Scaling 2-Cyano-4'-Methylbiphenyl Production: A Technical Breakthrough in Ni-Mn Catalytic Coupling
The pharmaceutical industry's relentless pursuit of efficient Angiotensin II Receptor Blockers (ARBs) has placed immense strategic value on the reliable supply of key intermediates, specifically 2-cyano-4'-methylbiphenyl, widely known as Sartan Biphenyl or OTBN. As detailed in patent CN103467341A, a transformative preparation method has been developed that leverages a novel Nickel-Manganese composite catalytic system to overcome historical bottlenecks in yield and purity. This intermediate serves as the foundational scaffold for blockbuster drugs such as Losartan, Valsartan, and Telmisartan, which are critical for managing hypertension and cardiovascular diseases globally. The structural integrity of this molecule, characterized by a biphenyl core with a cyano group and a methyl substituent, is paramount for downstream biological activity.
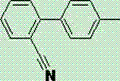
Beyond its pharmaceutical applications, this compound is increasingly recognized as a vital precursor in the synthesis of advanced liquid crystal materials, further diversifying its market demand. The patent discloses a robust synthetic pathway that initiates with a Grignard reaction using p-chlorotoluene and magnesium powder, followed by a crucial coupling step with o-chlorobenzonitrile. What sets this technology apart is the specific integration of Ni(II)/Mn(II) catalysts, which not only enhances reaction kinetics but also significantly simplifies the purification workflow. For R&D directors and procurement strategists, understanding the nuances of this catalytic innovation is essential for securing a competitive edge in the supply chain of high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sartan Biphenyl has been plagued by complex, multi-step protocols that impose severe economic and environmental burdens on manufacturers. One prominent conventional route, often referred to as the protection group multi-step method, relies on starting materials like o-methoxybenzoic acid or anisaldehyde. This archaic approach necessitates a Meyer reaction to generate an oxazoline protecting group, followed by a Grignard reaction with p-bromotoluene and a subsequent elimination of the methoxy anion. The inherent flaws in this methodology are manifold: it suffers from low total yields due to the accumulation of losses across multiple steps, generates substantial chemical waste, and requires expensive auxiliary materials and reagents. Furthermore, the equipment requirements for handling such diverse reaction conditions are stringent, rendering this method economically unviable for modern, large-scale industrial production where margin compression is a constant threat.
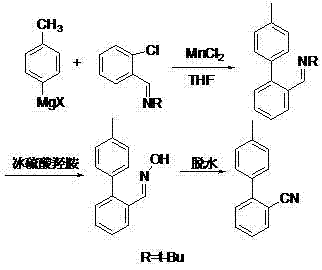
Another alternative, the aromatization ring-closing synthesis method described by Dormoy et al., attempts to streamline the process using imine derivatives. While this route reduces the number of steps compared to the protection group method, it still fails to deliver optimal efficiency, typically capping overall yields at approximately 65%. Such yield limitations translate directly into higher raw material consumption and increased waste disposal costs. Additionally, transition metal catalytic reduction methods utilizing systems like Pd(0) or Zn(II)/Ni(0) have been explored, but they often encounter issues with expensive catalyst costs, difficult recovery processes, and the formation of unwanted by-products that complicate downstream purification. These conventional limitations create a significant barrier to entry for new suppliers and constrain the supply continuity for major pharmaceutical clients.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN103467341A introduces a paradigm shift through the application of a Ni(II)/Mn(II) composite catalyst in a direct coupling strategy. This novel approach bypasses the need for cumbersome protecting groups and complex ring-closing maneuvers, opting instead for a direct cross-coupling between a Grignard reagent derived from p-chlorotoluene and o-chlorobenzonitrile. The strategic inclusion of the nickel-manganese dual catalyst system dramatically improves reaction activity and selectivity, enabling the process to achieve total yields exceeding 87% under optimized conditions. Crucially, this method allows for a simplified workup procedure where the reaction mixture is acidified and subjected to direct distillation, effectively eliminating the need for resource-intensive organic solvent extraction steps. This streamlining of the post-treatment phase not only reduces operational time but also minimizes the generation of hazardous waste, aligning perfectly with modern green chemistry principles and cost-reduction mandates.
Mechanistic Insights into Ni(II)/Mn(II) Composite Catalyzed Coupling
The core scientific breakthrough of this patent lies in the synergistic interaction between the Nickel and Manganese species within the catalytic cycle. Unlike traditional Palladium-catalyzed cross-couplings which rely on expensive ligands and rigorous exclusion of oxygen, the Ni(II)/Mn(II) system operates with remarkable robustness. The mechanism likely involves the in-situ reduction of the Ni(II) precursor to an active Ni(0) species by the Grignard reagent or the Mn(II) co-catalyst, which then undergoes oxidative addition with the aryl chloride of the o-chlorobenzonitrile. The presence of Manganese is hypothesized to facilitate the transmetallation step or stabilize the reactive nickel intermediates, thereby preventing the formation of homocoupling by-products which are common in nickel-only systems. This enhanced selectivity is critical for pharmaceutical intermediates, where impurity profiles must be tightly controlled to meet regulatory standards for downstream API synthesis.

Furthermore, the patent specifies precise temperature controls, ranging from 30-80°C for the Grignard formation and -20 to 20°C for the coupling step, which are instrumental in managing the exothermic nature of the reaction and suppressing side reactions. The choice of solvent, whether it be THF, toluene, or xylene, plays a pivotal role in solubilizing the organometallic species and ensuring efficient mass transfer. From an impurity control perspective, the final recrystallization step using solvents like ethyl acetate or petroleum ether is highly effective at removing residual metal salts and organic impurities, resulting in a white crystalline product with high purity. This level of mechanistic control ensures that the process is not only chemically efficient but also reproducible on a commercial scale, providing R&D teams with a reliable blueprint for technology transfer.
