, primarily stemming from the corrosive nature of the reaction environment. In conventional one-step hydrolysis and decarboxylation processes, the presence of isomeric impurities in the starting material, such as 2-amino-3,4,5,6-tetrafluorobenzonitrile, triggers detrimental side reactions. These impurities can undergo nucleophilic substitution with the target product or other intermediates, releasing hydrogen fluoride (HF) as a byproduct. HF is notoriously aggressive towards glass-lined and stainless steel reactors, leading to pitting, thinning of vessel walls, and eventual equipment failure. Furthermore, to mitigate this, prior art often necessitates the use of ultra-high purity starting materials, which requires expensive and time-consuming purification steps like recrystallization. This reliance on pristine feedstocks not only inflates raw material costs but also introduces bottlenecks in the supply chain, making the commercial scale-up of complex polymer additives or pharmaceutical precursors economically challenging. The inability to tolerate impurities forces manufacturers into a cycle of high operational expenditure and frequent maintenance downtime.
The Novel Approach
The methodology disclosed in CN1369479A circumvents these limitations through a sophisticated multi-stage reaction design that fundamentally alters the chemical environment to favor the desired pathway. Instead of attempting to drive hydrolysis and decarboxylation simultaneously under uniform conditions, the process divides the reaction into a leading portion and a latter portion with distinct parameters. In the initial stage, the aromatic cyano compound is introduced into a highly concentrated acidic medium, typically sulfuric acid ranging from 80% to 98% by mass. Under these conditions, the amino groups on the aromatic ring are rapidly protonated to form ammonium salts, which significantly reduces their nucleophilicity. This protonation acts as a protective shield, preventing the amino group from attacking adjacent fluorine atoms and thus inhibiting the release of hydrogen fluoride. By controlling the conversion rate in this first stage to stop primarily at the amide intermediate, the process minimizes the contact time between reactive species that could generate corrosive byproducts. This strategic segmentation allows for the use of cost-effective, industrial-grade raw materials without compromising reactor integrity or final product yield.
Mechanistic Insights into Acid-Catalyzed Hydrolysis and Decarboxylation
To fully appreciate the efficacy of this synthesis route, one must delve into the mechanistic interplay between acid concentration, temperature, and molecular reactivity. The core innovation lies in the manipulation of the electronic properties of the aromatic ring through protonation. In the leading portion of the reaction, the high concentration of sulfuric acid ensures that the amino substituent exists predominantly in its protonated form (-NH3+). This positive charge exerts a strong electron-withdrawing inductive effect, which deactivates the aromatic ring towards nucleophilic aromatic substitution. Consequently, even if isomeric impurities are present, their ability to displace fluorine atoms is drastically diminished. The reaction is carefully monitored to ensure that the conversion of the nitrile group proceeds to the amide stage (e.g., 4-amino-2,3,5,6-tetrafluorobenzamide) without fully progressing to the amine. This intermediate state is crucial because it locks the molecule in a less reactive configuration, preventing the cascade of side reactions that typically lead to tar formation and equipment corrosion. The precise control of temperature, generally maintained between 50°C and 180°C, further fine-tunes the kinetics to favor hydrolysis over substitution.
In the latter stage, the reaction conditions are shifted to facilitate the final transformation into the target aromatic amine. Water is added to the reaction mixture to dilute the acid concentration to a range of 50% to 95%, and the temperature is often increased. This dilution reduces the protonation equilibrium, gradually freeing the amino group while providing the necessary medium for the hydrolysis of the amide to the carboxylic acid and its subsequent decarboxylation. The presence of water also aids in the removal of ammonia or ammonium salts formed during the process. Crucially, because the bulk of the potentially corrosive side reactions were suppressed in the first stage, the system remains stable even as the final product forms. The final step involves component distillation with water, where the target aromatic amine, being less soluble or forming an azeotrope, is separated from the acidic aqueous phase. This continuous removal of the product drives the equilibrium forward and prevents the product from remaining in the harsh acidic environment for extended periods, further preserving its purity and stability.
How to Synthesize 2,3,5,6-Tetrafluoroaniline Efficiently
The practical implementation of this patented technology offers a streamlined pathway for producing high-value fluorinated intermediates. The process begins with the preparation of a reaction vessel, preferably glass-lined or stainless steel, charged with concentrated sulfuric acid. The starting material, which may contain up to 6% isomeric impurities, is fed into the reactor at a controlled rate to manage the exotherm and maintain the desired acid concentration profile. The detailed standardized synthesis steps, including specific molar ratios, agitation speeds, and precise temperature ramps for both the leading and latter stages, are outlined in the guide below. Following the reaction, the mixture undergoes a workup procedure involving dilution and distillation to isolate the pure crystalline product. This robust protocol ensures that manufacturers can achieve yields exceeding 95% while virtually eliminating the risk of reactor corrosion, making it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates.
- Conduct a leading portion reaction by adding aromatic cyano compounds to high-concentration aqueous sulfuric acid (80-98%) at 50-180°C to achieve partial hydrolysis to the amide stage.
