Industrial Scale-Up of Gemcitabine Hydrochloride via Stable TBDPS Protection Strategy
Introduction to Advanced Gemcitabine Synthesis
The pharmaceutical industry continuously seeks robust synthetic routes for critical oncology agents, and the synthesis process detailed in patent CN101492482B represents a significant advancement in the industrial production of Gemcitabine Hydrochloride. This known compound, chemically defined as 2'-deoxy-2',2'-difluorocytidine hydrochloride, acts as a cell cycle-specific antimetabolite primarily targeting the DNA synthesis phase (S phase) of tumor cells. While existing technologies have established pathways for its creation, they often suffer from critical inefficiencies related to intermediate stability. The present invention addresses these deficiencies by introducing a novel protective group strategy that utilizes 2,3-O-isopropylidene-D-glyceraldehyde as the raw material. Through a sophisticated sequence involving addition, ring-opening, and cyclization, the process employs both TBDPSCl and benzoyl chloride to selectively protect hydroxyl groups. This dual-protection methodology not only enhances the chemical stability of the intermediates but also streamlines the downstream condensation with cytosine, ultimately yielding a high-purity final product suitable for rigorous clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the landscape of nucleoside analog manufacturing, prior art techniques for producing Gemcitabine Hydrochloride have been plagued by a specific chemical vulnerability: the instability of the intermediate double benzoyl protecting group. In traditional synthetic routes, the reliance on double benzoyl protection creates a fragile molecular architecture that is highly susceptible to deprotection under alkaline conditions. This instability is not merely a theoretical concern but a practical bottleneck that severely influences both product quality and overall process yield. When the protecting groups are prematurely removed or rearranged during the synthesis, it leads to the formation of difficult-to-remove impurities and side products. Consequently, manufacturers face significant challenges in purification, often requiring extensive chromatography or recrystallization steps that drive up costs and extend lead times. Furthermore, the inconsistency in intermediate stability translates to batch-to-batch variability, a critical risk factor for supply chain managers who require predictable output for reliable API intermediate supplier operations.
The Novel Approach
The innovative methodology outlined in the patent data fundamentally restructures the protection strategy to overcome these historical limitations. By adopting a mixed protection scheme where TBDPSCl is used to protect one of the hydroxyls while benzoyl chloride protects the other, the process dramatically increases the stability of the intermediate. The tert-butyldiphenylsilyl (TBDPS) group is renowned for its robustness against basic conditions compared to ester-based protectors, thereby preventing the premature deprotection that plagues conventional methods. This strategic modification facilitates the separation and purification of the product, as the intermediates remain intact throughout the harsh reaction conditions required for reduction and mesylation. The result is a streamlined workflow where the intermediate is condensed with cytosine after reduction and methyl sulfonylation, followed by a controlled deprotection and salification sequence. This approach effectively solves the problem of intermediate instability, ensuring that the final Gemcitabine Hydrochloride is produced with consistent high quality and improved yield metrics.
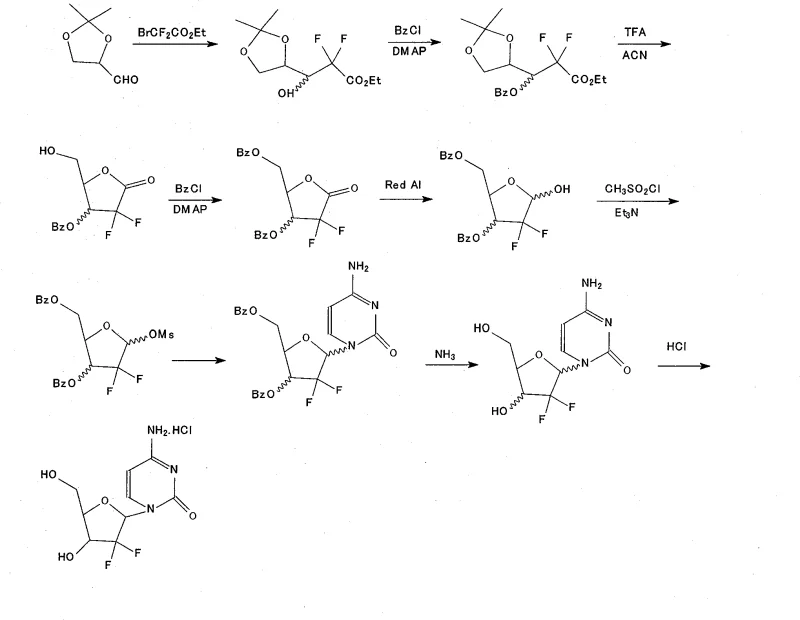
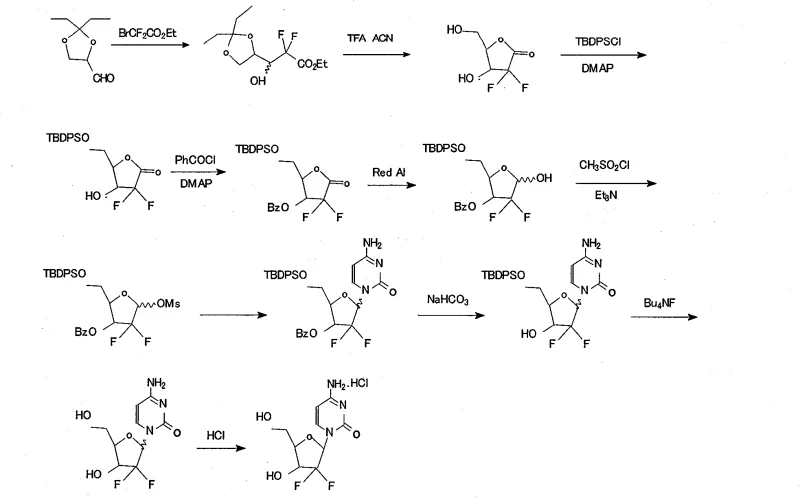
