Advanced Selective Hydrogenation of Citral for High-Purity Citronellol Manufacturing
The global demand for high-purity fragrance intermediates continues to drive innovation in catalytic hydrogenation technologies, particularly for valuable terpenoids like citronellol. Patent CN102295531B introduces a groundbreaking methodology for the direct selective reduction of citral to citronellol, addressing long-standing challenges in chemoselectivity and process economics. Traditional hydrogenation routes often struggle to differentiate between the multiple unsaturated bonds present in the citral molecule, leading to over-reduced byproducts such as dihydrocitronellol or incomplete conversion. This patent discloses a sophisticated four-phase reaction system utilizing a novel M2 type metal skeletal alloy catalyst, which achieves a remarkable conversion rate exceeding 99.3% and a selectivity greater than 96%. By integrating a specific combination of non-polar solvents and aqueous amine solutions, the process effectively manipulates the molecular orientation of the substrate at the catalyst interface. This technical breakthrough offers a compelling value proposition for manufacturers seeking to optimize their flavor & fragrance intermediates supply chain while adhering to stricter environmental and cost constraints.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of citronellol has relied heavily on catalysts containing precious metals or toxic heavy elements, presenting significant operational and regulatory hurdles. Prior art, such as methods utilizing copper chromite or chromium-modified Raney nickel, often suffers from severe environmental liabilities due to the presence of hexavalent chromium, necessitating complex and costly waste treatment protocols to meet modern discharge standards. Furthermore, processes employing noble metals like Ruthenium or Palladium, while effective, impose a substantial financial burden on production budgets due to the high volatility and intrinsic cost of these materials. Another critical limitation in existing technologies is the difficulty in controlling selectivity; high-temperature conditions required by some older catalysts frequently promote the hydrogenation of the isolated double bond in the hydrophobic tail, resulting in the formation of dihydrocitronellol, a less valuable byproduct that complicates downstream purification. These inefficiencies collectively erode profit margins and introduce supply chain risks associated with the sourcing of specialized, often hazardous, catalytic materials.
The Novel Approach
The methodology described in CN102295531B represents a paradigm shift by replacing expensive noble metals with a cost-effective M2 skeletal alloy composed of Cobalt, Nickel, Molybdenum, and Aluminum. This novel catalyst system operates within a unique hydrogen-oil-water-catalyst four-phase environment, which fundamentally alters the reaction kinetics to favor the desired product. By introducing a non-polar solvent, the process creates a protective microenvironment around the hydrophobic terminus of the citral molecule, physically shielding the isolated carbon-carbon double bond from hydrogenation. Simultaneously, the addition of an aqueous amine solution enhances the interaction between the hydrophilic carbonyl end of the molecule and the catalyst surface dispersed in the water phase. This strategic spatial arrangement ensures that hydrogenation occurs selectively at the conjugated double bond and the carbonyl group, preserving the structural integrity required for high-quality citronellol. The result is a robust, scalable process that delivers purity levels previously attainable only through far more expensive catalytic routes.
Mechanistic Insights into M2 Skeletal Alloy Catalyzed Selective Hydrogenation
The core innovation of this technology lies in the precise engineering of the interfacial interactions within the multiphase reaction system. Citral is an amphiphilic molecule, possessing a hydrophilic carbonyl group at one end and a hydrophobic hydrocarbon chain with an isolated double bond at the other. In the absence of phase modifiers, the molecule adsorbs randomly onto the catalyst surface, leading to non-selective reduction. However, the patented process leverages density differences and solubility parameters to enforce a specific orientation. The M2 catalyst, being denser, resides primarily in the aqueous phase along with the amine promoter. The citral is dissolved in a light petroleum hydrocarbon solvent, forming an oil phase that floats above the water. Under agitation, oil droplets are dispersed into the water layer, but the non-polar solvent preferentially solvates the hydrophobic tail of the citral molecule. This effectively 'hides' the terminal double bond within the oil droplet, while the polar carbonyl head group extends into the aqueous phase where the active catalytic sites are located.
