Revolutionizing N-Phenylmaleimide Production for High-Performance Polymer Applications
Revolutionizing N-Phenylmaleimide Production for High-Performance Polymer Applications
The global demand for high-performance engineering plastics, particularly heat-resistant ABS resins, has driven an urgent need for more efficient and sustainable manufacturing routes for key modifiers like N-phenylmaleimide. As detailed in the groundbreaking patent CN103664732A, a novel synthetic methodology has emerged that fundamentally shifts the production paradigm from wasteful, multi-step batch processes to a streamlined, catalytic one-pot system. This technological leap addresses critical pain points in the fine chemical industry, offering a pathway to significantly lower production costs while enhancing product quality and environmental compliance. By leveraging advanced solid acid catalysis, this method not only improves reaction kinetics but also simplifies downstream processing, making it an ideal candidate for large-scale industrial adoption by forward-thinking chemical manufacturers.
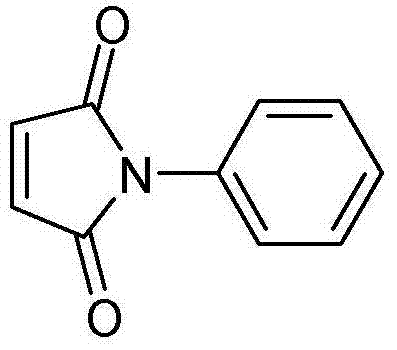
N-phenylmaleimide serves as a vital heat-proof modifier, capable of elevating the heat distortion temperature of ABS resin to the 125~130 ℃ range, thereby allowing it to substitute for more expensive engineering plastics in automotive and electronic applications. The structural integrity and purity of this molecule are paramount for its performance in polymer matrices. Consequently, the shift towards a cleaner, higher-yielding synthesis route described in the patent represents a strategic opportunity for supply chain optimization. For R&D directors and procurement managers alike, understanding the mechanistic advantages of this solid-acid catalyzed route is essential for securing a reliable supply of high-purity intermediates that meet the rigorous specifications of the modern polymer industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of N-phenylmaleimide has been plagued by inefficient, environmentally burdensome processes that struggle to meet modern green chemistry standards. One prevalent traditional method, often referred to as the two-step acetic anhydride method, involves the initial formation of N-phenylmaleamic acid followed by a dehydration step using excessive amounts of acetic anhydride and sodium acetate. As illustrated in the reaction scheme below, this approach is inherently flawed due to its reliance on stoichiometric dehydrating agents rather than catalytic ones.
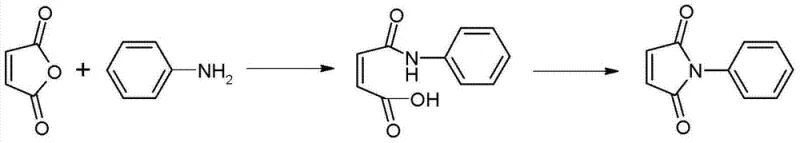
The drawbacks of this conventional acetic anhydride route are multifaceted and severe. Firstly, the operation steps are lengthy and complex, requiring the isolation of the intermediate acid before proceeding to cyclization, which drastically reduces overall throughput. Secondly, the yield is suboptimal, typically hovering around 82.32%, leading to significant material loss. Most critically from an environmental and cost perspective, the massive consumption of acetic anhydride generates substantial volumes of acidic wastewater during the workup phase. This not only inflates the cost of raw materials but also imposes a heavy burden on wastewater treatment facilities, rendering the process economically unviable for large-scale operations in regions with strict environmental regulations.
The Novel Approach
In stark contrast to these legacy methods, the innovative process disclosed in patent CN103664732A introduces a highly efficient one-pot synthesis strategy that utilizes maleic anhydride and aniline directly in an aromatic solvent system. This novel approach eliminates the need for isolating intermediates and replaces corrosive liquid acids or stoichiometric dehydrating agents with a reusable solid acid catalyst. The reaction proceeds under reflux conditions where water is continuously removed azeotropically, driving the equilibrium towards the desired imide product with exceptional efficiency.
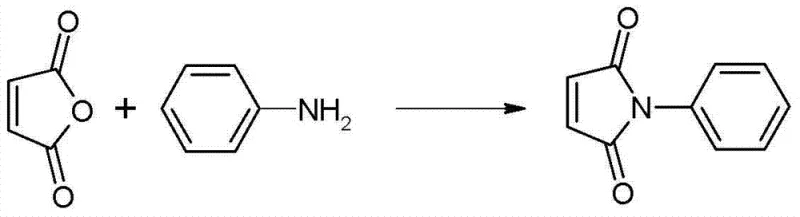
The superiority of this new method lies in its ability to transform a heterogeneous or difficult liquid-solid reaction into a manageable homogeneous-like process within the solvent phase. By employing aromatic hydrocarbons such as toluene or xylene as both solvent and water-entraining agents, the system achieves rapid dehydration without the need for external chemical dehydrating agents. The result is a dramatic reduction in reaction time—down to approximately 3 hours compared to the 6 hours or more required by older methods—and a significant boost in yield to the 90-92% range. This streamlined workflow not only enhances productivity but also drastically simplifies the post-reaction workup, as the solid catalyst can be simply filtered off and reused.
