Advanced Synthetic Route for 2,3,5,6-Tetrafluoroaniline: Technical Breakthroughs and Commercial Scalability
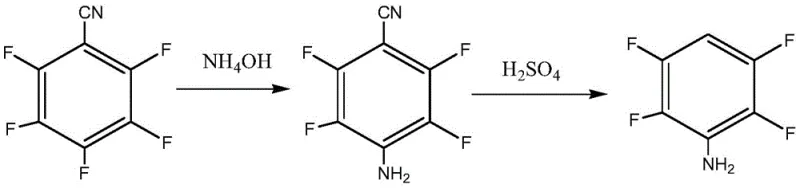
The global demand for high-performance fluorinated building blocks continues to surge, driven by the expanding pharmaceutical and electronic materials sectors. A pivotal development in this domain is documented in patent CN107915647B, which discloses a robust and highly efficient synthetic method for producing 2,3,5,6-tetrafluoroaniline. This critical intermediate serves as a cornerstone for the manufacture of advanced medicaments and fluorescent compounds, yet its domestic industrial production has historically been constrained by high import costs and limited availability. The patented methodology addresses these bottlenecks by introducing a novel two-step sequence that begins with the selective ammoniation of 2,3,4,5,6-pentafluorobenzonitrile. By leveraging ammonia water as the nitrogen source in conjunction with a specific phase transfer catalyst system, the process achieves exceptional regioselectivity at low temperatures. This technical advancement not only secures a high-purity intermediate exceeding 99.5% but also establishes a foundation for a streamlined, cost-effective manufacturing workflow that is ideally suited for modern chemical supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3,5,6-tetrafluoroaniline has relied on methodologies that are fraught with economic and operational inefficiencies, rendering them suboptimal for large-scale commercial adoption. One prominent prior art approach involves the reductive defluorination of pentafluoroaniline using potassium iodide and hydroiodic acid under prolonged heating conditions. This traditional pathway suffers from inherently low yields due to the formation of complex side products and the difficulty in controlling the reaction selectivity. Furthermore, the reliance on stoichiometric amounts of iodine-based reagents introduces a significant cost burden, as these materials are substantially more expensive than commodity chemicals. The generation of iodine-containing waste streams also poses severe environmental challenges, necessitating costly treatment protocols to meet regulatory standards. Additionally, the harsh thermal conditions required for these older methods often lead to equipment corrosion and increased energy consumption, further eroding the profit margins for manufacturers attempting to produce this valuable intermediate.
The Novel Approach
In stark contrast, the innovative route detailed in the patent data utilizes a strategic nitrile-based precursor to bypass the limitations associated with direct aniline modification. By starting with 2,3,4,5,6-pentafluorobenzonitrile, the process exploits the strong electron-withdrawing nature of the cyano group to activate the aromatic ring for nucleophilic attack. The use of ammonia water as the ammoniating agent represents a paradigm shift towards greener chemistry, replacing expensive halides with a ubiquitous and affordable nitrogen source. The integration of tetrabutylammonium bisulfate as a phase transfer catalyst facilitates the reaction in an ethyl acetate medium, ensuring efficient mass transfer between the aqueous ammonia and the organic substrate. This combination allows the reaction to proceed smoothly at mild temperatures ranging from -5 to -15°C, effectively suppressing the formation of unwanted isomers such as 2-amino-3,4,5,6-tetrafluorobenzonitrile. The result is a direct, high-yielding pathway that delivers the intermediate with purity levels exceeding 99.5%, thereby eliminating the need for complex purification steps that typically plague conventional syntheses.
Mechanistic Insights into Phase Transfer Catalyzed Ammoniation
The core of this synthetic breakthrough lies in the precise control of nucleophilic aromatic substitution (SnAr) kinetics through phase transfer catalysis. In the absence of a catalyst, the reaction between the lipophilic pentafluorobenzonitrile and hydrophilic ammonia water would be kinetically sluggish due to phase separation. The tetrabutylammonium cation acts as a molecular ferry, transporting the nucleophilic ammonia species into the organic ethyl acetate phase where the substrate resides. Once in the organic phase, the ammonia attacks the carbon atom at the para-position relative to the nitrile group, which is the most electrophilic site on the ring due to resonance and inductive effects. The low temperature regime of -5 to -15°C is critical; it provides sufficient energy for the desired substitution while kinetically freezing out competing reactions at the ortho-positions. This thermodynamic control ensures that the fluorine atom para to the cyano group is selectively displaced, leading to the exclusive formation of 4-amino-2,3,5,6-tetrafluorobenzonitrile. The subsequent hydrolysis step utilizes concentrated sulfuric acid to convert the nitrile functionality into a carboxylic acid, which then undergoes thermal decarboxylation to yield the final aniline product.
Impurity control is another dimension where this mechanistic approach excels, particularly regarding the suppression of regioisomers. In standard ammoniation reactions without phase transfer agents or temperature control, the statistical probability of attacking various fluorine positions increases, leading to a cocktail of amino-isomers that are notoriously difficult to separate. However, the specific interaction between the phase transfer catalyst and the transition state lowers the activation energy specifically for the para-substitution pathway. The patent data indicates that this selectivity is so profound that the intermediate purity reaches greater than 99.5% directly after concentration, without requiring recrystallization or chromatography. This high level of chemical fidelity is paramount for downstream applications in pharmaceutical synthesis, where trace impurities can compromise the safety profile of the final active ingredient. By maintaining a molar ratio of nitrile to ammonia between 1:1.6 and 1:1.8, the process ensures complete conversion of the starting material while minimizing the presence of unreacted ammonia in the waste stream.
