Advanced One-Step Synthesis of 2-Cyano-4'-methylbiphenyl for Scalable Sartan Drug Manufacturing
Advanced One-Step Synthesis of 2-Cyano-4'-methylbiphenyl for Scalable Sartan Drug Manufacturing
The global demand for angiotensin II receptor antagonists, commonly known as Sartans, continues to drive the need for efficient manufacturing of their key intermediates. Patent CN102964271B introduces a transformative synthesis method for 2-cyano-4'-methylbiphenyl, a critical building block for drugs like Losartan and Valsartan. This technology addresses long-standing challenges in the pharmaceutical industry by replacing multi-step, hazardous processes with a streamlined, one-step catalytic coupling. By utilizing readily available chlorinated precursors instead of expensive brominated or lithiated reagents, this method offers a robust pathway for reliable pharmaceutical intermediate supplier networks seeking to optimize their production lines. The technical breakthrough lies in the specific palladium catalyst system that activates inert carbon-chlorine bonds under mild conditions, ensuring high purity and yield suitable for stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-cyano-4'-methylbiphenyl has relied on chemically aggressive and operationally complex routes that pose significant barriers to industrial scale-up. Traditional methodologies, such as those described in prior art literature, often involve the use of Grignard reagents or organolithium species which require strictly anhydrous and oxygen-free environments. For instance, one established route utilizes p-bromotoluene reacting with t-BuLi at cryogenic temperatures of -78°C followed by transmetallation with ZnCl2. 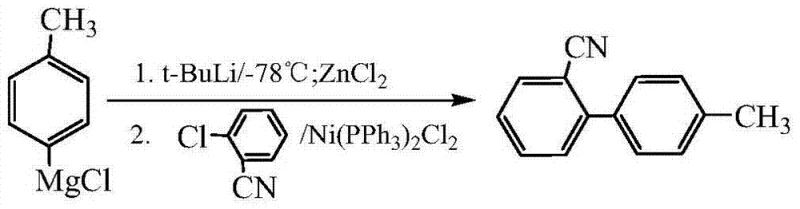 Such extreme low-temperature conditions necessitate specialized refrigeration equipment and result in substantial energy consumption, drastically increasing the operational expenditure for manufacturers. Furthermore, the handling of pyrophoric reagents like t-BuLi introduces severe safety hazards, complicating the supply chain management and requiring extensive safety protocols that slow down production throughput.
Such extreme low-temperature conditions necessitate specialized refrigeration equipment and result in substantial energy consumption, drastically increasing the operational expenditure for manufacturers. Furthermore, the handling of pyrophoric reagents like t-BuLi introduces severe safety hazards, complicating the supply chain management and requiring extensive safety protocols that slow down production throughput.
The Novel Approach
In stark contrast to these legacy methods, the invention disclosed in CN102964271B presents a direct cross-coupling strategy that fundamentally simplifies the manufacturing landscape. The core innovation involves the reaction between o-chlorobenzonitrile and p-chlorotoluene catalyzed by a specialized palladium complex. 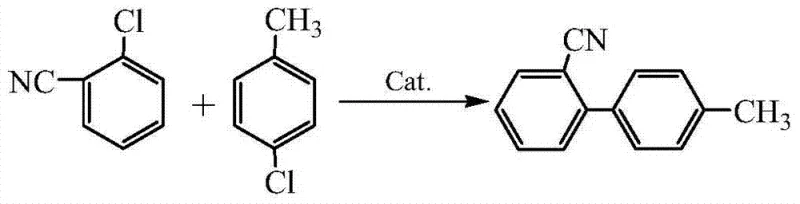 This approach eliminates the need for pre-functionalized organometallic intermediates, allowing the reaction to proceed in a single pot. The process operates at moderate temperatures ranging from 60°C to 90°C, which is easily achievable with standard heating infrastructure found in most chemical plants. By shifting from bromine or lithium-based chemistry to chlorine-based chemistry, the method leverages the significantly lower cost and higher availability of chlorinated starting materials, directly addressing the procurement manager's goal of cost reduction in API manufacturing without compromising on reaction efficiency or product quality.