Mechanistic Insights into Solid Acid Catalyzed Cyclodehydration
The core of this technological breakthrough is the utilization of macroporous strong acidic ion exchange resins, such as D001 resin or perfluorinated sulfonic resins, as heterogeneous catalysts. Mechanistically, these solid acids provide abundant proton sources (-SO3H groups) on their surface and within their pore structures, which activate the carbonyl oxygen of the maleic anhydride. This activation facilitates the nucleophilic attack by the amino group of aniline, leading to the formation of the N-phenylmaleamic acid intermediate in situ. Unlike traditional liquid acids which remain dissolved and require neutralization, the solid nature of the catalyst allows for a clean separation post-reaction.
Furthermore, the inclusion of specific polymerization inhibitors, such as 2,6-di-tert-butyl-p-cresol or resorcinol, plays a critical role in maintaining product quality. During the high-temperature reflux (80-160 ℃), there is a risk of radical polymerization of the maleimide double bond, which would lead to oligomeric impurities and reduced yield. The inhibitor scavenges these free radicals effectively, ensuring that the reaction pathway remains selective for the cyclodehydration to the monomeric imide. This precise control over side reactions is what enables the process to consistently achieve purity levels exceeding 98.5%, a critical specification for applications in high-end optical and electronic materials.
How to Synthesize N-Phenylmaleimide Efficiently
Implementing this advanced synthesis route requires careful attention to the molar ratios of reactants and the specific choice of catalyst to maximize efficiency. The patent outlines a robust protocol where maleic anhydride and aniline are reacted in a molar ratio of roughly 1.2:1, ensuring complete conversion of the amine while minimizing excess anhydride waste. The process is designed to be scalable, moving seamlessly from laboratory glassware to industrial reactors with minimal modification to the core parameters.
- Charge maleic anhydride, aromatic solvent (toluene/xylene), solid acid catalyst (e.g., D001 resin), and a polymerization inhibitor into a reactor equipped with a water separator.
- Heat to reflux and dropwise add aniline over 0.5-1 hour, maintaining reflux to remove water azeotropically for 0.5-2.5 hours.
- Filter to recover the solid catalyst, wash the organic phase with alkali solution, and distill off the solvent to isolate high-purity N-phenylmaleimide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this solid-acid catalyzed process offers profound economic and logistical benefits that extend far beyond simple yield improvements. The elimination of acetic anhydride as a reagent removes a major cost driver and a significant source of hazardous waste, directly impacting the bottom line through reduced raw material expenditure and lower waste disposal fees. Additionally, the ability to recover and reuse the aromatic solvent and the solid catalyst creates a closed-loop system that minimizes resource consumption and stabilizes long-term production costs against market volatility.
- Cost Reduction in Manufacturing: The replacement of stoichiometric dehydrating agents with a catalytic amount of reusable solid acid fundamentally alters the cost structure of N-phenylmaleimide production. By avoiding the purchase of expensive acetic anhydride and the associated costs of neutralizing acidic waste streams, manufacturers can achieve substantial cost savings. Furthermore, the high yield of 90-92% means that less raw material is required per kilogram of finished product, maximizing the return on investment for every ton of maleic anhydride and aniline purchased.
- Enhanced Supply Chain Reliability: The simplicity of the one-pot process reduces the risk of production bottlenecks associated with multi-step syntheses that require intermediate isolation and purification. With a total reaction cycle time of approximately 3 hours, facility throughput is significantly increased, allowing suppliers to respond more rapidly to fluctuating market demands. The use of common, commodity-grade solvents like toluene and xylene ensures that supply chain disruptions due to specialized reagent shortages are virtually eliminated.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent globally, the ability to operate a process with minimal wastewater generation is a decisive competitive advantage. This method produces negligible acidic waste compared to traditional routes, simplifying compliance with environmental protection laws. The scalability is further enhanced by the ease of catalyst filtration and solvent recovery, making it straightforward to ramp up production from pilot scale to hundreds of tons annually without encountering the engineering challenges typical of complex multiphase reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of what partners can expect when adopting this methodology for their supply chains.
Q: What are the primary advantages of the solid acid catalyst method over traditional acetic anhydride dehydration?
A: The solid acid catalyst method eliminates the need for stoichiometric amounts of acetic anhydride, significantly reducing wastewater generation and raw material costs. Furthermore, the solid catalyst can be filtered and reused, unlike homogeneous acids which require neutralization and create salt waste.
Q: How does this synthesis route impact the purity of N-phenylmaleimide for ABS resin applications?
A: This optimized one-pot process achieves product purity greater than 98.5% with yields reaching 90-92%. The use of specific polymerization inhibitors prevents unwanted side reactions, ensuring the thermal stability required for high-performance engineering plastics.
Q: Is the aromatic solvent recoverable in this industrial process?
A: Yes, the process utilizes aromatic hydrocarbons like toluene or xylene which form an azeotrope with water. After the reaction and filtration steps, the solvent is easily recovered via distillation and recycled, minimizing environmental impact and operational expenses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Phenylmaleimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing processes like the solid-acid catalyzed synthesis of N-phenylmaleimide requires a partner with deep technical expertise and proven industrial capability. As a leading CDMO and fine chemical manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this process, including high-temperature reflux systems and efficient solvent recovery units, ensuring that we can deliver product with stringent purity specifications and rigorous QC labs validation.
We invite procurement leaders and R&D teams to engage with us to explore how this optimized route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
