Revolutionizing Electronic Grade TGIC Production with Advanced Halogen-Free Catalytic Technology
The rapid evolution of the global electronics industry has placed unprecedented demands on the purity and reliability of foundational chemical materials, particularly for applications in printed circuit boards (PCBs) and photo-imaging solder resist inks. Patent CN108947987B introduces a groundbreaking methodology for the production of high-quality electronic grade triglycidyl isocyanurate (TGIC), addressing critical bottlenecks related to halogen contamination and process inefficiency. This technical disclosure outlines a sophisticated synthesis route that replaces traditional halogenated phase transfer catalysts with advanced organic strong bases, fundamentally altering the impurity profile of the final product. For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, this innovation represents a pivotal shift towards sustainable, high-performance manufacturing. By integrating a vacuum-assisted alkali addition strategy, the process effectively mitigates the hydrolysis of epichlorohydrin, a common side reaction that plagues conventional methods and degrades product quality. The result is a robust, scalable protocol that delivers exceptional epoxy values while adhering to stringent environmental regulations, positioning it as a superior choice for next-generation electronic material supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of TGIC has relied heavily on two-step routes utilizing catalysts such as benzyltriethylammonium chloride, which introduce significant challenges for high-end electronic applications. The primary drawback of these legacy systems is the inevitable residue of halogen ions, specifically chlorine, which persists through purification stages and compromises the dielectric integrity of cured coatings. Furthermore, the cyclization phase in traditional processes often involves the coexistence of alkaline water, creating a thermodynamic environment conducive to the hydrolysis of epichlorohydrin and the undesirable ring-opening of the TGIC molecule itself. This leads to a complex impurity spectrum that requires extensive and costly downstream processing to mitigate. Additionally, conventional crystallization protocols mandate the use of large volumes of methanol as a cosolvent, introducing severe safety hazards due to methanol's volatility and flammability. The recovery and recycling of this solvent impose a heavy energy burden on the facility, while fugitive emissions contribute to substantial environmental pollution, making these older methods increasingly untenable in a regulatory landscape focused on green chemistry and carbon footprint reduction.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a halogen-free organic strong base catalyst, such as tetramethylammonium hydroxide or 1,8-diazabicyclo[5.4.0]undec-7-ene, to drive the addition ring-opening reaction with exceptional selectivity. This strategic substitution eliminates the source of halogen contamination at the molecular level, ensuring that the final electronic grade TGIC is free from corrosive ions that could damage sensitive microelectronics. The process innovates further by implementing a vacuum-assisted alkali addition technique during the cyclization stage, where flake caustic soda is introduced at controlled temperatures between 35°C and 40°C under negative pressure. This engineering control effectively removes water generated during the reaction in real-time, preventing the formation of alkaline aqueous phases that trigger hydrolysis. Moreover, the crystallization step is re-engineered to utilize the residual epichlorohydrin from the reaction mixture as the crystallization medium, completely obviating the need for external methanol addition. This not only simplifies the unit operations but also drastically reduces solvent consumption and waste generation, offering a compelling value proposition for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Organic Base-Catalyzed Cyclization
The core of this technological advancement lies in the precise manipulation of nucleophilic substitution and ring-closure mechanisms facilitated by the organic strong base. In the initial stage, the organic base activates the nitrogen atoms of the cyanuric acid ring, enhancing their nucleophilicity towards the epichlorohydrin molecules. This promotes a clean addition reaction where the epoxide ring of the epichlorohydrin remains intact while the chlorohydrin intermediate is formed efficiently. Unlike quaternary ammonium salts that may decompose or leave ionic residues, the selected organic bases are thermally stable and can be managed to minimize entrapment within the crystal lattice. The subsequent cyclization is a delicate intramolecular nucleophilic substitution where the hydroxyl group attacks the adjacent carbon-chlorine bond to close the epoxide ring. The presence of water is the enemy here, as it competes as a nucleophile, leading to diol byproducts. By maintaining a high vacuum during the caustic soda addition, the system continuously strips away water vapor, shifting the equilibrium towards the desired triglycidyl product and suppressing the kinetic pathways that lead to ring-opened impurities.
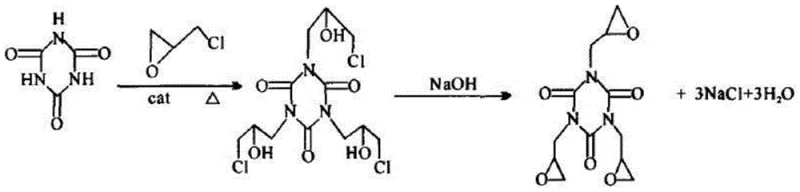
Furthermore, the control of impurity profiles is achieved through the rigorous management of reaction thermodynamics and phase behavior. The absence of halogenated catalysts means that the ionic strength of the reaction medium is lower, reducing the likelihood of salt inclusion in the final crystal structure. The vacuum distillation step is calibrated to remove only a specific fraction (30-40%) of the excess epichlorohydrin, leaving enough solvent to maintain a homogeneous solution upon cooling. As the temperature drops to 8-10°C, the solubility of TGIC decreases sharply, prompting the formation of high-purity crystals while soluble impurities and residual salts remain in the mother liquor. This fractional crystallization effect, driven by the specific solvent properties of epichlorohydrin rather than methanol, ensures that the solid phase obtained after centrifugation possesses the narrow particle size distribution and high epoxy value required for electronic grade specifications. The microwave drying final step further ensures that no thermal degradation occurs, preserving the integrity of the sensitive epoxide groups.
