Advanced Purification Technology for Gemcitabine Hydrochloride: Ensuring Clinical Safety and Commercial Scalability
The landscape of oncology drug manufacturing demands uncompromising standards for purity and safety, particularly for potent antimetabolites like Gemcitabine Hydrochloride. As detailed in patent CN102659884B, a novel purification methodology has been established that significantly elevates the quality profile of this critical antitumor agent. This technology addresses the persistent challenges associated with removing stereoisomeric impurities and residual organics that often plague conventional synthesis routes. By integrating a precise acid-base liberation step followed by activated carbon adsorption and high-resolution preparative chromatography, manufacturers can achieve a refined product with exceptional pharmaceutical grade specifications. This report analyzes the technical merits of this approach, demonstrating how it serves as a robust foundation for reliable pharmaceutical intermediates supplier networks aiming to deliver high-quality active ingredients to the global market.
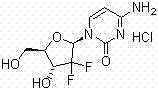
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Gemcitabine Hydrochloride, often relying on D-glyceraldehyde acetonide and bromo difluoro acetate ethyl ester as starting materials, face significant hurdles during the final purification stages. The core issue lies in the glycosylation step where two (trimethylsilyl) cytosines are condensed onto the ribose sugar. This reaction inherently produces a mixture of alpha and beta anomers due to the simultaneous occurrence of SN1 and SN2 nucleophilic substitution mechanisms. Conventional crystallization techniques frequently struggle to completely separate these stereoisomers, leading to crude products with suboptimal purity profiles. Furthermore, the presence of colored impurities and residual solvents from the complex multi-step synthesis requires rigorous downstream processing. Without an advanced purification strategy, these impurities pose potential safety hazards for clinical application, necessitating a more sophisticated approach to ensure the therapeutic efficacy and safety of the final drug substance.
The Novel Approach
The patented method introduces a streamlined yet highly effective three-stage purification protocol that overcomes the deficiencies of standard recrystallization. Initially, the process employs a controlled acid-base reaction where the crude hydrochloride salt is dissolved in water and the pH is adjusted to an alkaline range of 8 to 9. This crucial step liberates the free base of Gemcitabine, allowing for the separation of water-soluble inorganic salts and acidic impurities. Subsequently, the free base is subjected to activated carbon treatment in an alcoholic solution, which effectively adsorbs colored impurities and high molecular weight organic byproducts. The final and most critical stage involves preparative chromatography using a specifically tuned mobile phase of acetone or acetonitrile mixed with hydrochloric acid. This system provides the resolution necessary to separate the target beta-anomer from the unwanted alpha-anomer, resulting in a highly refined product that meets the strictest regulatory standards for antitumor medications.
Mechanistic Insights into Chromatographic Separation and Impurity Control
The success of this purification strategy hinges on the precise manipulation of physicochemical properties during the chromatographic separation phase. The stationary phase, composed of silica gel or aluminum oxide, interacts differentially with the Gemcitabine anomers based on their polarity and spatial configuration. The mobile phase, consisting of a volume ratio of 30-50 parts acetone or acetonitrile to 70-50 parts hydrochloric acid solution (pH 2-3), plays a dual role. Firstly, the acidic environment ensures that the Gemcitabine remains in its stable hydrochloride salt form during elution, preventing degradation. Secondly, the organic modifier adjusts the polarity to optimize the retention time difference between the alpha and beta isomers. This fine-tuned balance allows for the baseline separation of the target compound from structurally similar impurities that co-elute in less optimized systems. The result is a product stream that is chemically homogeneous, minimizing the risk of immunogenic responses or off-target effects in patients.
Impurity control is further enhanced by the preceding activated carbon adsorption step, which acts as a broad-spectrum scavenger for trace organic contaminants. By dissolving the intermediate in alcohol at a concentration of 0.1-0.2g/mL and treating it with 4.8% to 5% activated carbon at temperatures between 20°C and 30°C, the process effectively removes conjugated systems responsible for coloration. This step is vital not only for aesthetic quality but also for reducing the load on the chromatography column, thereby extending its operational life and maintaining consistent separation efficiency over multiple batches. The combination of physical adsorption and chemical partitioning creates a synergistic effect that drives the final HPLC purity to levels exceeding 99.9%, ensuring that the high-purity pharmaceutical intermediates produced are safe for human administration.