Mechanistic Insights into Selective Silylation and Cyclization
The core of this technological breakthrough lies in the precise orchestration of protective group chemistry and stereoselective transformations. The synthesis initiates with a Reformatsky-type addition reaction where activated zinc powder reacts with ethyl bromodifluoroacetate and 2,3-O-isopropylidene-D-glyceraldehyde. This step is critical for introducing the gem-difluoro motif, which is essential for the biological activity of the final drug. Following the addition, the process employs trifluoroacetic acid (TFA) in acetonitrile to induce cyclization and remove the isopropylidene group, generating the difluoro-furanose ketone. The mechanistic elegance continues with the selective protection steps. The primary hydroxyl group is first targeted by TBDPSCl in the presence of pyridine and DMAP, leveraging the steric accessibility of the primary alcohol. Subsequently, the secondary hydroxyl is benzoylated. This order of operations is vital; attempting to install the bulky silyl group after benzoylation could lead to steric clashes or incomplete reactions. The resulting 5-tert-butyl diphenyl silane-3-benzoyl intermediate serves as a robust scaffold for the subsequent reduction by Red-Al, which stereoselectively generates the ribofuranose configuration required for the natural nucleoside structure.
Furthermore, the mechanism ensures tight control over the impurity profile, which is a primary concern for R&D Directors focusing on purity specifications. The stability of the TBDPS group prevents the formation of deprotected byproducts during the mesylation step, where methanesulfonyl chloride activates the anomeric position. This activation is a prerequisite for the nucleophilic attack by the silylated cytosine base. In conventional routes, the lability of benzoyl groups during this activation can lead to competing reactions or degradation. However, in this novel pathway, the integrity of the sugar moiety is preserved until the deliberate deprotection stage. The final steps involve the removal of the benzoyl group using sodium bicarbonate in a fluorinated alcohol solvent, followed by the cleavage of the silyl ether using tetrabutylammonium fluoride (TBAF). This orthogonal deprotection strategy allows for the sequential removal of protecting groups without affecting the sensitive glycosidic bond or the difluoro substitution, ensuring that the final crystallization yields a product with purity above 99.8 percent.
How to Synthesize Gemcitabine Hydrochloride Efficiently
Executing this synthesis requires strict adherence to the reaction conditions and stoichiometry defined in the patent to maximize the 17.9 percent total yield. The process begins with the preparation of the difluoro ester, followed by cyclization to the lactone, and then the critical divergent step of selective protection. Operators must maintain precise temperature controls, such as keeping the benzoylation between 0~5 ℃ to prevent side reactions, and managing the reduction with Red-Al at sub-zero temperatures (-5 ℃) to ensure stereochemical fidelity. The coupling with cytosine requires heating to 80~85 ℃ in the presence of a silylating agent to facilitate the formation of the N-glycosidic bond. For a comprehensive breakdown of the specific reagent quantities, solvent volumes, and workup procedures required to replicate this high-purity synthesis, please refer to the standardized protocol below.
- Perform a Reformatsky-type addition reaction using activated zinc powder and ethyl bromodifluoroacetate with 2,3-O-isopropylidene-D-glyceraldehyde to form the difluoro ester intermediate.
- Execute acid-catalyzed cyclization and deprotection using trifluoroacetic acid (TFA) in acetonitrile to generate the difluoro-furanose ketone.