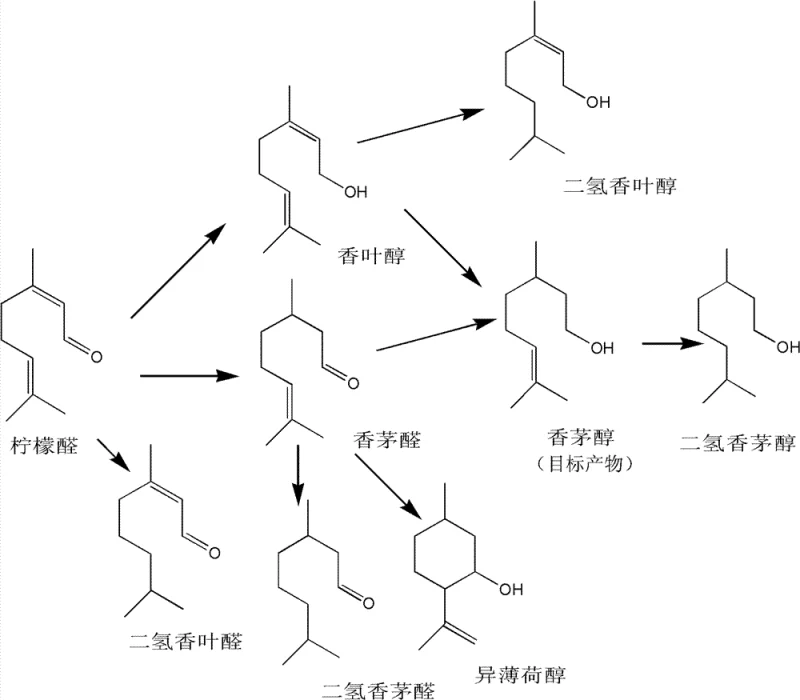
This mechanistic control is visually represented in the reaction network, where the pathway to citronellol is favored over the fully saturated dihydrocitronellol or the isomerized geraniol. The M2 alloy itself, prepared by leaching aluminum from a fused Co-Ni-Mo-Al precursor, possesses a high surface area and specific electronic properties that further enhance the activation of hydrogen and the carbonyl group. The amine component in the water phase plays a dual role: it adjusts the pH to optimize catalyst stability and acts as a co-catalyst that strengthens the adsorption of the carbonyl oxygen. This synergistic effect allows the base metal catalyst to mimic the high selectivity typically associated with precious metals like Ruthenium. Consequently, the reaction proceeds with minimal formation of over-reduced impurities, simplifying the subsequent vacuum rectification step and significantly improving the overall yield of the target synthetic flavors & fragrances product.
How to Synthesize Citronellol Efficiently
Implementing this advanced hydrogenation protocol requires careful attention to the preparation of the skeletal catalyst and the precise formulation of the four-phase reaction mixture. The process begins with the high-temperature fusion of the constituent metals followed by a controlled leaching process to generate the active porous structure. Once the catalyst is prepared and preserved under water, it is introduced into a standard hydrogenation autoclave along with the substrate, solvent, and amine promoter. The reaction is then conducted under moderate hydrogen pressure and temperature conditions, which are significantly milder than those required by traditional copper chromite methods. For a comprehensive understanding of the specific operating parameters, reagent ratios, and safety precautions necessary for reproducible results, please refer to the standardized synthesis guide below.
- Prepare the M2 catalyst by fusing Cobalt, Nickel, Molybdenum, and Aluminum, followed by alkaline leaching to remove aluminum and create a porous skeletal structure.
- Establish a four-phase reaction system comprising citral, a non-polar solvent, an aqueous amine solution, and the solid catalyst in a hydrogenation autoclave.