- Dilute the reaction mixture with water to lower acid concentration (50-95%) and increase temperature to facilitate the decarboxylation of the intermediate carboxylic acid or amide.
- Perform component distillation with water to separate and recover the final aromatic amine product from the reaction solution while minimizing hydrogen halide generation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this multi-stage hydrolysis technology translates into tangible operational efficiencies and risk mitigation. Traditional methods often require the sourcing of ultra-high purity precursors, which are not only expensive but also subject to limited availability from specialized suppliers. By enabling the use of technical-grade starting materials with acceptable impurity profiles, this process diversifies the supplier base and reduces dependency on niche vendors. This flexibility enhances supply chain resilience, ensuring that production schedules are not disrupted by raw material shortages. Furthermore, the elimination of corrosive byproducts like hydrogen fluoride extends the lifespan of capital equipment. Reactors that would previously require frequent lining repairs or replacement due to HF attack can now operate for extended campaigns with minimal maintenance. This reduction in unplanned downtime directly contributes to lower overhead costs and more predictable production cycles, aligning perfectly with the goals of a reliable agrochemical intermediate supplier seeking to optimize asset utilization.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substantial reduction in both raw material and maintenance costs. By removing the necessity for rigorous purification of the starting cyano compounds, manufacturers save significantly on solvent usage, energy for recrystallization, and labor associated with purification steps. Additionally, the suppression of hydrogen fluoride generation means that expensive corrosion-resistant alloys or frequent glass-lining repairs are no longer a recurring budget item. The process allows for the use of standard industrial reactors, lowering the barrier to entry for production and reducing the total cost of ownership for the manufacturing facility. These savings can be passed down the value chain, offering competitive pricing for downstream customers without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of sourcing high-purity intermediates. This method mitigates that risk by tolerating a broader specification range for incoming raw materials. Suppliers can provide technical-grade 4-amino-2,3,5,6-tetrafluorobenzonitrile without the need for additional refining, speeding up the procurement cycle. The robustness of the reaction also means that batch-to-batch variability is minimized, leading to consistent output quality. This consistency is vital for long-term contracts with pharmaceutical clients who require strict adherence to specifications. By stabilizing the production process against feedstock fluctuations, companies can offer more reliable delivery commitments and build stronger partnerships with their clients.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, particularly when handling corrosive acids and gases. This multi-stage approach inherently improves safety by minimizing the generation of hazardous hydrogen fluoride gas, simplifying waste gas treatment requirements. The aqueous workup and distillation steps are well-established unit operations that scale linearly from pilot plant to commercial production volumes. The reduced corrosivity also lowers the risk of leaks or containment failures, enhancing overall plant safety. From an environmental perspective, the higher atom economy and reduced need for purification solvents result in a smaller waste footprint. This alignment with green chemistry principles facilitates easier regulatory compliance and supports corporate sustainability goals, making the process attractive for modern, eco-conscious manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrolysis technology. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this process into existing production lines. The answers are derived directly from the experimental data and theoretical framework provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Whether you are concerned about equipment compatibility, raw material specifications, or product purity, these insights provide a clear roadmap for adoption. For further technical discussions or custom feasibility studies, our team is available to provide detailed route assessments tailored to your specific production needs.
Q: How does this multi-stage method prevent reactor corrosion compared to conventional one-step processes?
A: Conventional methods often generate hydrogen fluoride (HF) as a byproduct due to side reactions between impurities and the product. This patent's method uses high acid concentration in the first stage to protonate amino groups, reducing their nucleophilicity and suppressing the substitution of fluorine atoms that leads to HF generation.
Q: Can this process tolerate lower purity starting materials without affecting yield?
A: Yes, the process is specifically designed to handle raw materials containing isomeric impurities, such as 2-amino-3,4,5,6-tetrafluorobenzonitrile. By controlling the reaction stages, the interaction between these impurities and the target product is minimized, allowing for high yields even with starting materials below 97% purity.
Q: What are the typical reaction conditions for the decarboxylation stage?
A: In the latter stage, water is added to reduce the sulfuric acid concentration to between 50% and 95%, and the temperature is typically raised to between 60°C and 180°C. This shift in conditions favors the hydrolysis of the amide to the acid and subsequent decarboxylation to the final amine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5,6-Tetrafluoroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this multi-stage hydrolysis method are fully realized in a commercial setting. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 2,3,5,6-tetrafluoroaniline meets the exacting standards required by the global pharmaceutical and agrochemical industries. Our infrastructure is designed to handle complex fluorinated chemistries safely and efficiently, leveraging the corrosion-resistant advantages of this technology to deliver consistent supply.
We invite potential partners to engage with us for a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this advanced synthesis route for your supply chain. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your production goals. By collaborating with us, you gain access to a supply partner that prioritizes both technological excellence and commercial viability. Contact us today to discuss how we can optimize your sourcing strategy for high-purity fluorinated intermediates and drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