This approach eliminates the need for pre-functionalized organometallic intermediates, allowing the reaction to proceed in a single pot. The process operates at moderate temperatures ranging from 60°C to 90°C, which is easily achievable with standard heating infrastructure found in most chemical plants. By shifting from bromine or lithium-based chemistry to chlorine-based chemistry, the method leverages the significantly lower cost and higher availability of chlorinated starting materials, directly addressing the procurement manager's goal of cost reduction in API manufacturing without compromising on reaction efficiency or product quality.
Mechanistic Insights into Pd-Catalyzed Chloride Activation
The success of this synthesis hinges on the unique electronic and steric properties of the palladium catalyst employed. Unlike standard palladium catalysts which struggle to activate the strong carbon-chlorine bond, the catalyst described in the patent features a sophisticated ligand environment designed to facilitate oxidative addition. The catalyst structure incorporates a bulky DBCPh-derived ligand system coordinated to the palladium center. 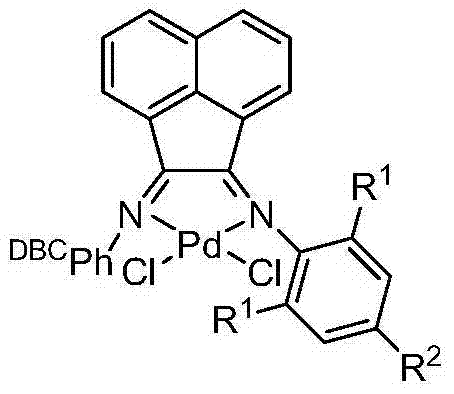 This specific architecture creates an electron-rich metal center that can effectively insert into the C-Cl bond of the chlorotoluene substrate. The mechanism likely proceeds through the formation of a transient palladium-chlorotoluene complex where the chlorine atom is weakened and primed for departure. This activation step is the rate-determining factor in many cross-coupling reactions involving chlorides, and the proprietary catalyst design overcomes this kinetic barrier efficiently.
This specific architecture creates an electron-rich metal center that can effectively insert into the C-Cl bond of the chlorotoluene substrate. The mechanism likely proceeds through the formation of a transient palladium-chlorotoluene complex where the chlorine atom is weakened and primed for departure. This activation step is the rate-determining factor in many cross-coupling reactions involving chlorides, and the proprietary catalyst design overcomes this kinetic barrier efficiently.
Furthermore, the reaction kinetics are heavily influenced by the solvent system, which plays a dual role in stabilizing the catalytic species and solubilizing the reactants. The patent data indicates that a mixture of tetrahydrofuran and pyridine provides a synergistic effect that enhances both conversion rates and selectivity. The nitrogen atom in pyridine likely acts as a weak base or auxiliary ligand, assisting in the stabilization of the palladium intermediate during the catalytic cycle. This precise control over the reaction environment minimizes the formation of homocoupling by-products and ensures that the desired biphenyl linkage is formed with high fidelity. For R&D directors, understanding this solvent-catalyst interplay is crucial for troubleshooting and optimizing the process for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-Cyano-4'-methylbiphenyl Efficiently
Implementing this synthesis requires careful attention to the order of addition and temperature control to maximize the turnover number of the expensive palladium catalyst. The process begins by dissolving p-chlorotoluene in the optimized solvent mixture under an inert atmosphere to prevent catalyst deactivation by oxygen. Once the solution is heated to the target range of 60-90°C, the catalyst is introduced and allowed to equilibrate with the substrate before the electrophile is added.
- Dissolve p-chlorotoluene in an optimized organic solvent mixture such as tetrahydrofuran and pyridine, then add the specialized Pd complex catalyst.
- Heat the mixture to 60-90°C under stirring, then slowly dropwise add o-chlorobenzonitrile over 0.5 to 1 hour while maintaining temperature.