How to Synthesize Triglycidyl Isocyanurate Efficiently
The synthesis of high-purity TGIC via this patented route requires precise control over reaction parameters, particularly temperature ramps and vacuum levels, to maximize yield and minimize byproduct formation. The process begins with the homogeneous mixing of cyanuric acid, epichlorohydrin, the organic strong base catalyst, and a controlled amount of water in a synthesis reactor. Detailed standard operating procedures regarding stoichiometry, heating rates, and vacuum thresholds are essential for reproducibility at scale. For a comprehensive understanding of the specific operational parameters and safety protocols required to implement this technology in a pilot or commercial plant, please refer to the standardized synthesis guide below.
- Mix cyanuric acid, epichlorohydrin, organic strong base, and water, reacting at 75-80°C followed by a 100°C hold to form the intermediate.
- Transfer to a cyclization kettle, apply vacuum, cool to 35-40°C, and slowly add flake caustic soda to induce ring closure without hydrolysis.
- Distill off partial epichlorohydrin under vacuum, then utilize the remaining solvent for low-temperature crystallization to isolate the final electronic grade product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel TGIC synthesis route offers transformative advantages that extend far beyond simple chemical yield improvements. The elimination of methanol from the crystallization process represents a massive reduction in raw material costs and logistical complexity, as there is no longer a need to procure, store, and recover large volumes of this hazardous solvent. This simplification of the material flow directly translates to enhanced supply chain reliability, as the production process becomes less dependent on external solvent markets and more self-contained within the reaction mass balance. Furthermore, the removal of halogenated catalysts simplifies the wastewater treatment profile, reducing the load on effluent treatment plants and lowering the costs associated with environmental compliance and hazardous waste disposal. These factors combine to create a manufacturing process that is not only more economically efficient but also significantly more resilient to regulatory changes and raw material price volatility.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic reduction in solvent usage and energy consumption. By eliminating the methanol crystallization step, the facility avoids the capital and operational expenditures associated with methanol recovery distillation columns and explosion-proof storage infrastructure. Additionally, the organic strong base catalyst, while potentially higher in unit cost than simple salts, is used in catalytic quantities and contributes to a cleaner product that requires less refining, thereby lowering the overall cost per ton of qualified electronic grade material. The energy savings are substantial because the process avoids the high latent heat requirements of evaporating large volumes of methanol, focusing thermal energy solely on the reaction and the recovery of epichlorohydrin, which is recycled back into the process.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this method ensures consistent delivery of high-specification materials. Traditional methods often suffer from batch-to-batch variability due to the sensitivity of the cyclization step to water content; the vacuum alkali addition technique provides a much tighter control window, ensuring that every batch meets the strict epoxy value and halogen content specifications required by PCB manufacturers. This consistency reduces the risk of downstream production failures for customers, strengthening the supplier-customer relationship. Moreover, the use of widely available raw materials like cyanuric acid and flake caustic soda, combined with a generic class of organic bases, ensures that the supply chain is not vulnerable to shortages of exotic or highly regulated reagents, guaranteeing long-term continuity of supply.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing standard unit operations such as vacuum distillation and centrifugation that are well-understood in the fine chemical industry. The environmental benefits are profound, as the process generates significantly less hazardous waste and VOC emissions compared to methanol-based routes. This aligns perfectly with global trends towards green manufacturing and helps customers meet their own Scope 3 emission targets by sourcing cleaner intermediates. The absence of halogen residues also means the final product is compliant with increasingly strict international regulations regarding halogen-free electronics, opening up premium market segments that are inaccessible to producers using legacy technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this halogen-free TGIC synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is crucial for evaluating the feasibility of adopting this route for your specific application needs.
Q: Why is a halogen-free catalyst critical for electronic grade TGIC?
A: Traditional catalysts like benzyltriethylammonium chloride leave chlorine residues that cause corrosion and reliability issues in printed circuit boards (PCBs). The halogen-free organic base ensures zero halogen ion residue, meeting strict electronic material standards.
Q: How does the vacuum alkali addition process improve yield?
A: Adding alkali under vacuum at controlled temperatures (35-40°C) prevents the formation of alkaline water, which typically causes epichlorohydrin hydrolysis and unwanted TGIC ring-opening side reactions, thereby significantly boosting product purity and yield.
Q: What are the cost benefits of eliminating methanol crystallization?
A: By utilizing residual epichlorohydrin for cooling crystallization instead of adding large volumes of methanol, the process eliminates solvent recovery costs, reduces volatile organic compound (VOC) emissions, and lowers overall energy consumption for distillation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triglycidyl Isocyanurate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced electronic materials requires a partner who understands both the intricate chemistry and the rigorous demands of industrial scale-up. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN108947987B are fully realized in practical manufacturing environments. We are committed to delivering high-purity Triglycidyl Isocyanurate that meets stringent purity specifications, leveraging our rigorous QC labs to verify every batch for halogen content, epoxy value, and particle size distribution. Our capability to handle complex catalytic systems and vacuum-sensitive operations allows us to offer a supply of electronic grade TGIC that is both consistent and cost-effective, bridging the gap between laboratory innovation and commercial reality.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your product performance and reduce your total cost of ownership. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate precisely how our manufacturing capabilities align with your project milestones and quality standards.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