- Selectively protect the primary hydroxyl group using tert-butyldiphenylsilyl chloride (TBDPSCl) followed by benzoylation of the secondary hydroxyl with benzoyl chloride.
- Reduce the ketone to the ribofuranose alcohol using Red-Al, followed by mesylation to activate the anomeric position for nucleophilic substitution.
- Condense the activated sugar with silylated cytosine, followed by selective deprotection of the benzoyl group and final desilylation with TBAF to yield Gemcitabine Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this stabilized synthetic route offers tangible benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. Because the intermediates are significantly more stable, there is a reduced generation of degradation byproducts that typically necessitate expensive and time-consuming purification steps. This directly correlates to cost reduction in pharmaceutical intermediates manufacturing, as fewer resources are consumed in chromatography columns, solvents, and labor hours dedicated to reworking off-spec batches. Moreover, the robustness of the TBDPS-protected intermediate means that the process is more forgiving to minor fluctuations in reaction parameters, leading to higher batch success rates. This reliability is crucial for maintaining supply continuity, especially when scaling from pilot plant operations to multi-ton commercial production. The ability to consistently achieve purity levels above 99.8 percent minimizes the risk of downstream rejection by API manufacturers, thereby securing long-term contracts and reducing the financial liability associated with quality failures.
- Cost Reduction in Manufacturing: The elimination of unstable double benzoyl intermediates removes the need for complex stabilization protocols and reduces the loss of valuable materials due to decomposition. By preventing premature deprotection, the process ensures that the majority of the starting material is converted into the desired intermediate rather than waste. This efficiency gain translates into substantial cost savings, as the effective utilization of expensive reagents like Red-Al and specialized silylating agents is maximized. Additionally, the simplified workup procedures reduce the consumption of extraction solvents and drying agents, further lowering the operational expenditure per kilogram of produced API intermediate.
- Enhanced Supply Chain Reliability: The improved stability of the key intermediates directly enhances the predictability of the manufacturing schedule. In traditional processes, unexpected deprotection events can halt production lines for days while troubleshooting occurs. With this novel approach, the risk of such interruptions is significantly mitigated, allowing for tighter delivery windows and more reliable inventory planning. This stability is particularly beneficial for reducing lead time for high-purity pharmaceutical intermediates, as the consistent quality of the crude product allows for faster turnover in the quality control laboratory and quicker release for shipment to downstream customers.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial production, utilizing reagents and conditions that are amenable to large-scale reactor setups. The use of standard solvents like ethyl acetate, toluene, and methanol facilitates solvent recovery and recycling, aligning with modern green chemistry principles. Furthermore, the high selectivity of the reaction reduces the load on wastewater treatment systems by minimizing the discharge of complex organic impurities. This environmental compliance is increasingly becoming a prerequisite for vendor qualification by major multinational pharmaceutical companies, making this synthesis route a strategically sound choice for long-term supply partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios or for procurement specialists assessing the quality credentials of potential suppliers.
Q: Why is the TBDPS protection strategy superior to conventional double benzoyl protection for Gemcitabine synthesis?
A: Conventional double benzoyl protection is unstable under alkaline conditions, leading to premature deprotection which negatively impacts product quality and yield. The novel approach utilizes TBDPSCl to protect one hydroxyl group, significantly increasing the stability of the intermediate during subsequent alkaline processing steps, thereby facilitating easier separation and purification.
Q: What are the expected yield and purity metrics for this industrial synthesis process?
A: According to the patent data, the total yield of this optimized synthesis process is approximately 17.9 percent. Crucially for pharmaceutical applications, the final product achieves a purity level above 99.8 percent, meeting stringent requirements for active pharmaceutical ingredients (APIs).
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process is specifically designed for industrial production. By solving the stability issues associated with intermediate double benzoyl protectors, the method ensures consistent product quality and yield, making it particularly applicable for the commercial scale-up of complex nucleoside analogs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemcitabine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the production of life-saving oncology medications. Our technical team has extensively analyzed the protective group strategies outlined in recent patents, and we possess the expertise to implement these advanced methodologies at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of Gemcitabine Hydrochloride or related nucleoside analog meets the exacting standards required by global regulatory bodies. We are committed to delivering high-purity pharmaceutical intermediates that empower our partners to accelerate their drug development timelines.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how switching to this stabilized synthesis route can optimize your specific procurement budget. We encourage you to contact our technical procurement team today to request specific COA data for our current inventory and to discuss route feasibility assessments for your upcoming projects. Let us demonstrate how our commitment to chemical innovation and operational excellence can serve as the foundation for your continued success in the competitive pharmaceutical market.