- After reaction completion, cool to room temperature, adjust pH to 6-7 with dilute hydrochloric acid, separate layers, and purify via distillation and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the transition to this catalytic chloride coupling method represents a strategic opportunity to enhance margin stability and supply security. The primary economic driver is the substitution of expensive raw materials; p-chlorotoluene and o-chlorobenzonitrile are commodity chemicals produced on a massive scale, whereas the brominated or lithiated alternatives used in older methods are specialty chemicals with volatile pricing and limited supplier bases. This shift mitigates the risk of raw material shortages and price spikes, ensuring a more predictable cost structure for long-term contracts. Additionally, the elimination of cryogenic steps removes a major bottleneck in batch processing times, allowing facilities to increase their annual output capacity without significant capital investment in new cooling infrastructure.
- Cost Reduction in Manufacturing: The economic benefits extend beyond simple raw material costs to include significant savings in utility and waste management expenditures. By operating at ambient pressure and moderate temperatures, the process reduces the energy load on the plant's HVAC and heating systems. Moreover, the high selectivity of the reaction means fewer by-products are generated, which simplifies the downstream purification workup. This reduction in waste volume lowers the costs associated with solvent recovery and hazardous waste disposal, contributing to a leaner and more environmentally sustainable manufacturing profile that aligns with modern green chemistry initiatives.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of stable, non-pyrophoric reagents simplifies storage and transportation requirements. Unlike t-BuLi, which requires specialized containers and strict temperature control during shipping, the chlorinated starting materials can be handled with standard industrial protocols. This ease of handling reduces the administrative burden on the supply chain team and minimizes the risk of delivery delays caused by hazardous material regulations. The robustness of the reaction also implies a wider operating window, making the process less susceptible to minor fluctuations in utility supply or operator error, thereby guaranteeing consistent delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The simplicity of the one-pot procedure makes it inherently scalable from pilot plant to multi-ton production. The absence of sensitive organometallic intermediates reduces the need for complex quenching procedures that often generate large volumes of saline wastewater. Instead, the workup involves a straightforward acid wash and extraction, generating a cleaner waste stream that is easier to treat. This environmental advantage is increasingly critical as regulatory bodies impose stricter limits on industrial effluent, positioning manufacturers who adopt this technology as preferred partners for environmentally conscious pharmaceutical companies seeking to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of adoption.
Q: Why is the Pd-catalyzed chloride coupling superior to traditional Grignard methods for this intermediate?
A: Traditional methods often require expensive brominated starting materials or harsh cryogenic conditions (e.g., -78°C with t-BuLi). The patented Pd-catalyzed method utilizes cheaper chlorinated raw materials and operates at mild temperatures (60-90°C), significantly reducing energy costs and safety risks associated with cryogenic operations.
Q: What represents the critical innovation in the catalyst system described in CN102964271B?
A: The innovation lies in the specific Pd complex catalyst structure featuring a bulky ligand system (DBCPh derivative). This structure facilitates the activation of the less reactive aryl chloride bonds, enabling high conversion rates and selectivity without the need for expensive activators or extreme conditions.
Q: How does the solvent choice impact the purity of the final 2-cyano-4'-methylbiphenyl product?
A: The patent highlights that solvent polarity and coordination ability are critical. A specific mixture of tetrahydrofuran and pyridine (1:1 to 1:2 ratio) was found to provide optimal conversion and selectivity, minimizing side reactions and simplifying the downstream purification process compared to single-solvent systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyano-4'-methylbiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of Sartan drugs depends on the reliability and quality of their key intermediates. Our technical team has extensively analyzed the catalytic mechanisms described in CN102964271B and possesses the expertise to adapt this laboratory-scale innovation into a robust industrial process. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench to plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-cyano-4'-methylbiphenyl meets the exacting standards required for global pharmaceutical registration.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your specific supply chain. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your position in the competitive antihypertensive drug market.