How to Synthesize Gemcitabine Hydrochloride Efficiently
The synthesis and purification of Gemcitabine Hydrochloride require a disciplined approach to process parameters to ensure reproducibility and high yield. The patented workflow outlines a clear sequence of operations that transforms a crude reaction mixture into a clinical-grade API. Operators must strictly adhere to the specified pH ranges, solvent ratios, and temperature controls to maximize the recovery of the active ingredient while minimizing the formation of degradation products. The following guide summarizes the critical operational steps derived from the patent examples, providing a roadmap for implementing this technology in a GMP-compliant environment. For a comprehensive breakdown of the specific equipment setup and quality control checkpoints, please refer to the detailed technical documentation provided below.
- Dissolve crude Gemcitabine Hydrochloride in water, adjust pH to 8-9 using alkali to liberate the free base, and extract.
- Dissolve the free base in alcohol (0.1-0.2g/mL), treat with activated carbon at 20-30°C, and filter to remove organic impurities.
- Separate the filtrate using a preparative chromatography column with silica or alumina stationary phase and an acetone/acetonitrile-HCl mobile phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology offers tangible benefits that extend beyond mere technical compliance. The process is designed with industrial scalability in mind, utilizing common solvents and standard equipment that are readily available in the global chemical market. This reduces dependency on exotic reagents that might suffer from supply chain disruptions or volatile pricing. Furthermore, the high yield reported in the patent embodiments indicates efficient material utilization, which directly correlates to lower raw material costs per kilogram of finished product. By simplifying the purification train to three distinct and manageable unit operations, manufacturers can reduce cycle times and increase throughput, addressing the growing global demand for this essential oncology medication without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The elimination of complex derivatization steps often required in older purification methods leads to a significant reduction in operational expenditures. By relying on straightforward acid-base extraction and standard chromatography, the process minimizes the consumption of expensive chiral resolving agents or specialized catalysts. This streamlined approach lowers the overall cost of goods sold (COGS), allowing for more competitive pricing strategies in the generic pharmaceutical market. Additionally, the high recovery rates observed in the patent examples suggest that waste generation is minimized, further contributing to cost efficiency through reduced disposal fees and improved atom economy.
- Enhanced Supply Chain Reliability: The robustness of this method ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream drug product manufacturers. The use of stable and non-hazardous solvents like acetone and ethanol simplifies logistics and storage requirements, reducing the risk of delays associated with the transportation of regulated chemicals. Moreover, the scalability of the chromatographic step means that production capacity can be easily expanded to meet surge demands or long-term contract obligations. This reliability makes the manufacturer a preferred partner for multinational pharmaceutical companies seeking a dependable source of critical cancer therapeutics.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with green chemistry principles by avoiding the use of heavy metal catalysts or chlorinated solvents that pose significant disposal challenges. The aqueous workup in the initial step and the use of recyclable alcohols in the second step facilitate easier waste treatment and solvent recovery. This reduces the environmental footprint of the manufacturing facility and ensures compliance with increasingly stringent international environmental regulations. The ability to scale this process from laboratory benchtop to multi-ton commercial production without fundamental changes to the chemistry provides a clear pathway for rapid technology transfer and commercialization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the purification of Gemcitabine Hydrochloride. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature. They are intended to provide clarity on the feasibility, quality, and operational aspects of this manufacturing route for potential partners and stakeholders evaluating the technology for integration into their supply chains.
Q: How does this purification method address the issue of alpha/beta anomer impurities?
A: The method utilizes preparative chromatography with a specific mobile phase ratio (acetone or acetonitrile to hydrochloric acid solution at 30-50:70-50). This high-resolution separation effectively isolates the desired beta-anomer from the alpha-anomer and other structural isomers formed during the initial nucleophilic substitution synthesis.
Q: What is the expected purity level achievable with this process?
A: According to the patent data (CN102659884B), the implementation of this three-step purification protocol consistently yields Gemcitabine Hydrochloride with an HPLC purity of 99.9%, meeting stringent requirements for clinical antitumor applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial suitability. It relies on standard unit operations such as pH adjustment, filtration, and column chromatography using common solvents like acetone, acetonitrile, and alcohols, avoiding exotic reagents that would hinder scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemcitabine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality antitumor agents to the global healthcare community. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of Gemcitabine Hydrochloride meets or exceeds the 99.9% purity benchmark established by advanced purification technologies. Our infrastructure is designed to support the complex requirements of oncology API manufacturing, providing our clients with the confidence they need to bring life-saving medications to market.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized purification processes, we can help you achieve significant reductions in manufacturing costs while ensuring the highest standards of product safety. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to enhance your supply chain resilience and secure a steady supply of this vital pharmaceutical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