- Conduct batch hydrogenation at temperatures between 40-120°C and hydrogen pressures of 0.3-6.0 MPa to achieve selective reduction of the carbonyl and conjugated double bond.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this M2 catalyst technology translates into tangible strategic benefits beyond mere technical performance. The elimination of precious metals like Ruthenium or Palladium from the catalyst formulation immediately decouples production costs from the volatile fluctuations of the noble metal market, providing greater financial predictability for long-term contracts. Furthermore, the removal of toxic chromium species from the process stream drastically reduces the complexity and expense of wastewater treatment and hazardous waste disposal, aligning the manufacturing process with increasingly stringent global environmental regulations. The robustness of the M2 catalyst also implies a longer operational lifespan and the potential for multiple reuse cycles without significant loss of activity, which further drives down the cost per kilogram of the final product. These factors collectively enhance the resilience of the supply chain against raw material shortages and regulatory shocks.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with a base metal M2 alloy fundamentally alters the cost structure of citronellol production. By removing the need for high-value ruthenium or palladium, manufacturers can achieve substantial savings on catalyst procurement and regeneration. Additionally, the high selectivity of the process minimizes the loss of raw material citral to unwanted byproducts, thereby improving the overall atom economy and reducing the cost of goods sold. The simplified downstream processing, resulting from a cleaner reaction profile, also lowers energy consumption during distillation, contributing to a leaner and more cost-efficient manufacturing operation.
- Enhanced Supply Chain Reliability: Reliance on scarce precious metals often introduces supply chain bottlenecks, as the availability of materials like ruthenium can be constrained by geopolitical factors and mining outputs. Transitioning to a catalyst based on abundant metals such as nickel, cobalt, and aluminum mitigates these risks, ensuring a more stable and continuous supply of critical production inputs. The ability to prepare the catalyst in-house or source it from a broader range of suppliers further diversifies the supply base, reducing dependency on single-source vendors and enhancing the overall security of the flavor & fragrance intermediates supply chain.
- Scalability and Environmental Compliance: The batch nature of this hydrogenation process, combined with the use of standard autoclave equipment, facilitates straightforward scale-up from pilot plants to full commercial production volumes. The absence of hazardous heavy metals simplifies the permitting process for new manufacturing facilities and reduces the liability associated with environmental compliance. This ease of scaling, coupled with the green chemistry principles inherent in the high atom economy and reduced waste generation, positions this technology as a sustainable choice for meeting growing global demand without compromising on regulatory standards or corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selective hydrogenation technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of adopting this route for your specific production needs. Understanding these nuances is critical for R&D teams planning process validation and for procurement specialists assessing vendor capabilities.
Q: How does the four-phase reaction system improve selectivity in citronellol synthesis?
A: The system utilizes the amphiphilic nature of citral. A non-polar solvent protects the hydrophobic terminal double bond, while an aqueous amine phase exposes the hydrophilic carbonyl group to the catalyst, ensuring only specific bonds are reduced.
Q: What are the economic advantages of the M2 skeletal alloy catalyst over noble metals?
A: The M2 catalyst is composed of abundant base metals like Nickel and Cobalt, eliminating the high procurement costs and supply volatility associated with Ruthenium or Palladium, while maintaining comparable selectivity performance.
Q: Is this hydrogenation process suitable for large-scale industrial production?
A: Yes, the process operates in standard batch autoclaves with robust catalyst recovery. The absence of toxic heavy metals like Chromium simplifies waste treatment and regulatory compliance for mass manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citronellol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102295531B are fully realized in practical, industrial settings. Our rigorous QC labs and stringent purity specifications guarantee that every batch of citronellol meets the exacting standards required by the global fragrance and flavor industry. We are committed to delivering high-purity intermediates that empower our clients to formulate superior end-products with confidence and consistency.
We invite you to engage with our technical procurement team to discuss how this innovative hydrogenation technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this M2 catalyst system. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your volume requirements, allowing us to demonstrate our commitment to driving efficiency and quality in the commercial scale-up of complex flavor & fragrance intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