How to Synthesize 2-Cyano-4'-Methylbiphenyl Efficiently
The synthesis protocol outlined in the patent provides a clear, actionable roadmap for producing high-quality Sartan Biphenyl. The process begins with the careful preparation of the Grignard reagent under anhydrous, nitrogen-protected conditions to prevent hydrolysis. Following this, the coupling reaction is executed with the specific Ni/Mn catalyst loading, followed by a straightforward acidic quench and distillation. This sequence represents a significant optimization over prior art, balancing reaction speed with product quality. For detailed operational parameters, stoichiometry, and safety considerations, please refer to the standardized synthesis guide below.
- Preparation of Grignard Reagent: React p-chlorotoluene with magnesium powder in anhydrous solvents like THF or toluene at 30-80°C under nitrogen protection.
- Catalytic Coupling: React the Grignard reagent with o-chlorobenzonitrile using a Ni(II)/Mn(II) composite catalyst at controlled temperatures between -20°C and 20°C.
- Workup and Purification: Acidify the mixture, perform direct distillation to remove solvents, and recrystallize the crude product to obtain high-purity white crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this Ni-Mn catalytic technology translates into tangible strategic benefits that extend far beyond simple chemical yield. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By eliminating the need for organic solvent extraction—a unit operation that typically consumes vast quantities of solvents and energy for recovery—this process significantly lowers the variable cost per kilogram of the final product. Moreover, the catalyst system itself utilizes Nickel and Manganese salts, which are substantially more abundant and cost-effective than precious metal catalysts like Palladium or Platinum. This shift in raw material sourcing mitigates exposure to the volatile price fluctuations often seen in the precious metals market, ensuring more stable long-term pricing for buyers.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the removal of expensive purification steps. Traditional methods often require chromatography or multiple recrystallizations to remove metal residues and by-products, whereas this novel route achieves high purity through simple distillation and a single recrystallization. The reduction in solvent usage and energy consumption for distillation versus extraction creates a leaner manufacturing profile. Additionally, the high catalytic activity means lower catalyst loading is required to achieve optimal conversion, further driving down the bill of materials. These cumulative efficiencies allow suppliers to offer more competitive pricing structures without sacrificing margin, providing a distinct advantage in cost-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Supply continuity is often jeopardized by complex synthesis routes that are prone to failure or batch-to-batch variability. The robustness of the Ni(II)/Mn(II) catalytic system enhances reliability by tolerating a wider range of operating conditions compared to sensitive palladium systems. The raw materials, p-chlorotoluene and o-chlorobenzonitrile, are commodity chemicals with well-established global supply chains, reducing the risk of raw material shortages. Furthermore, the simplified post-treatment process reduces the turnaround time between batches, allowing manufacturers to respond more agilely to sudden spikes in demand from downstream API producers. This operational agility is crucial for maintaining just-in-time inventory levels and preventing production stoppages.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden engineering challenges, particularly regarding heat management and waste treatment. This patent addresses scalability by utilizing standard industrial solvents like toluene and xylene, which are familiar to plant operators and compatible with existing infrastructure. The elimination of complex extraction steps reduces the volume of wastewater generated, simplifying compliance with increasingly stringent environmental regulations. The ability to recycle the catalyst or easily separate metal residues also contributes to a smaller environmental footprint. For supply chain heads, this means the technology is not just theoretically sound but is practically deployable in large-scale facilities with minimal retrofitting, ensuring a secure and sustainable supply of this critical intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-cyano-4'-methylbiphenyl using this advanced catalytic method. These insights are derived directly from the experimental data and claims presented in the patent literature, offering clarity on process capabilities and product specifications. Understanding these details is vital for technical procurement teams evaluating potential suppliers for long-term partnerships.
Q: What are the primary advantages of the Ni(II)/Mn(II) catalyst system over traditional Palladium catalysts?
A: The Ni(II)/Mn(II) composite catalyst offers a significant cost advantage due to the abundance of nickel and manganese compared to precious metals like palladium. Furthermore, this system demonstrates high activity and selectivity, allowing for easier catalyst recovery and simplified post-treatment processes without compromising yield.
Q: How does this patent address the environmental concerns associated with traditional biphenyl synthesis?
A: Traditional methods often require multi-step protection and deprotection sequences generating substantial waste. This novel approach eliminates the need for organic solvent extraction during workup by utilizing direct distillation after acidification, drastically reducing solvent consumption and three-waste pollution.
Q: Is this synthesis route suitable for large-scale industrial production of Losartan intermediates?
A: Yes, the process is designed for industrial scalability. It utilizes readily available raw materials like p-chlorotoluene and o-chlorobenzonitrile, operates under relatively mild conditions, and achieves high yields (over 87%) with a simple recrystallization purification step, making it highly viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyano-4'-Methylbiphenyl Supplier
The technological advancements detailed in patent CN103467341A represent a significant leap forward in the manufacturing of Sartan Biphenyl, offering a pathway to higher purity and lower production costs. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this Ni-Mn catalytic route are fully realized in our manufacturing facilities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards. We understand that for R&D directors, consistency is key, and our advanced process controls guarantee that the impurity profile remains stable across all production scales.
We invite global pharmaceutical and chemical companies to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this optimized synthesis route, we can help you reduce the total cost of ownership for your ARB drug supply chain. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our engineering expertise and commitment to innovation can become a cornerstone of your supply chain strategy, ensuring you have access to high-quality intermediates when you need them most.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