How to Synthesize 2,3,5,6-Tetrafluoroaniline Efficiently
The operational protocol for this synthesis is designed for reproducibility and safety, utilizing standard reactor configurations found in most fine chemical facilities. The process begins with the preparation of the reaction mixture at ambient temperature, followed by a controlled cooling phase to reach the critical -5 to -15°C window before the addition of ammonia. Maintaining this temperature throughout the 10-hour reaction period is essential to preserve the regioselectivity discussed in the mechanistic section. Following the ammoniation, the workup involves a simple phase separation and washing with saturated brine, which removes inorganic salts and excess ammonia efficiently. The second stage involves the careful addition of the isolated intermediate into heated sulfuric acid, where temperature ramping is used to drive the hydrolysis and decarboxylation to completion. For a comprehensive understanding of the exact parameters, reagents ratios, and safety precautions required for execution, please refer to the standardized guide below.
- Perform selective ammoniation of 2,3,4,5,6-pentafluorobenzonitrile using ammonia water and a phase transfer catalyst in ethyl acetate at -5 to -15°C to obtain 4-amino-2,3,5,6-tetrafluorobenzonitrile.
- Hydrolyze the intermediate nitrile using 90% sulfuric acid at elevated temperatures (100°C to 135°C) to effect decarboxylation.
- Neutralize the reaction mixture with sodium hydroxide to pH 8-10 and isolate the final product via steam distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this patented synthetic route offers tangible benefits that extend far beyond mere technical feasibility. The primary value driver is the substantial reduction in raw material costs achieved by substituting expensive iodine reagents with commodity-grade ammonia water and sulfuric acid. This shift not only lowers the direct cost of goods sold but also mitigates the volatility associated with sourcing specialized halogenated reagents from a limited supplier base. Furthermore, the simplified workup procedure, which relies on basic layering and distillation rather than complex chromatographic purification, drastically reduces solvent consumption and processing time. These operational efficiencies translate directly into improved throughput and lower utility costs, making the final product more competitive in the global market. The robustness of the process also implies a lower risk of batch failure, ensuring a more consistent and reliable supply of this critical intermediate for downstream customers.
- Cost Reduction in Manufacturing: The elimination of stoichiometric potassium iodide and hydroiodic acid removes a major cost center from the production budget, as these iodine sources are significantly more expensive per kilogram than ammonia and sulfuric acid. Additionally, the high selectivity of the reaction means that yield losses due to isomer formation are virtually eliminated, maximizing the output from every kilogram of starting pentafluorobenzonitrile. The use of ethyl acetate as a solvent is also advantageous, as it is a widely available, recoverable solvent that is cheaper and easier to handle than many specialized polar aprotic solvents. By avoiding the need for extensive purification steps to remove isomeric impurities, the process saves on both labor and consumable costs associated with column chromatography or multiple recrystallizations.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified because the key reagents—ammonia water, ethyl acetate, and sulfuric acid—are bulk commodities available from numerous global suppliers. This diversity of supply sources reduces the risk of disruption caused by geopolitical issues or single-supplier dependencies that often plague the procurement of specialty halides. The mild reaction conditions also reduce wear and tear on reactor vessels, extending equipment lifespan and minimizing unplanned maintenance downtime that could interrupt production schedules. Consequently, manufacturers can offer more stable lead times and commit to larger volume contracts with greater confidence, knowing that the supply of raw materials is secure and the process is resilient.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial parameters such as heat transfer and mixing efficiency in mind, allowing for seamless transition from pilot plant to multi-ton production. From an environmental perspective, the avoidance of heavy metal catalysts and iodine waste streams simplifies wastewater treatment and reduces the burden on effluent processing facilities. The high atom economy of the ammoniation step ensures that minimal waste is generated relative to the product mass, aligning with modern green chemistry principles and corporate sustainability goals. This compliance advantage is increasingly critical for maintaining operating licenses in regions with stringent environmental regulations, thereby securing long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,3,5,6-tetrafluoroaniline, derived directly from the specifications and examples provided in the patent literature. These insights are intended to clarify the operational nuances of the new method and its implications for quality and cost. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines or for procurement specialists assessing supplier capabilities.
Q: What is the primary advantage of using ammonia water over potassium iodide for this synthesis?
A: Using ammonia water significantly reduces raw material costs compared to expensive iodine reagents and avoids the generation of hazardous iodine-containing waste streams, simplifying environmental compliance.
Q: How does the new method control regioselectivity to avoid isomer byproducts?
A: The process utilizes a phase transfer catalyst (tetrabutylammonium bisulfate) at low temperatures (-5 to -15°C), which directs the nucleophilic attack specifically to the para-position relative to the nitrile group, achieving over 99.5% purity.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method employs readily available commercial reagents like ethyl acetate and sulfuric acid, operates under manageable thermal conditions, and achieves a total yield of approximately 96%, making it highly viable for tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5,6-Tetrafluoroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful implementation of advanced synthetic routes requires not just intellectual property, but deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a manufacturing setting. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2,3,5,6-tetrafluoroaniline meets the exacting standards required for pharmaceutical and electronic applications. Our commitment to quality assurance means that we can consistently deliver the high-purity profiles necessary for sensitive downstream reactions, providing our partners with the reliability they need to maintain their own production schedules.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic advantages of switching to this method for your projects. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs, allowing you to make informed decisions based on concrete technical and commercial data.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
